Case
A Moroccan man, 45 years of age, was referred to the dermatology department because of the progressive occurrence of asymptomatic cutaneous lesions on his hands and feet during the past three years. He had received several topical treatments, including corticosteroids, antifungals and antibiotics, but with no response. Physical examination revealed the presence of purplish-red, infiltrated plaques, symmetrically located only on the dorsum of proximal and distal interphalangeal joints of both hands (Figure 1A). The patient also had a pronounced hyperkeratosis on his palms and soles (Figure 1B), oral hyperpigmentation (Figure 2A) and nail changes in the form of longitudinal melanonychia (Figure 2B). He had been diagnosed with essential thrombocytosis, and had initiated treatment with oral hydoxyurea one year before the onset of the skin manifestations.
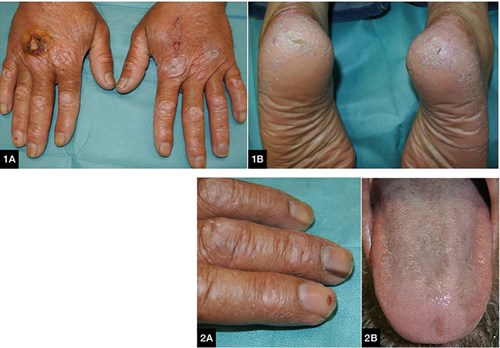
Figure 1. A. Purplish-red infiltrated plaques on the dorsum of proximal and distal interphalangeal joints of both hands; B. Cracked hyperkeratosis on both heels
Figure 2. A. Fingernails with dark brownish longitudinal bands; B. Patched tongue hyperpigmentation
Question 1
Considering the whole dermatological picture, how would you investigate this patient?
Answer 1
The clinical morphology of acral lesions, mimicking the dermatomyositis traditionally known as ‘mechanic’s hands’, along with the other mucocutaneous manifestations in the context of a haematological disease, is highly suggestive of cytostatic-induced cutaneous toxicity. Therefore, a detailed medical history and complete physical examination of the entire skin and mucosal surfaces are the most important investigations to achieve a final diagnosis in this case.
Hydroxyurea is a cytostatic agent currently used in clinical practice to treat myeloproliferative disorders, sickle-cell disease and few cases of refractory psoriasis. Myelosuppression, teratogenicity and hepatotoxicity are the most common side effects observed with the drug, although dermatological events have been described in up to 30% of cases.1,2
Case continued
The patient´s work-up was completed with an examination to assess muscle weakness and levels of muscle enzymes, and an electromyographic study to exclude muscular involvement. An extended autoimmunity panel was performed and results were negative. Finally, systemic manifestations were excluded by performing laboratory tests, pulmonary function tests and electrocardiography study.
Question 2
What is the main differential diagnoses?
Answer 2
Differential diagnoses should necessarily include amyopathic dermatomyositis (Table 1). Similarly to this, dermatomyositis-like eruptions secondary to hydroxyurea are characterised by the presence of acral erythematous lesions over the dorsal region of interphalangeal joints (Gottron’s papules), along with a marked hyperkeratosis on the palms and soles, without signs of muscular involvement. Both features are clinically indistinguishable from cutaneous manifestations of the classical disease. However, patients with cytostatic-induced dermatomyositis rarely develop other clinical features traditionally linked to dermatomyositis (holster sign and periungual abnormalities) and photodistributed rashes (heliotrope erythema, shawl and ‘V’ signs). In addition, although positive autoimmunity and systemic complications are common findings in the course of amyopathic dermatomyositis, they are typically absent when the disorder appears related to hydroxyurea.3–5
Table 1. Differential diagnoses: Differences between hydroxyurea-induced cutaneous toxicity and dermatomyositis
|
|
Classical dermatomyositis
|
Hydroxyurea-induced dermatomyositis eruptions
|
|---|
|
Age of onset
|
Fifth–sixth decade of life
|
Sixth–seventh decade of life
|
|
Cutaneous findings
|
Gottron’s papules
Heliotrope eruption
Facial erythema
Shawl sign
‘V’ sign
Holster sign
Palmoplantar keratoderma
Periungueal abnormalities
|
Gottron’s papules
Palmoplantar keratoderma Longitudinal melanonychia Onychodystrophy, xerosis or icthyosis Painful leg ulcers
Oral patched hyperpigmentation
|
|
Systemic symptoms
|
Cardiac, pulmonary, gastrointestinal, renal and/or peripheral vascular involvement
|
Extremely rare
|
|
Autoimmunity
|
Positive
|
Negative
|
|
ESR, CRP
|
High levels
|
Normal limits
|
|
Need for immunosuppressive therapy
|
Yes
|
No
|
|
Improvement with drug withdrawal
|
No
|
Yes
|
|
Need for screening malignances
|
Always
|
Not necessary (only if suspect)
|
|
CRP, C-reactive protein; ESR, erythrocyte sedimentation rate
|
Case continued
An extended autoimmunity panel, including antinuclear antibodies (SSA/Ro antibodies, SSA/Ro52 antibodies, SSB/La antibodies, double-stranded DNA, antiRNP, antiRNP-A, antiRNP 68, anti-topoisomerase antibodies, antiJo1, antiSM antibodies and antiSM-RNP antibodies), anti-cromatine, antiMi2 and complement molecules C3 and C4 was performed. All results were negative for these antibodies. Inflammatory markers, creatine kinase levels and thyroid function tests were normal. Serological status was negative for human immunodeficiency virus, hepatitis B and C viruses, and syphilis.
The patient did not show any muscle weakness on physical examination. He denied the presence of myalgia and motor limitation, and there were no signs of synovitis or joint swelling. Electromyography showed normal electrical activity in the muscle tissue, and serum levels of creatine phosphokinase and aldolase were normal.
Pulmonary and renal function tests, as well as an electrocardiography studies, were performed to rule out systemic involvement, which can appear in the idiopathic forms. All of these tests were normal.
Question 3
How would you confirm the diagnosis?
Question 4
What is the most appropriate management?
Question 5
What are the most common skin disorders related to this diagnosis?
Answer 3
Diagnosis of hydroxyurea-induced cutaneous toxicity is mainly based on the co-existence of compatible clinical findings and a positive history of drug intake. In these cases, a careful physical examination that includes mucosal surfaces and nails is mandatory to identify other cutaneous manifestations linked to the use of hydroxyurea. In this patient, the concomitant presence of palmoplantar keratoderma, oral hyperpigmentation and longitudinal melanonychia is suggestive of hydroxurea-induced cytotoxicity as the diagnosis. However, hydroxyurea-induced dermatomyositis must be excluded.
A plausible time relationship between hydroxyurea administration and the onset of dermatological manifestations, and an improvement in the skin lesions after drug withdrawal are clues to making the diagnosis.
A skin biopsy of the rash may be helpful to rule out other specific entities, but there are no definite histopathological features associated with the diagnosis of hydroxyurea-induced dermatomyositis. Hyperkeratosis with epidermal atrophy, vacuolar degeneration of the basal cell layer and moderate dermal mononuclear perivascular inflammatory infiltrate may be equally found in skin biopsies of both idiopathic dermatomyositis and dermatomyositis-like eruptions.6 In this patient, microscopic examination of the rash showed non-specific changes consistent with a remarkable hyperkeratosis, along with a mild superficial perivascular infiltrate.
Answer 4
Although mild cases can improve with symptomatic topical treatments, such as potent or hyper-potent corticosteroids, dermatomyositis-like eruptions secondary to hydroxyurea usually have a poor response and require removal of the drug for complete resolution.7 Cutaneous lesions observed in this patient did not change with topical steroid therapy, so hydroxyurea was finally switched to melphalan therapy. Six months after hydroxyurea was withdrawn, cutaneous lesions showed a notable improvement with only topical emollients.
To our knowledge, there are no cases reporting a link between drug-induced dermatomyositis and malignancy. Therefore, the presence of dermatomyositis-like dermatological manifestations triggered by a plausible drug avoids unnecessary malignancy screening in those patients.
Answer 5
Cutaneous side effects in patients following hydroxyurea treatment are described in 10–35% of cases subjected to long-term therapy (Table 2).8,9 The skin is the largest visible organ of the body and thus physicians must recognise the clinical spectrum of cutaneous drug reactions related to the use of hydroxyurea. Knowing how to properly read the clinical signs in the skin is essential to minimise unnecessary complementary investigations and correctly direct the treatment approach.
Table 2. Cutaneous adverse effects of hydroxyurea
|
Hair
|
Alopecia
|
|---|
|
Nails
|
Longitudinal melanonychia
Onychodystrophy
|
|
Skin lesions
|
Xerosis or icthyosis
Cutaneous hyperpigmentation
Skin atrophy
Painful leg ulcers
Acral erythema
Palmoplantar keratoderma
Allergic vasculitis
Dermatomyositis-like eruptions
Actinic keratosis
Squamous cell carcioma
|
|
Mucous membranes
|
Oral patched hyperpigmentation
Oral ulceration
|
Authors
Alba Calleja Algarra MD, Dermatologist, Dermatology Department, 12 de Octubre University Hospital, 28041 Avenida de Córdoba, S/N, Madrid, Spain. albacallejaalgarra@gmail.com
Raquel Aragón Miguel MD, Dermatologist, Dermatology Department, 12 de Octubre University Hospital, 28041 Avenida de Córdoba, S/N, Madrid, Spain
Fátima Tous Romero MD, Dermatologist, Dermatology Department, 12 de Octubre University Hospital, 28041 Avenida de Córdoba, S/N, Madrid, Spain
Lidia Maroñas Jiménez, MD, Dermatologist, Dermatology Department, 12 de Octubre University Hospital, 28041 Avenida de Córdoba, S/N, Madrid, Spain
Competing interest and funding: None.
Provenance and peer review: Not commissioned, externally peer reviewed.