Case
A man, aged 59 years, with a 30‑year history of genotype 1 hepatitis C, mild cirrhosis and a past history of asthma requiring combination therapy, presented to his general practitioner (GP) complaining of insomnia, fatigue and muscle weakness, which caused him to fall when climbing stairs. Recently, he had received treatment with a newly available antiviral regimen for hepatitis C eradication.
On examination, the patient was found to have centralised adiposity, moon facies (Figure 1) and a ‘buffalo hump’. The skin on his arms was thin and hairless, there was evidence of easy bruising, and several eccymoses in different stages of resolution were present. His abdomen was distended and had purple striae, but there was no shifting dullness. Proximal muscle weakness was demonstrable at the hip flexors. He had hypertension and a significant postural drop. There was mild pitting oedema at the ankles. Chest auscultation revealed vesicular breath sounds with no wheeze or basal crackles.
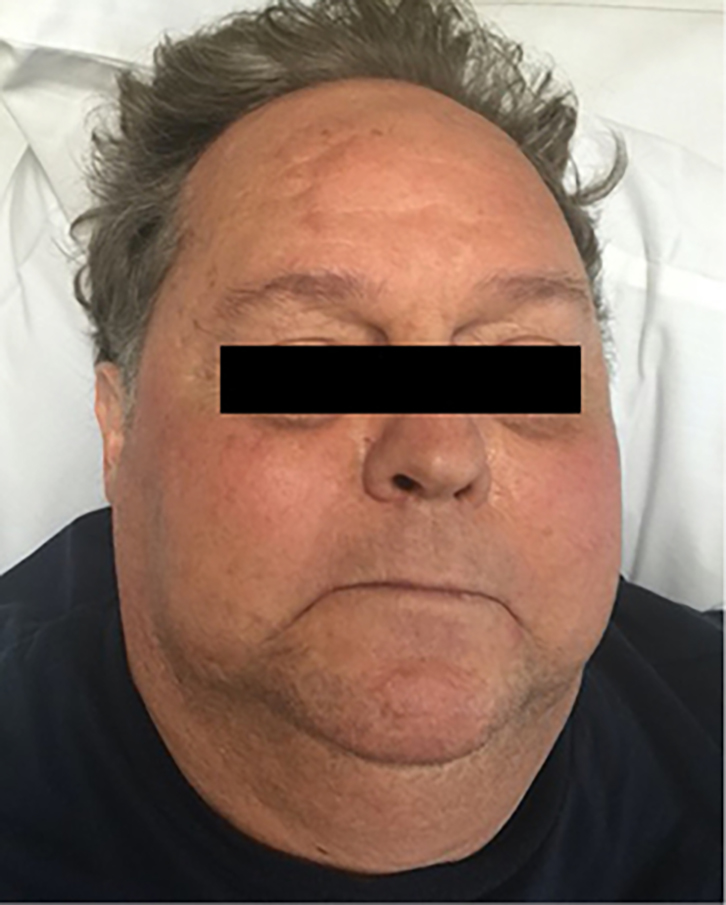
Figure 1. Moon-shaped facies
The patient was adherent to a medication regime that included fluticasone/salmeterol 250/50 μg and a combination antiviral containing dasabuvir, ombitasvir, paritaprevir and ritonavir, along with ribavirin.
Early morning urine and serum cortisol levels were measured. The results are shown in Table 1.
Table 1. Urine and cortisol levels
|
Test
|
Result
|
Reference range
|
|
Serum cortisol
|
112 nmol/L
|
120–620 nmol/L (morning)
|
|
24-hour urine free cortisol
|
<p"><72 nmol/24 hours |
100–330 nmol/24 hours
|
Question 1
Which syndrome explains this constellation of signs and symptoms?
Question 2
How do you interpret the cortisol results?
Question 3
What is the cause of steroid excess?
Answer 1
The best explanation for the signs and symptoms in this patient is Cushing’s syndrome.
Answer 2
On examination of the results in Table 1, it is important to note that endogenous cortisol production is low, which suggests suppression of the hypothalamic–pituitary–adrenal axis.
Answer 3
The combination of clinical signs of steroid excess and low endogenous steroid suggest an iatrogenic Cushing’s syndrome.
Discussion
Iatrogenic Cushing’s syndrome has been reported to occur with the co‑administration of inhaled steroids and protease class antivirals. In particular, the combination of fluticasone and ritonavir has been observed to cause iatrogenic Cushing’s syndrome in adults and children undergoing treatment for human immunodeficiency virus (HIV).1
In this case, it is thought that ritanovir interacted with fluticasone by saturating the CYP3A4 pathway shared by both drugs.1 The effect may have been increased because of reduced hepatic capacity related to the patient’s mild cirrhosis. The patient went on to experience complications of Cushing’s syndrome and asthma, which required admission to an intensive care unit (ICU) and prolonged rehabilitation before he was able to return to the community. Recent advances in, and availability of, outpatient hepatitis treatment and its funding in Australia mean that many general practice patients are likely to be exposed to antiviral medications. Given the prevalence of inhaled steroid use in general practice populations, interactions are likely to occur. Early recognition and management of patients with drug–drug interactions is a key role of the GP in ensuring the safe use of antiviral medications.
Key points
- Cushing’s syndrome presents with characteristic clinical signs and symptoms that are illustrated in this case.
- Protease inhibitors may prevent the metabolism of medications by inhibition of CYP3A4, which may led to unwanted effects.
- An early morning urinary cortisol test is useful in distinguishing between endogenous and exogenous steroid excess.
- Clinical vigilance needs to be used to detect side effects early.
- Use of minimal effective doses of inhaled and intranasal steroids should be considered to minimise the risk of drug–drug interactions with antivirals.
- The combination of ritonavir and fluticasone should be avoided where possible.
Author
George Marshall BA, MBBS (Hons), FRACGP, GradDip Uni Teaching, Adjunct Lecturer, School of Medicine, University of Notre Dame Australia, Darlinghurst, NSW; General Practitioner, Rozelle Total Health, Rozelle, NSW. george.marshall@nd.edu.au
Competing interests: None.
Provenance and peer review: Not commissioned, externally peer reviewed.
References