Asthma is among the most commonly managed chronic problems in general practice.1 About 2.3 million Australians (10.2%) had asthma in 2011–12, a significantly lower prevalence than in 2001 (11.6%).2 About 11% of boys and 6% of girls aged 0–14 years have diagnosed asthma.3 Inhaled corticosteroids (ICS) are increasingly used for first-line management in adults and children and the 2014 Australian Asthma Handbook advocates commencement at a lower threshold of symptoms than was recommended previously for adults and adolescents.4 For children, ICS are recommended as a first choice prevention treatment only in those with moderate or severe persistent asthma, and at the lowest effective dose.4 Regular review is advised in children to avoid the risk of adverse effects associated with long-term ICS use.5
Our aims were to determine changes in general practice asthma management rates among children over the past decade, and to examine current medication use and level of control in a recent sample of children with asthma aged <15 years.
Method
This is a secondary analysis of data from the Bettering the Evaluation And Care of Health (BEACH) program and of a program sub-study. BEACH methods are described in detail elsewhere.6
Changes in rates of asthma management in children from 2005–2014 are reported. We calculated proportions and robust 95% confidence intervals using survey procedures in SAS (version 9.3)7 that adjust for the study’s cluster design.
Prevalence data were gathered during July and August 2014 from a Supplementary Analysis of Nominated Data (SAND) sub-study.8 GPs recorded whether the patient had diagnosed asthma;8 current asthma medications; and asthma control assessed using the Global Initiative for Asthma (GINA) tool.9 Specifically, patients were asked if they had experienced asthma symptoms in the last 4 weeks, used reliever medication more than twice a week, or had been limited in their activity9 (carers were asked to respond on behalf of children). Although patients of all ages were sampled, these results are limited to children aged <15 years.
Results
Management of asthma: From 2005–2014, there were 61,229 BEACH encounters with children aged <5 years and 50,968 encounters with children aged 5–14 years. The rate of asthma management did not change significantly over the time period for patients in either age group, averaging 3.4 per 100 encounters for those <5 years and 6.3 per 100 encounters for those aged 5–14 years across the decade (Figure 1).
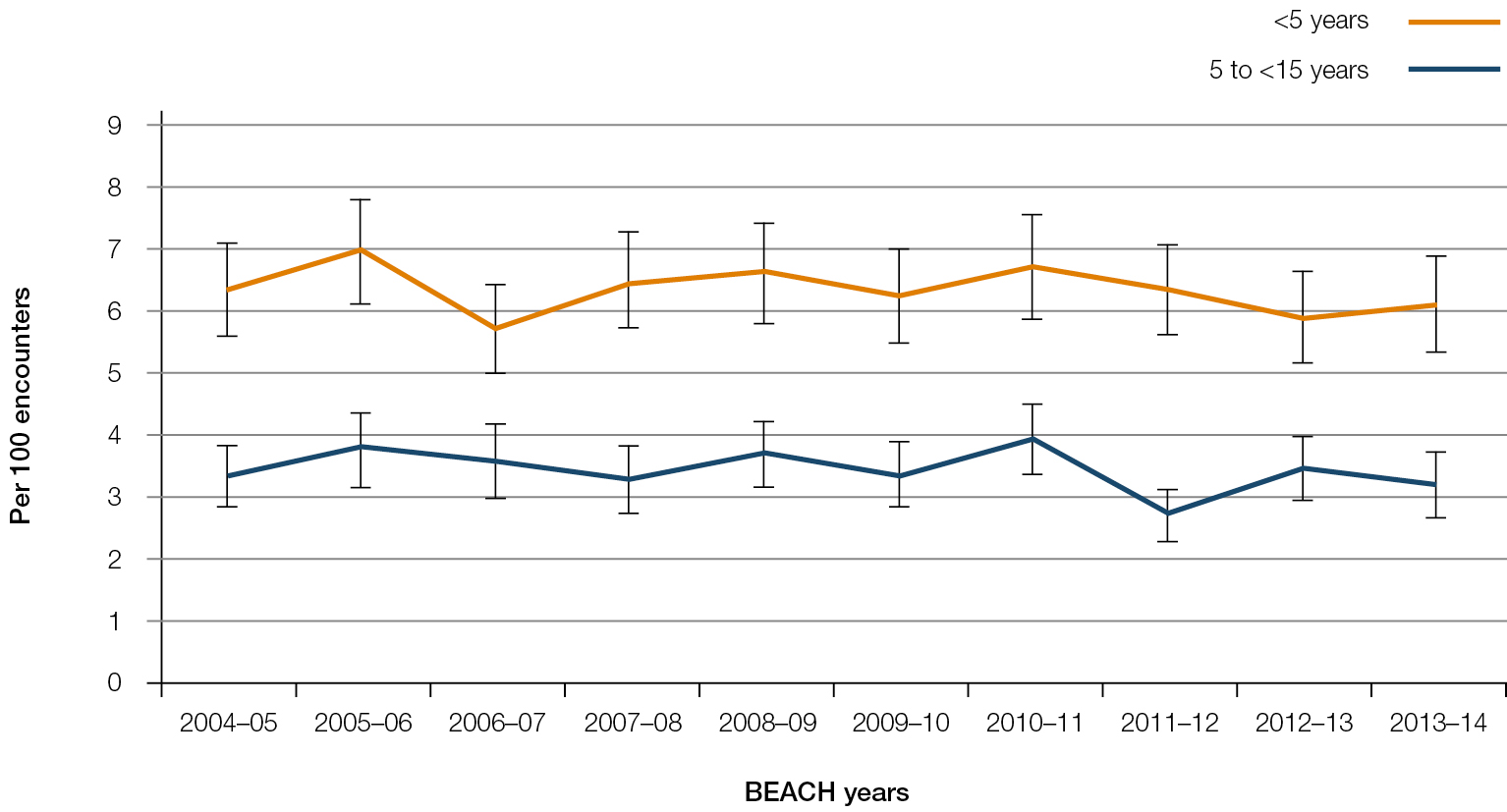 |
| Figure 1. Asthma management rate at GP–patient encounters |
Prevalence: Completed asthma SAND questions were returned by 97 of 125 general practitioners (GPs; 77.6%). Among these, GPs surveyed 409 patients aged <15 years, the age and sex distribution of whom did not differ significantly from those of children who attended all BEACH encounters in 2013–14. Of the 409 patients, 56 (13.7%, 95% CI: 10.3–17.1) had diagnosed asthma (17.5% of boys and 9.2% of girls). Two-thirds of the patients with asthma were boys. Of children aged 1–4 years, 8.7% had diagnosed asthma as did 23.8% of those aged 5–14 years. Because the sample size was small (n = 56), the group was analysed as a whole rather than separating them into <5 years and 5 to <15 years age groups.
Medication use and asthma control: Forty-five patients were taking medication for their asthma; salbutamol accounted for almost two-thirds (63.8%) of the 69 medications recorded. Twenty-four children taking medication for asthma (42.8%) were taking an ICS or ICS/LABA (combination).
Using the GINA guidelines, half of the patients with asthma had experienced symptoms in the previous 4 weeks. Twenty-nine patients (51.8%, 95% CI:37.9–65.7) had no symptoms (considered well controlled), 17 patients (30.4%; 95% CI: 18.9–41.8) had one or two (partly controlled) and 10 patients (17.9%, 95% CI: 6.4–29.3) had experienced three or four symptoms (uncontrolled). Among patients on ICS or ICS/long-acting â-agonists (LABA), eight were well controlled, nine were partly controlled and seven were uncontrolled.
All nine patients taking no medication were categorised as having well-controlled asthma. There were 21 patients for whom ashort-acting â-agonist (SABA) was reported to be the only asthma medication taken. Of these, 12 were well controlled, seven were partly controlled and two were uncontrolled. ICS/LABA were taken in combination by 10 patients: three well controlled, four partly controlled and three uncontrolled.
The reported daily doses of ICS aligned with those recommended,10 with the exception of six patients on twice the recommended dose. Two of these had uncontrolled asthma and the other four were well controlled.
Discussion
These results show that the rate of asthma management in general practice for children <15 years did not change significantly over the decade. The prevalence was higher in boys than girls, supporting results of the Australian Health Survey 2011–12, which also found asthma in children <15 years to be significantly more common in males than females.2
A previous study found good correlation between GPs’ perception of asthma control and patients’ individual ACQ5 scores.11 There seems a similar fit for the patients in this study, given they were, overall, appropriately medicated for their asthma control level as determined by the GINA guidelines.9 The patients who took no medication were all in the well-controlled group, and no partly controlled or uncontrolled patients were unmedicated.
Patients taking only SABAs were mostly well controlled, although there may be patients who could benefit from ICS, given there were nine patients ranked as partly controlled or uncontrolled who were taking only SABAs. There are differences in GP attitudes and familiarity with current guidelines relating to childhood asthma management;12 however, while some GPs may be less familiar with guidelines, others may be cautiously concerned about adverse events associated with ICS use, particularly in the long term.5 Others may consider the benefits of ICS use outweigh the risk for individual patients with uncontrolled asthma, but risk can be minimised by using the lowest effective dose.5 Constant review is important for all users, particularly in the case of patients on higher than recommended doses, such as the six patients reported in this study. Also of concern are the 10 patients on ICS/LABA combination therapy, given the potential for adverse effects associated with LABAs in children.13 These children may have been prescribed according to asthma control level initially, but only three of these patients were ranked as uncontrolled currently.
Some limitations should be considered. The GINA guidelines are designed for patients >5 years of age and were used for this sub-study because, although only children’s results are reported here, it surveyed all patients. However, we believe they are a reasonable guide for asthma control in these young patients. Encounters with children contribute a small proportion of total GP workload (about one in eight),1 reflected in the small sample size. Some results (denominator n < 30) should be interpreted with caution. Further research involving larger patient numbers would provide more reliability.
One in eight children aged <15 years have diagnosed asthma and the GP management rate has not changed over the past decade. While most children are appropriately medicated according to their asthma status, this study highlights the need to continually monitor appropriate medication use for asthma control in children so that ICS, and particularly LABA, are not prescribed for children who do not need them.
Competing interests: None.
Provenance and peer review: Commissioned, peer reviewed.
Acknowledgements
The authors thank the GP participants in the BEACH program and all members of the BEACH team. Financial contributors to BEACH between 2000 and 2014: Abbott Australasia Pty Ltd; AstraZeneca Pty Ltd (Australia); Australian Government Department of Health and Ageing; Australian Government Department of Veterans’ Affairs; Bayer Australia Ltd; bioCSL (Australia) Pty Ltd; GlaxoSmithKline Australia Pty Ltd; Janssen-Cilag Pty Ltd; Merck, Sharpe and Dohme (Australia) Pty Ltd; National Occupational Health and Safety Commission; National Prescribing Service; Novartis Pharmaceuticals Australia Pty Ltd; Pfizer Australia Pty Ltd; Roche Products Pty Ltd; Sanofi-Aventis Australia Pty Ltd; Wyeth Australia Pty Ltd. BEACH is approved by the Human Research Ethics Committee of the University of Sydney.