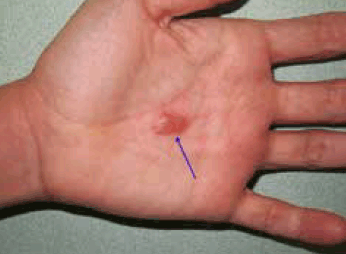
Figure 1. Palmar blister
Question 1
What is the most likely diagnosis?
Question 2
What is the differential diagnosis?
Question 3
What is the most appropriate management?
Question 4
What is the most useful investigation to confirm the diagnosis?
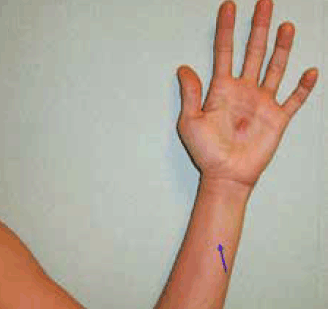
Figure 2. Lymphangitis
Answer 1
Given the absence of medication use, the most likely diagnosis is recurrent Herpes simplex infection. H. simplex viruses produce primary and recurrent vesicular eruptions that favour the orolabial and genital regions. The palmar area is involved less commonly.1 Viral lymphangitis can complicate the clinical presentation, as in this case, and it is frequently misdiagnosed as a bacterial infection.1,2 It should be noted that the infection can be transmitted to others through contact with skin vesicles.
Answer 2
Recurrent H. simplex needs to be differentiated from other causes of bullous skin lesions:
In a fixed drug eruption, one round sharply demarcated erythematous plaque is seen, sometimes with a central blister and violaceous hue. However, previous pharmacological exposure is a prerequisite and was not a feature of this case.
Culicosis bullosa are bullous reactions to insect bites. These reactions are common and are often multiple, pruritic, excoriated papules or plaques. Although the cutaneous manifestations could be compatible with an insect bite, the recurrent course and painful lesions make this option improbable.
Localised bullous pemphigoid could present as a tense blister with perilesional erythema, but lymphangitis is not a frequent finding.3 Local factors, such as trauma, ultraviolet light or topical therapy, seem to be frequently implicated in the production of lesions.4 The disease has a benign course and generally responds to cessation of exacerbating factors and initiation of topical steroids.
H. zoster infection begins with a prodrome of pruritus, hyperaesthesia and intense pain. Most patients develop a painful eruption of grouped vesicles on an erythematous base in a dermatomal distribution. It is rarely recurrent.
Bullous erysipelas represents a superficial cellulitis with lymphatic involvement. It is usually caused by group A-haemolytic streptococci. It typically appears as a sharply demarcated, tender area of erythema and oedema with an indurated border. The clinical course may be complicated by the local appearance of bullae. Accompanying signs and symptoms include lymphangitis and chills. The absence of fever and resolution with symptomatic treatment make this an unlikely diagnosis here.
Answer 3
Although valaciclovir, famciclovir and aciclovir have all shown high clinical efficacy in the treatment of herpesvirus infections, valaciclovir is preferable for long-term suppressive therapy from a treatment adherence perspective as it requires only daily dosing (500 mg/day).5 Valaciclovir is an oral pro-drug that is converted to aciclovir after administration.1 Chronic suppressive therapy is usually reserved for patients with six or more recurrences per year. In our patient, the acute episode was not treated with valacyclovir but prophylactic treatment was instituted and no recurrence was reported at 1-year follow-up. It was not neccesary to add oral broad spectrum antibiotics in this case because the lymphangitis was due to viral infection.1,2
Answer 4
Detection of herpesvirus DNA using real-time polymerase chain reaction (RT-PCR) is the best test to confirm the diagnosis. In addition to being the preferred method for identifying H. simplex in cerebrospinal fluid, RT-PCR is increasingly being used as a rapid, sensitive and specific method to detect H. simplex DNA in specimens from the skin and other organs.6 To ensure an adequate sample and avoid false-negative results, the vesicle should be unroofed and the base of the ulcer scraped with a flocked swab in universal transport media (Copan, Italy).7 H. simplex type 1 was isolated from this patient.
Tzanck smear test is rarely used now for diagnosis. It reveals multinucleated epithelial giant cells but it does not distinguish between H. simplex and Varicella zoster virus.6 Viral culture was the traditional gold standard for detection of H. simplex and was the reference method against which all other tests were measured. A cell culture positive for H. simplex suggests probable active infection. However, a negative cell culture result does not rule out H. simplex infection, particularly if the specimen is from cerebrospinal fluid or nonvesicular lesions.8 Stained biopsy specimens show intra-epidermal vesiculation associated with balloning degeneration of keratinocytes, which often fuse to form multinucleated giant cells.9
Competing interests: None.
Provenance and peer review: Not commissioned; externally peer reviewed.