What is obstructive sleep apnoea?
Obstructive sleep apnoea (OSA) is a condition that causes repetitive episodes of upper airway obstruction during sleep, leading to hypoxia and/or sleep disturbance. The condition affects 1–5% of children,1 and peaks in the pre-school years. The features of the condition differ in a few key areas from those in adults (Table 1). Children with OSA often do not have complete apnoea, but more frequently experience episodic reduction in flow (hypopnoea) due to partial upper airway obstruction. About one-third of children snore; about 10% snore most nights2 and this is the cardinal symptom of OSA. In childhood, males and females are equally affected.
Table 1. Key differences between adults and children in the clinical features of OSA1,3
|
|
Adults
|
Children
|
|---|
|
Alternating snoring with obstructive apnoeas
|
Continuous snoring – episodic reduction in flow (hypopnoea) without obstructive apnoea is most common
|
|
Daytime sleepiness is a cardinal feature
|
Daytime sleepiness is uncommon
|
|
Obesity is common
|
Most children are normal weight, although failure to thrive may occur in severe cases and obesity increases the risk
|
|
Male predominance
|
No prevalence difference by gender
|
What causes obstructive sleep apnoea in children?
In adults, obesity is the primary association with OSA. Gender, airway anatomy and neuromuscular control also play a role.3 In paediatrics, the majority of children presenting with OSA are not obese and may even be underweight.4 The most common cause of OSA in childhood is enlargement of the tonsils and adenoids. Tonsils and adenoids grow most quickly in the pre-school years, and the adenoids are large in some children even in the latter part of the first year of life.5 Enlargement of lymphoid tissue in the confined space of the pharynx can result in airway obstruction during sleep, with relaxation of the dilating muscles of the pharynx contributing to the obstruction. In addition to OSA, enlarged upper airway lymphoid tissue may present with problematic nasal obstruction during wakefulness, or difficulty with swallowing larger items of food. Obesity, when present, does increase the risk of OSA, affecting up to 50% of overweight and obese children,6 compared with 1–5% of the general paediatric population. Allergic rhinitis or other causes of nasal obstruction may also cause OSA. Children with certain medical conditions are also more likely to have OSA, particularly those associated with muscle weakness, hypotonia, craniofacial abnormalities including retro/micrognathia, previous upper airway surgery including repair of cleft palate, and syndromes such as Down syndrome or achondroplasia.
The consequences of OSA in childhood
Studies on the impact of OSA in children have been focused on two main areas: cardiovascular and neurocognitive/behavioural. OSA is associated with increases in blood pressure during sleep7,8 and structural changes, such as increased left and right ventricular wall thickness, evident on echocardiography.9 Long-term consequences of these changes are not known, but long-term studies in other conditions suggest that increases in blood pressure in childhood increase the risk of lifelong hypertension.10 Evidence for the reversibility of these structural cardiac changes with resolution of OSA11,12 adds weight to the argument for early detection and treatment of this condition.
The relationship of OSA with impairments in memory, attention, learning and behaviour has been recognised for many years. Hypoxia or sleep disturbance in the absence of hypoxia have the potential to impair cognitive functioning and lead to daytime behavioural difficulties. One early study in the US demonstrated OSA in 18% of children performing in the lowest 10% of the first grade (aged approximately 6 years), with subsequent improvement in performance after adenotonsillectomy.13 Subsequent studies have found specific cognitive deficits in learning and mental processing in children with OSA.14 Somewhat surprisingly, children with mild OSA, or even those who snore but do not have OSA, have been shown in many studies to have measurable cognitive deficits as a group.15,16 The explanation for this remains unclear and is the subject of ongoing studies.
How is obstructive sleep apnoea detected?
Features on history raise the possibility of OSA. Snoring is very common in childhood1 but should not be regarded as normal, even where there is a strong family history. Children with persistent snoring or noisy breathing during sleep, present during three or more nights/week in the absence of an upper respiratory tract infection, are at risk of OSA. Although features of history, such as the loudness of snoring, have been shown to be poor predictors of the presence or severity of OSA, there are features that increase the likelihood of OSA in a snoring child (Table 2). Unrefreshing sleep despite a seemingly adequate quantity of sleep (wakes tired and grumpy), secondary enuresis (a relapse of bedwetting after being dry for more than six months)17 and poor weight gain4 are also possible consequences of OSA. Unlike OSA in adults, children are usually not obviously sleepy but are more likely to manifest difficulties with behaviour or concentration, which overlaps with the phenotype of attention deficit hyperactivity disorder.18 The original descriptions of the disease reported significant failure to thrive (poor weight gain) and clinical signs of right heart failure,4 but with earlier diagnosis, these features have been uncommon in recent years.
Table 2. Features on history that increase the likelihood of OSA in a snoring child33
|
|
Snoring or noisy breathing during sleep present ≥3 nights/week
|
|
Difficulty breathing while asleep (including increased effort of breathing, choking, gasping or snorting during sleep) observed by parents
|
|
Parents who reports being afraid for their child’s health because of the child’s breathing while asleep
|
|
Frequent daytime mouth breathing
|
|
Witnessed obstructive apnoea during sleep (parent describes cessation of airflow with clear ongoing breathing effort*)
|
|
*All children have central apnoea during sleep where there is cessation of airflow without respiratory effort, and care should be taken to distinguish this on history from obstructive apnoea if possible
|
Important features on clinical examination should include assessment of conditions predisposing to OSA and complications of severe OSA (Table 3). Measurements of growth (serial if possible) are important, as obesity predisposes a child to OSA and severe OSA can be associated with failure to thrive. Nasal examination for features of allergic rhinitis (eg mucosal inflammation, swollen turbinates) or reduced airflow suggestive of obstruction by enlarged adenoids, and assessment of the oropharynx, including tonsillar size, should be performed.
The diagnosis of OSA can be confirmed with a sleep study (polysomnography), which involves non-invasively monitoring multiple physiological channels to record sleep and respiratory parameters overnight including oxygen and carbon dioxide levels (Figure 1). This can be arranged through referral to a sleep centre or specialist sleep paediatrician. The sleep centre chosen must have experience in sleep studies in children, as performing sleep studies can be challenging in young children. There are also different rules for defining the condition and different thresholds for treatment, compared with sleep studies in adults.19 Alternatives to polysomnography for children are being actively investigated owing to the very restricted availability of this test in Australia. Simplified studies with limited channels performed at home show promise, especially if abnormal, and specific abnormalities on overnight oximetry have a strong predictive value for OSA.20
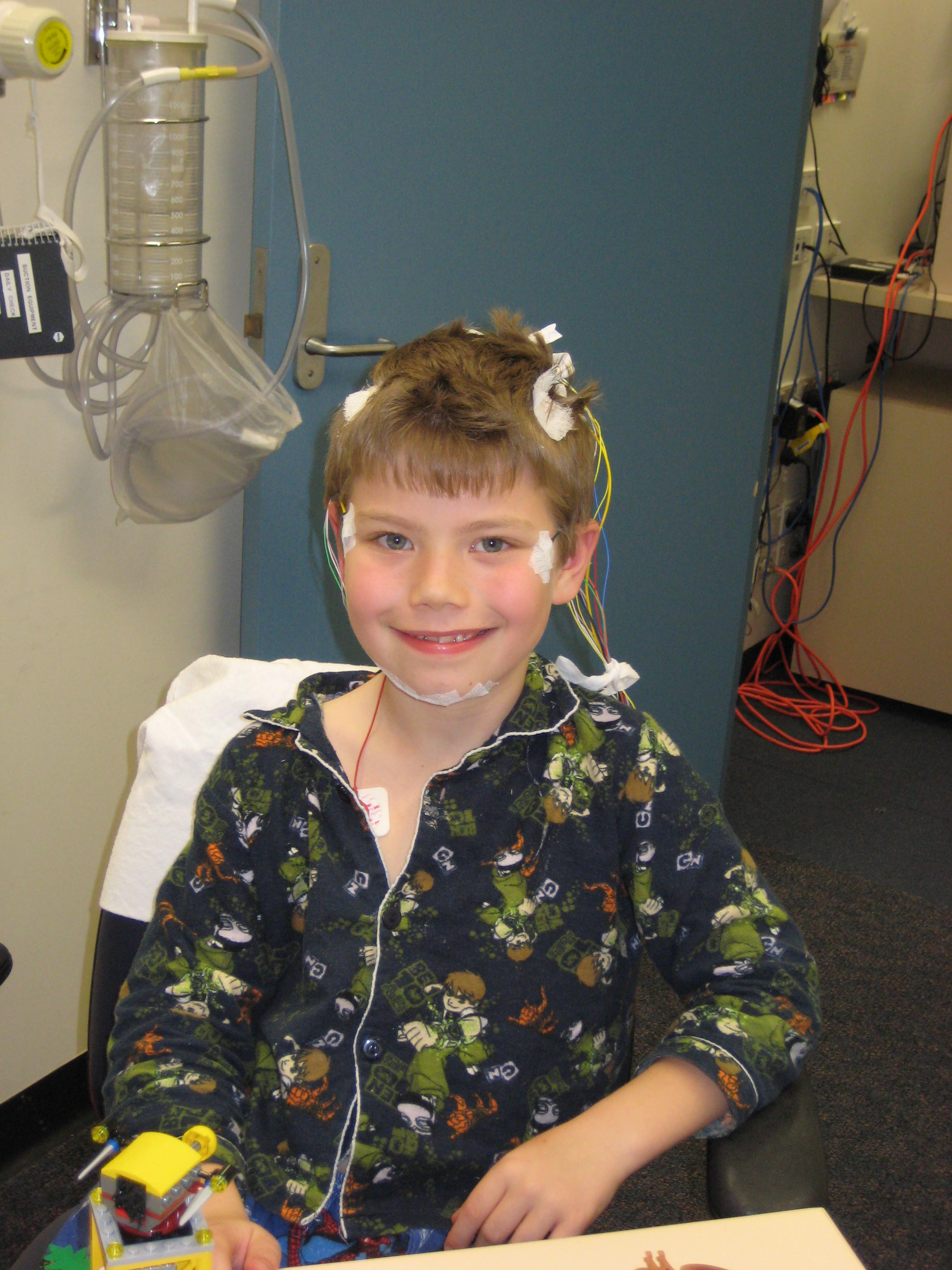 |
| Figure 1. A child undergoing polysomnography for assessment of possible obstructive sleep apnoea. |
Table 3. Important aspects of clinical examination in a child with suspected OSA
|
|
Growth: either slowing of weight gain or obesity
|
|
Craniofacial structure (retro/micrognathia, midface hypoplasia, adenoidal facies)
|
|
Nasal airflow, septum, turbinates
|
|
Tongue, pharynx, palate, uvula, tonsils
|
|
Pectus excavatum or Harrison’s sulci
|
|
Right ventricular hypertrophy, pulmonary hypertension, systemic hypertension
|
What treatment should be offered and when?
A range of treatment options is available, including surgical removal of the tonsils and/or adenoids, anti-inflammatory treatments, dental therapies and continuous positive airway pressure (CPAP).
In cases where the history is very strong and the child has enlarged tonsils (occupying more than 50% of the pharyngeal diameter21) and no other medical problems, direct referral to an ear, nose and throat surgeon for consideration of adenotonsillectomy may be appropriate, given the scarcity of paediatric sleep medicine services. Examples of features of a strong history are given in Table 2. Assessment of adenoidal size can be achieved using a lateral X-ray of the head or by an experienced operator using nasendoscopy.
Children under the age of 3 years, those with significant comorbidity and those with severe OSA are at increased risk of post-operative respiratory complications after adenotonsillectomy (approximately 25% for those with severe OSA), such as the need for supplemental oxygen or even re-intubation.22–24 Therefore, the choice of a location for the surgery should reflect that possibility.
The evidence for benefit of adenotonsillectomy has been accumulating over many years and a recent randomised controlled trial25 has shown improvements in sleep-related symptoms and quality of life. Surgery may not reverse all of the associated decrements in neurocognitive functioning, especially for children with more mild OSA.25 The reason for this remains unclear. Adenotonsillectomy often improves OSA in children who are obese25,26 or who have an underlying medical condition, such as Down syndrome, but in these groups in particular, the increased possibility of persisting disease after treatment should be borne in mind. A small number of children in whom surgical treatment is not appropriate or effective will require CPAP treatment.
Several studies have supported the use of anti inflammatory agents in children with mild OSA as defined by polysomnography.27,28 This includes topical intranasal steroid sprays29 and leukotriene receptor antagonists.28 Nasal steroids take at least 2 weeks to improve symptoms, and a 6-week trial is recommended before determining the extent of benefit. These agents are helpful in controlling allergic rhinitis as a contributing factor to nasal obstruction, but may also reduce the size of the adenoids sufficiently to improve nasal obstruction in unselected patients with mild OSA.29 Where snoring is not associated with the features outlined in Table 2, and where there is clinical nasal obstruction with small tonsils, or allergic rhinitis in an older child, consideration of a trial of nasal steroids is warranted before referral for surgery. Commonly used agents in paediatrics include mometasone furoate 50 µg in each nostril once daily and fluticasone furoate 27.5 µg in each nostril once daily. There is evidence for the benefit of leukotriene receptor antagonists for mild OSA in children, but these agents are not approved by the Therapeutic Goods Administration (TGA) for this indication.
Dental therapies, such as rapid maxillary expansion may alter airway size, and be beneficial in children with OSA,30 but the literature supporting this is in its early stages.31
A suggested approach to case finding and treatment in primary care
An awareness of the potential significance of poor quality sleep in children is paramount. Parents may not appreciate the significance of snoring, nor mention sleep problems when consulting a general practitioner about their child’s health.16,32 A simple screening question regarding noisy breathing during sleep may uncover symptoms that had not alerted parents to a significant problem. This question is especially important in the context of problematic night wakings, nasal obstruction and tonsillar hypertrophy. Where there is a strong clinical suspicion of OSA, referral for assessment by an ear, nose and throat surgeon is reasonable, especially in a child older than 2 years with tonsillar hypertrophy, recurrent tonsillitis or daytime symptoms of pharyngeal obstruction such as frequent choking on foods. In children aged less than 2 years, or less clear clinical scenarios or where there is a possibility of comorbid sleep problems contributing to the symptom complex, such as inadequate sleep due to difficulty settling at the start of the night or marked daytime sleepiness in an older child, referral to a specialist paediatric sleep physician is warranted.
Key points
- Snoring is common in childhood but should not be regarded as normal as it is the key symptom of OSA.
- OSA has an adverse impact on sleep quality, daytime functioning and cardiovascular health, even in very young children.
- Symptoms of OSA should be sought in any child with enlarged tonsils and/or disturbed or unrefreshing sleep.
- Treatment with adenotonsillectomy is usually effective in uncomplicated cases and should be considered in children with a strong history and enlarged tonsils.
- Anti-inflammatory agents should be considered for children with nasal obstruction and mild symptomatology.
Competing interests: None.
Provenance and peer review: Commissioned, externally peer reviewed.