Case
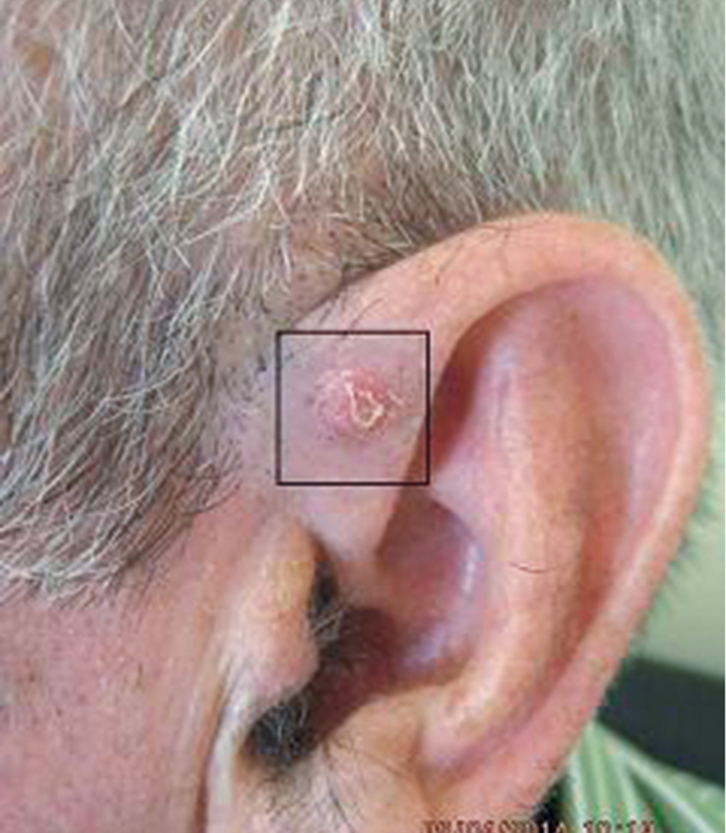
Mr JT, 63 years of age, had a long history of non-melanoma skin cancers (NMSCs). A suspicious skin lesion 6 mm in diameter was noted on his left ear during a routine review (Figure 1). The lesion was photographed and a 3-mm punch biopsy was performed. The subsequent histology report noted a nodular and infiltrating basal cell carcinoma (BCC) with a thickness of 2.3 mm and no perineural or vascular invasion.
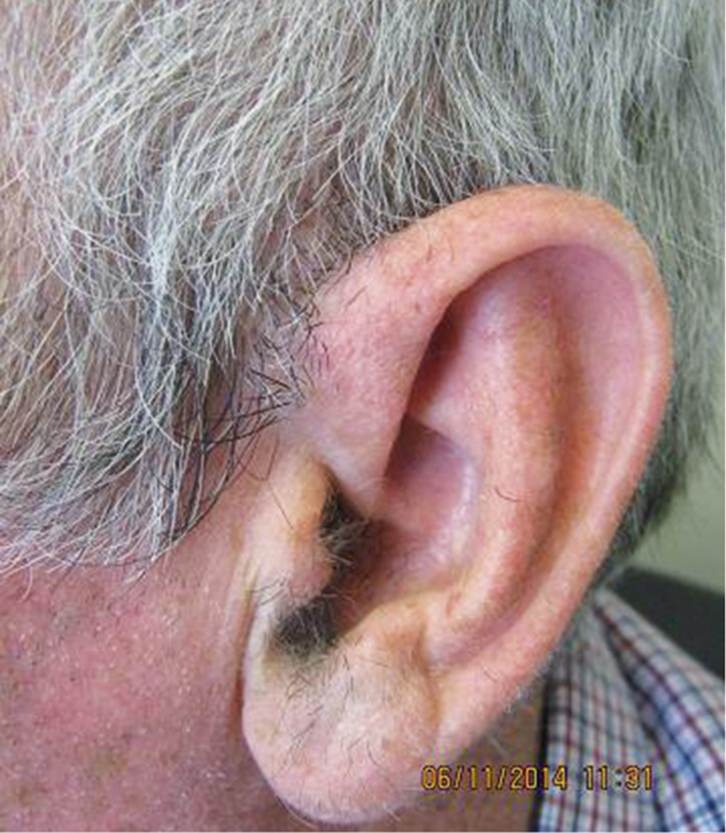
The results of the biopsy were discussed with Mr JT and a date for excision was arranged. However, by the time of the appointment, 2 months after the biopsy, the lesion had completely clinically regressed (Figure 2).
Mr JT felt the tumour had disappeared and was reluctant to have any further treatment ‘unless necessary’. He chose, instead, to have the area observed.
 |
 |
| Figure 1. Skin lesion on the left ear |
Figure 2. Skin lesion 2 months after punch biospy |
Question 1
Why had the lesion regressed?
Question 2
What is the significance of the BCC having an infiltrating component?
Question 3
What management options are available in this situation? What pros and cons are associated with each of these options?
Question 4
What is the optimal management modality in this case?
Answer 1
Regression of NMSCs following biopsy is not uncommon and is thought to be due to the host’s immune response.1
Answer 2
The most common types of BCC are nodular and superficial. Infiltrating BCC is considered to be an ‘aggressive’ subtype, along with micronodular and morphoeic BCCs. Aggressive subtypes tend to have fewer clinical signs and can spread widely at the site of the tumour before becoming clinically apparent. This usually results in the need for larger and more disfiguring procedures to achieve a cure. Aggressive subtypes also have a greater risk of incomplete excision and recurrence.2
A lesion such as this on the ear is particularly challenging, given the importance of the ear as a cosmetic structure and the potential complexity involved in repairing an excision defect in this location (eg skin grafts and/or flaps).
Answer 3
A number of treatment options are available in this situation:
- close observation for signs of clinical recurrence
- re-biopsy of the area
- excision of the area
- referral for Mohs surgery
- referral for radiotherapy.
It should be noted that photography of the lesion and biopsy site(s) has a pivotal role in determining the exact site to observe or to excise, or to plan radiotherapy.
Observation
Close observation of the area is the least invasive approach, although it is fraught with difficulties. A study in 1999 showed that in 42 consecutive cases where there was no clinical evidence of remaining tumour following BCC biopsy, subsequent excision revealed residual tumour in 66% of the cases.3 Furthermore, the BCC (particularly aggressive subtypes) may become extensive before becoming clinically apparent and require complicated and/or disfiguring treatment. Clinical recurrence may occur years later, where there is the risk of the patient having been lost to follow-up. However, observation is a reasonable approach in the frail or elderly who are unable to tolerate treatment, or who have a limited life expectancy.
Re-biopsy
The area may be re-biopsied. If the second biopsy is positive for residual BCC, this may prompt the patient and general practitioner (GP) to proceed to a more definitive management. However, false-negative rates are significant.
A clinical trial in 1992 found a false-negative rate of 63% when performing a second biopsy to determine if there was residual tumour after tumour regression following the initial biopsy.4 The second biopsy was substantial in this trial, and involved shave biopsy of the initial biopsy site and surrounding skin. It had a depth to the reticular dermis and width of ≥5 mm in 80% of the cases. It should be noted, however, that this study examined a cohort of ‘high-risk patients’ referred for Mohs surgery and may not accurately reflect the average patient seen in general practice.
When an area is biopsied, it is important to consider suturing the biopsy site and leaving the suture in place until the histopathology results are available or until the time of surgery. This will allow easy location of the biopsy site.
Excision
Given the location and type of lesion, excision of the area is a favourable option in terms of cure rate. However, as there are no clinical signs to define the tumour boundaries, there is an increased risk of incomplete tumour excision (if the tumour is still present). Repair could be complicated as it requires a skin flap or graft. The patient’s reluctance to have ‘unnecessary’ surgery could also pose a dilemma if there was no evidence of residual tumour in excised sample.
Mohs surgery
Mohs surgery is the gold standard for aggressive or poorly defined tumours in cosmetically sensitive areas. It offers the best cure rate (99%, compared with 95% with conventional excision). It also requires the least amount of tissue removal, which allows for a better cosmetic outcome. However, Mohs surgery is expensive, with significant out-of-pocket costs for the patients.
Radiotherapy
Radiotherapy is a good option for patients who refuse or who are unable to tolerate surgery. However, radiotherapy results in scarring, skin atrophy and telangiectasia, which worsens over time. It is therefore not usually a first-line treatment option in younger patients. It is also only available in larger centres and requires multiple treatments over a prolonged period of time.
Answer 4
The optimal management modality in this case is to provide the patient with the available options, explain the pros and cons of each, then allow the patient to make a fully informed decision. Having family involved in the decision making, with the patient’s consent, may be helpful. Good record keeping and having systems in place to ensure continuity of follow-up are paramount.
Outcome
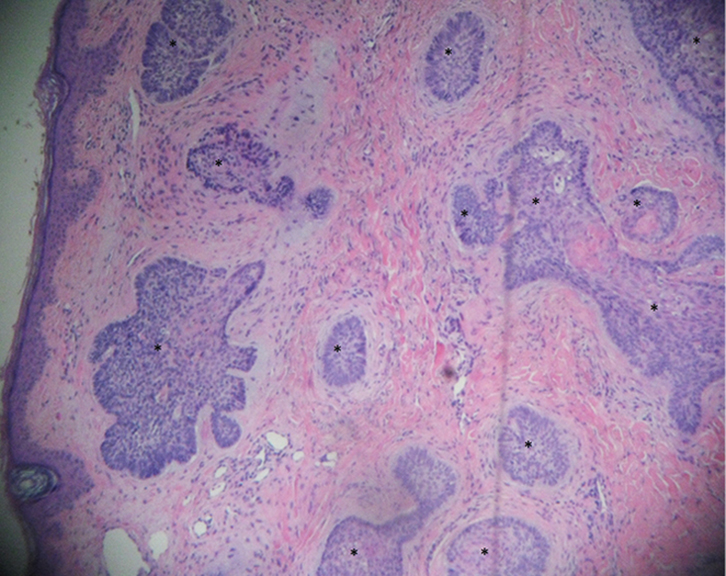 |
| Figure 3. Punch biopsy – micronodular BCC |
In this case, there was still no clinical evidence of recurrent tumour after 7 months of observation. However, using a dermatoscope, an area suspicious for BCC was detected. Numerous studies have noted the typical features of BCC under the dermatoscope. Mr JT was strongly advised to undergo, at least, a second biopsy using the dermatoscopic findings as a guide. A 3-mm punch biopsy was performed and showed definite micronodular (aggressive growth pattern) BCC (Figure 3). Mr JT was referred for Mohs surgery.
Mohs surgery was recommended as the lesion was not clinically visible, had an aggressive subtype and was in an area of cosmetic importance. It is worth noting that the initial biopsy was diagnosed as having an infiltrative component, whereas the second biopsy was diagnosed as having a micronodular component. These discrepancies are not uncommon and are often attributable to heterogeneity of the lesion and/or differing interpretation between histopathologists.
Conclusion
Managing a situation where an NMSC regresses after biopsy can be difficult. The tumour may still be present despite the absence of clinical features. Relying on observation may allow the tumour to progress before being visible, and a second biopsy of the area may only serve to add false reassurance. The GP may negotiate this difficult territory by:
- carefully informing the patient of the pros and cons of the various options
- keeping thorough records of discussions and treatments
- having robust systems in place to ensure strict follow-up of the patient.
Author
Steven Tomas MBBS (Hons), MMed, FRACGP Clinical Lecturer, University of Sydney, and General Practitioner, Gumtree Medical Practice, Dubbo, NSW. stomas101@australiaonline.net.au
Competing interests: None
Provenance and peer review: Not commissioned, externally peer reviewed.