Case
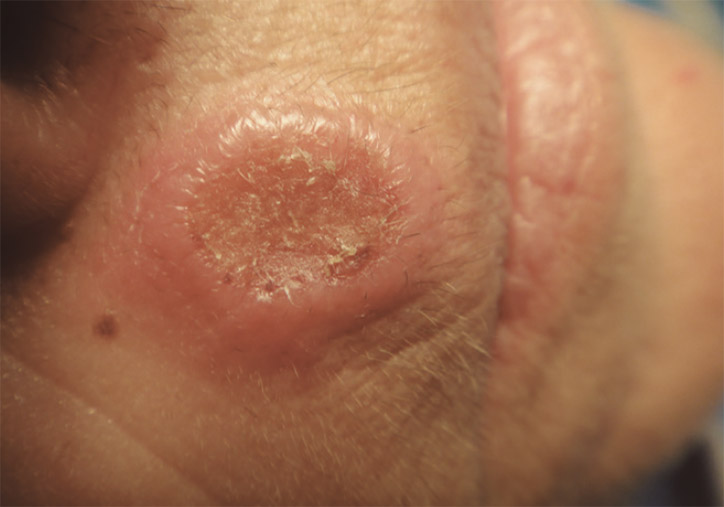
Figure 1. Asymptomatic ulcerated nodule localised above the upper lip with central crusting. The lesion had grown rapidly over a 3-week period
A woman aged 56 years presented with a nodule on her upper lip, which had rapidly evolved to central ulceration with crusting (Figure 1). Her medical history showed no relevant data. She was a housewife, living in the countryside in the Mediterranean southeast coast of Spain, and had a pet dog. No history of recent travels or sexual-risk practice was reported. She did not recall any signs of insect bite at the onset of the lesion. Within a few days of onset, the skin in the centre of the lesion turned darker and started to ulcerate. No pain or pruritic sensation was reported; however, the associated aesthetic disturbance was her main concern. No systemic symptoms, such as fever, lymphadenopathy, abdominal pain or asthenia, were found.
Question 1
What are the main differential diagnoses to be considered in this case?
Question 2
What are the main epidemiological features of localised cutaneous leishmaniasis?
Question 3
What are the main epidemiological features of keratoacanthoma?
Question 4
What are the shared clinical features of localised cutaneous leishmaniasis and keratoacanthoma?
Question 5
Which complementary test should be done in this case?
Question 6
What would be your diagnosis?
Question 7
What should be the primary therapeutic approach?
Question 8
There are other therapeutic options for kerathoacanthoma?
Answer 1
Differential diagnoses include, among other skin lesions, localised cutaneous leishmaniasis, keratoacanthoma, syphilitic chancre, deep fungal infection, mycobacterial infection, sarcoidosis and squamous cell carcinoma. In considering the diagnosis, we narrowed the possibilities to localised cutaneous leishmaniasis and keratoacanthoma. There are several similarities between these two entities and the epidemiological background could be the differentiating factor.
Answer 2
Leishmania is an intracellular protozoan parasite infection spread by a sandfly vector and has an estimated prevalence of 12 million people worldwide and an incidence of 1.5–2 million new cases each year.1 Rodents and dogs are the principal reservoir with geographical variations. In endemic areas of the Mediterranean basin, up to 20% of dogs are infected.1 Our patient lives in the Mediterranean basin, in the countryside where there are probably rodents and stray dogs nearby. In addition, she had a pet dog.
Answer 3
Keratoacanthoma is a relatively common skin tumour derived from the pilosebaceous glands, which shows low-grade malignancy, unpredictable biological behaviour and anecdotal cases of metastases. Sun exposure, chemical carcinogens such as tar, human papillomavirus infection and inmunocompromised conditions have been implicated in the aetiology of keratoacanthoma.2 Our patient presented the epidemiological features of being white-skinned, middle-aged and having chronic sun exposure. Signs of actinic damage, such as increase of skin lines and solar lentigines, were observed on her face.
Answer 4
Sun-exposed areas such as the head and limbs are the predilection sites for the development of keratoacanthoma or localised cutaneous leishmaniasis. The typical clinical manifestation of both conditions may be a pinkish-red nodule with a volcanic shape, a rolled border and a central crater. The lesion starts with a reddish papule, grows rapidly and progressively ulcerates in the centre. Pain or tenderness could be present during the natural evolution.3
Answer 5
Skin biopsies and microbiological samples are needed to complete the diagnostic process. Skin scrapings were obtained from the base of the ulcer and two 4-mm punch biopsies were taken from the edges. The sensitivity of direct visualisation of Leishmania in touch preparations and in tissue samples is estimated to be 75–85%. If there is a high suspicion of Leishmania and the results of other tests are not conclusive, other diagnostic tests such as polymerase chain reaction or in vitro cultures may be performed. In this case, no identification of Leishmania was made through microbiological testing. The histopathology section is shown in Figure 2.
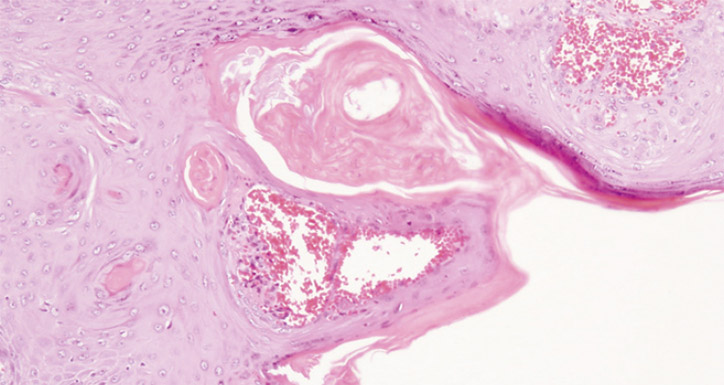
Figure 2. Well-differentiated squamous domized trial. Actas Dermosifiliogr 2011;102:605–15. epithelium exhibiting mild degree of pleomorphism with scattered keratin masses (hematoxylin-eosin staining, original magnification 10x)
Answer 6
The absence of intracellular protozoa in the microbiological samples and histological findings compatible with keratoacanthoma led to the definitive diagnosis of keratoacanthoma.
Answer 7
Keratoacanthoma is classified as a low-grade malignancy variant of squamous cell carcinoma (SCC) that has unpredictable biological behaviour. The risk of disfiguring scars and the aesthetic implications make surgical treatment the primary therapeutic option for keratoacanthoma.4 In our case, at the follow-up appointment 30 days after the initial presentation, when the surgical excision was scheduled, the lesion had almost disappeared and a small, fibrotic, pink-to-white area of scarring remained. The patient reported that the nodule had gradually become involuted since the biopsies were performed. Left to natural progression most cases of keratoacanthoma tend to resolve spontaneously.5 However, because of the difficulty to histologically differentiate keratoacanthomas from a low grade SCCs, surgical excision is mandatory.
Answer 8
Other therapeutic alternatives, such as intralesional injections of methotrexate, have shown effectiveness either alone or combined with surgery.6
Competing interest: None.
Provenance and peer review: Not commissioned, externally peer reviewed.