Current recommendations for the management of cryptorchidism are for orchidopexy in a timely manner.1–3 Hormonal therapy is not recommended because of limited efficacy and side effects.4 In Australia, orchidopexy is performed for congenital and acquired undescended testes (UDT),4 where the testes are known to be in the scrotum in the first three to six months of age but are found out of the scrotum later in childhood. Acquired UDT is likely to be the result of the spermatic cord not elongating at the same rate as pelvic enlargement with age, and the scrotum becomes further from the groin. The common cause of acquired UDT is persistence of a fibrous remnant of the processus vaginalis (the peritoneal diverticulum within which the testis descends). Retractile testes are now considered by some surgeons as the ‘grey zone’ between normally descended testes and acquired UDT.
Failure to treat congenital and acquired UDT at the right time has an impact on male fertility, and congenital UDT also increases the risk of testicular cancer. Cellular degeneration is a major risk in UDT as the higher inguinal or intra-abdominal temperature (35–37°C) versus that in the scrotum (33°C) inhibits postnatal germ cells from transforming into the stem cells for spermatogenesis at three to six months of age, which is thought to be the cause of long-term infertility and increased cancer risk.5
The incidence of orchidopexy is commonly used as a surrogate for the diagnosis and treatment of cryptorchidism.1–3 Trends regarding international incidence vary according to country, with the incidence in Austria increasing between 1993 and 2009.6 By contrast, Swedish, UK and local Victorian data indicate a decline in operations for UDT (especially where it is acquired) over the past three decades.7–9 Other developed countries have also shown a trend to decreased rates of orchidopexy, and by inference cryptorchidism (congenital and acquired), since the start of the 21st century. Thorup et al10 detailed the worldwide prevalence of congenital cryptorchidism to be around 1–2%, and orchidopexy rates around 2.4–3.8%, consistent with the notion that half of the orchidopexies in childhood are performed on acquired UDT.
Victoria’s Department of Human Services (DHS) data on orchidopexy revealed a decline in the number of orchidopexies performed between 1999 and 2006,9 whereas the proportion of bilateral versus unilateral operations over this period was similar to that reported in the existing literature. This coincided in the early 1990s with the removal of a physical examination from DHS’s primary school entry criteria in the state of Victoria. Currently, there are mandatory examinations at birth, six weeks of age, and between two and four years of age, depending on state/territory.11
Objectives
The aim of our study was to examine 20 years of Australian orchidopexy data, from 1995 to 2014, in order to explore the trends in national operation rates over time, and compare them with previous Victorian studies that showed an overall decline in operation rates between 1999 and 2006.10 We used orchidopexy as a marker for the diagnosis and treatment of UDT on the assumption that operative treatment is the accepted standard.
Methods
Orchidopexy data were collected from the federal Department of Human Services for the calendar years 1994–2014 after ethical approval by the Royal Children’s Hospital (Melbourne) Human Research Ethics Committee (reference number 35176A). Data were extracted using the operation codes for orchidopexies for all patients aged 0–24 years (public and private). Operation data for redo-surgery were excluded, as were scrotal explorations and orchidopexy for indications other than UDT (ie testicular torsion, torsion of a hydatid of Morgagni, hydrocele).
National population data were collected from the Australian Bureau of Statistics (ABS). Census data for each age group were available for years 1996, 2001, 2006 and 2011; demographic data for other years were ABS estimates. The number of orchidopexies per annum was divided by the ABS into three age groups: 0–4, 5–14 and 15–24 years. Each five-year quantum of data was averaged for each age group. A Poisson regression model was fitted to the data for each age group. An incident rate ratio (IRR) indicated the estimated annual (compound) rate of change over the 20-year period to reveal the trends in operation rates related to the whole population. Calculations were performed in Stata 14.
Results
Incidence of orchidopexy operations in Australia
The total number of orchidopexies in Australia over the 20-year period was 27,362. Overall, the incidence of operations per annum has remained constant over the past 20 years, with some decrease between the late 1990s and 2000–04. There was an average of 1816 operations per year between 1995 and 1999, compared with 1433 in the 2000–04 period. This was followed by an increase between 2005–09 and 2010–14 (Figure 1A), with an average of 1481 in 2005–09, compared with 1766 procedures in 2010–14 (Table 1).
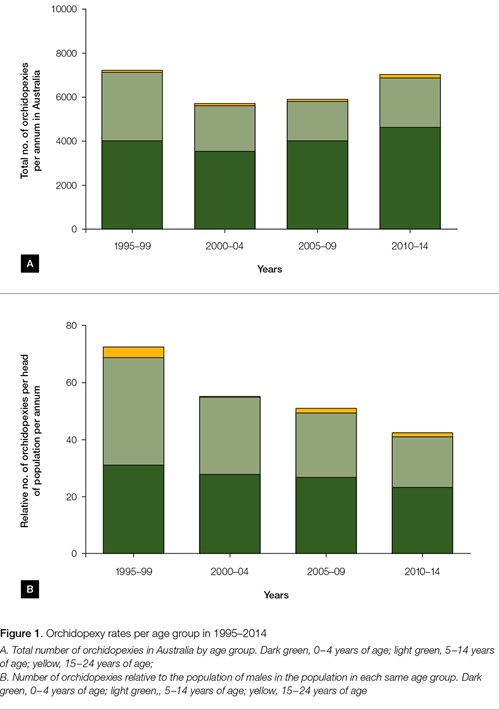
When the operation rates are divided into the three age groups (0–4, 5–14 and 15–24 years), it can be seen that operations in children under the age of five years increased by about 10%, from 1015 per year to 1163. By contrast, in the 5–14 years age group, the frequency of surgery decreased by about 30%, from 775 per year to 564 (Table 1). There were only very small numbers in the 15–24 years age group, precluding analysis.
Table 1. Orchidopexy rates in three age groups. Incidence expressed as average number/year for each five-year period
|
Incidence of orchidopexy
|
0–4 years of age
|
5–14 years of age
|
15–24 years of age
|
Total
|
|---|
|
1995–99
|
1015
|
775
|
27
|
1817
|
|
2000–04
|
889
|
519
|
25
|
1433
|
|
2005–09
|
1009
|
441
|
31
|
1481
|
|
2010–14
|
1163
|
564
|
40
|
1767
|
|
Total
|
4076
|
2299
|
123
|
6498
|
|
Incidence of orchidopexy relative to the total population in each age group
The number of orchidopexies in each age group was divided by the total population of boys in each age. This shows a progressive decrease in the number of orchidopexies per head over the 20-year period in the 5–14 years age group. By contrast, the number of orchidopexies per head of population in the 0–4 years age group remained constant (Figure 1B).
Meanwhile, the population of males aged 0–24 years increased from 3.4 million in the 1995–99 period to 3.9 million in the 2000–04 period. This means that in 1995, there were 0.66 million boys under the age of five years, compared with 0.8 million in 2014. In the 5–14 years age group, there were 1.32 million boys in the 1990s, compared with 1.49 million in 2014. In the 15–24 years age group, there were 1.37 million males in the 1990s, compared with 1.6 million in 2014.
Using a Poisson regression model for each age group (Figure 2), the IRR indicates the estimated annual (compound) rate of change over time. For boys under the age of five years, there was no change in rate. For the 5–14 years age group, there was a decrease in the rate of orchidopexy operations (P <0.005), with an average decline of 2.9% per year. For adolescent and young adults (15−24 years of age), there was an increase of 2% (P: 0.01).
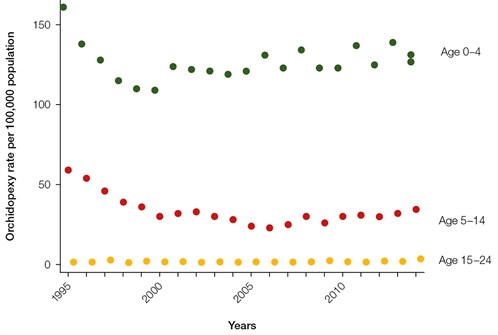
Figure 2. Poisson regression model graphing relative incidence of orchidopexies in each age group divided by the total of boys in each age group
Green, 0–4 years of age; orange, 5–14 years of age; yellow, 15–24 years of age
Discussion
This study found that the overall incidence of orchidopexy in Australia has remained relatively constant over the past 20 years. Compared with the general population, there was no change in the 0–4 years age group. By contrast, there was a decrease of 2.9% per annum over 20 years in the incidence of orchidopexy in boys aged 5–14 years, and an increase of 2% per annum over the 20 years in adolescents and young men (15−24 years of age).
The constant rate of orchidopexy in the 0–4 years age group is likely to reflect effective neonatal and early infant screening over the 20-year period. Neonates with no testis in the scrotum need to be re-examined at three months of age to see if there is any postnatal descent. For half of those with congenital UDT at birth, their testes spontaneously descend by 12 weeks of age and they will not require surgical intervention.4 Congenital UDT, which persists after three months of age, should be referred immediately for paediatric surgical assessment, aiming for orchidopexy between six and 12 months of age.1–3 Timely orchidopexy (at six to 12 months of age) is thought to preserve fertility and reduce the risk of testicular carcinoma in this age group when they become young adults.1–3 Bilateral testicular UDT (especially if neither testis is palpable) should have an urgent referral to a paediatric urologist or specialist for consideration of disorders of sex development (DSD).12
The rate of orchidopexies in boys aged 5–14 years has dropped by 30% over the past 20 years. This is consistent with decreasing intervention for putative ascending testes that have developed into acquired UDT (Figure 3). This second peak of surgery should occur later in childhood, between five and nine years of age. Initially, these boys may have a ‘retractile’ testis, which can be pulled into the scrotum and remains there. However, these cases should be reviewed in 6–12 months to ensure there is no deterioration in testicular position. Once the testes cannot remain in the scrotum, or if the testes are still high after a six-month observation period, paediatric surgical review is indicated. The decrease in numbers in this group suggests that many boys are not being diagnosed with acquired UDT or, alternatively, a decision has been made not to operate.
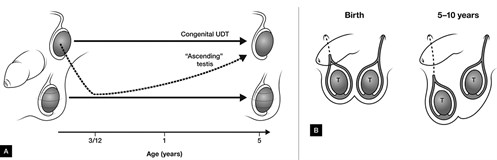
Figure 3. Differences between congenital and acquired undescended testes (UDT)
A. Presentations of congenital versus acquired UDT. The testis descended in infancy assumes a higher position later in childhood in acquired UDT, while congenital UDT is never in the scrotum;
B. Schematic demonstrating the length of the spermatic cord in cases of acquired UDT. At birth, the processus vaginalis has disappeared on the right (dotted line), but persisted (often as a fibrous remnant) on the left. Usual growth of the spermatic cord is emonstrated by a dashed line on the right, with the testis located in the scrotum.
Failure of the spermatic cord to elongate with age (because of the fibrous remnant of the processus vaginalis) is shown on the left side, with an ascending testis
The increasing number of patients aged 15–24 years who undergo orchidopexy may represent those with either missed diagnosis or inappropriate conservative management, or an increasing migrant population where no surgery was performed in their home country.
Whether the decline in surgery for acquired UDT is due to failure of detection, failed referral or failed follow-up is unknown. Data from Europe suggest we should be operating on 1–2% of boys overall,13 providing our care is equal to other developed countries. In most countries, acquired UDT is usually treated by operative intervention, once the testes no longer remain in the scrotum. The exception is in the Netherlands, where there is controversy regarding the management of acquired UDT. At present, many Dutch paediatric surgeons are not operating on boys with acquired UDT until they have reached puberty.14,15 Our recommendation is for operation once the testes no longer reside spontaneously in the scrotum.4
Limitations
Limitations to these data include the assumption that an appropriate number of orchidopexies were performed 20 years earlier. If too many cases of UDT were managed inappropriately with orchidopexy, the results in this study could be skewed. The overall incidence of UDT is also presumed to be the same as that in other developed countries. Given the limitations of privacy with DHS, these data could not be broken down into smaller age groups. This limits the data interpretation, and we cannot examine whether the 0–4 years age group were being operated on at younger ages, or when the 5–14 years age group were undergoing orchidopexy.
This study suggests that boys aged 5–14 years may not be receiving treatment for ascending, acquired UDT. The natural history of acquired UDT is that the testes are in the scrotum at six months of age, but ascend out of the scrotum between one and five years of age.16 From about five years of age, the testes are located in the groin, but then spontaneously descend again at puberty (thought to be a secondary effect of androgen).17 However, if the testis has been non-scrotal for most of the boy’s childhood, temperature-dependant damage to testicular function will have probably already occurred, and these boys are likely to have suboptimal fertility.18 The removal of formal screening for UDT on school entry leaves primary care as the only regular (informal) screening point, and puts the onus on general practitioners to actively screen boys around the time of school entry for acquired undescended testis.
Implications for general practice
- Examination for congenital UDT should be at birth and three months: refer persisting UDT at three to six months of age.
- To avoid missing the diagnosis of acquired UDT, scrotal exams should be performed yearly between one and three years of age, and two-yearly from age three to 10 years.
- Persisting UDT in a previously descended testis (more than six months) should be referred to a paediatric surgeon, as the boy may be developing acquired UDT.
- Failure of diagnosis and treatment of congenital and acquired UDT may result in infertility.
Authors
Jaya Vikraman MBBS, Diploma of Surgical Anatomy, Diploma of Child and Adolescent Health, PhD student, University of Melbourne, Douglas Stephens Surgical Research Laboratory, Murdoch Childrens Research Institute, Parkville, Vic; Royal Children’s Hospital, Parkville, Vic; Department of Paediatrics, University of Melbourne, Parkville, Vic. jaya.vikraman@mcri.edu.au
Susan Donath BSc (Hons), MS Women’s Studies (Hons), Senior Biostatistician, Clinical Epideminology and Biostatistics Unit, Murdoch Childrens Research Institute, Parkville, Vic; Department of Paediatrics, University of Melbourne, Parkville, Vic
John M Hutson AO, BSMD, MD, DSc, FRACS, FAAP, Professor, Chair of Paediatric Surgery, Douglas Stephens Surgical Research Laboratory, Murdoch Children’s Research Institute, Parkville, Vic; Royal Children’s Hospital, Parkville, Vic; Department of Paediatrics, University of Melbourne, Parkville, Vic
Competing interests: None.
Provenance and peer review: Not commissioned, externally peer reviewed.