Definitions
An aneurysm is defined as an enlargement of an artery resulting in a diameter of more than 1.5 times the normal size (Figure 1). True aneurysms have an intact arterial wall consisting of all three layers (intima, media and adventitia), while false or pseudoaneurysms lack a complete arterial wall. True aneurysms are mostly of atherosclerotic or degenerative origin. In contrast, false aneurysms are usually related to trauma, iatrogenic injury or infection.
Splanchnic artery aneurysms
Splanchnic artery aneurysms (SpAAs) encompass intra-abdominal aneurysms that are not part of the aorto-renal-iliac system, and include aneurysms of the coeliac artery, the superior and inferior mesenteric arteries, and their branches. Of all intra-abdominal aneurysms, only around 5% involve the splanchnic arteries.1 The prevalence of SpAAs has been estimated at 0.1–2.0%;2 their anatomic distribution is illustrated in Figure 2.3 The majority of SpAAs are found in splenic and hepatic arteries.
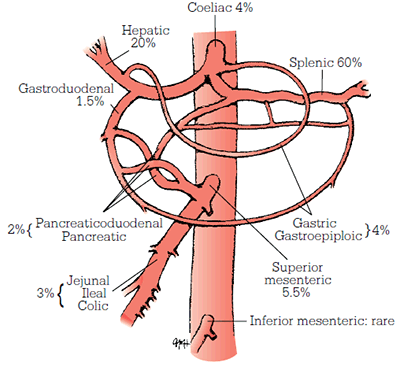
Figure 2. Anatomical distribution of SpAAs
Reproduced with permission from Cronenwett JL, Johnston KW. Rutherford’s Vascular Surgery. Philadelphia: Elsevier, 2010
Approximately one-third of SpAAs are associated with other aneurysmal disease. True SpAAs are mostly degenerative or atherosclerotic, but may also be associated with disorders such as Ehlers-Danlos syndrome, fibromuscular dysplasia and other collagen vascular diseases.1 Risk factors for splenic artery aneurysms are female gender, a history of multiple pregnancies and portal hypertension. Splanchnic pseudoaneurysms are commonly caused by (iatrogenic) trauma or injury, local inflammation or infection.
Splanchnic artery aneurysms are generally asymptomatic and discovered incidentally. They have been described to rupture and provide a major clinical challenge due to difficulty in reaching a diagnosis, technical issues in terms of locating and controlling ruptured aneurysms, and the rapid onset of haemodynamic shock if there is uncontrolled intraperitoneal bleeding. Rupture is often life-threatening and has a reported mortality of 25–100%, depending on anatomical location. In pregnant women, rupture of a splenic artery aneurysm, usually in the third trimester, has a devastating mortality of 80–90% for both mother and child. Although precise rates of rupture are lacking, the lifetime risk for rupture of SpAAs is high and reported between 13% (for coeliac artery aneurysms) to 90% (for gastric artery aneurysms). Furthermore, splanchnic artery pseudoaneurysms have a higher rupture potential than true aneurysms.
Early elective aggressive repair is recommended for SpAAs in pregnant women or young patients, for rapidly enlarging aneurysms or pseudoaneurysms, aneurysms in patients with a known connective tissue disorder, and for aneurysms greater than 2 cm. For smaller aneurysms, or in patients with high comorbidity, a conservative approach can be justified. Current treatment options include open surgical repair (ligation, excision, interposition) and endovascular treatment (embolisation, covered stent graft), both of which show favourable long term results (Figures 3a to 3d).4
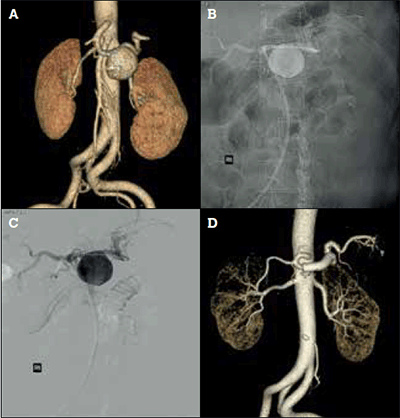
Figure 3. Endovascular treatment of a splenic artery aneurysm with a covered stent. A) Preoperative CT angiogram reconstruction of 5 cm splenic artery aneurysm; B) Conventional angiography with covered stent positioned across the splenic artery aneurysm before deployment; C) Digital subtraction angiogram image of 3B; D) Postoperative CT angiogram reconstruction with a covered stent (Gore Viabahn®, Flagstaff Ohio, USA) completely excluding splenic artery aneurysm
Renal artery aneurysms
Renal artery aneurysms (RAAs) are usually separately described from other intra-abdominal aneurysms, as they show a different natural history and are often associated with hypertension. Renal artery aneurysms have an incidence of 0.7–1.0% and are bilateral in about 10% of cases.5 True aneurysms are mostly due to atherosclerosis, fibromuscular dysplasia or a congenital wall defect, and lesions commonly occur at the main renal artery bifurcation, which makes repair challenging. False aneurysms generally arise from (iatrogenic) trauma. Hypertension is seen in 80% of patients with RAAs.6
The lifetime risk of rupture of RAAs is estimated at 3%, but rupture has a mortality of 10% in men and non-pregnant women. In pregnant women, rupture of a renal artery is associated with a 55% maternal and 85% fetal mortality. Renal artery aneurysms may also cause renovascular hypertension via distal embolisation. Indications for RAA repair are rupture, acute dissection, pseudoaneurysm, women of childbearing age and size greater than 2 cm. Repair can be performed with open surgery or endovascular surgery (coiling, covered stent) with excellent results (Figures 4a to 4d).
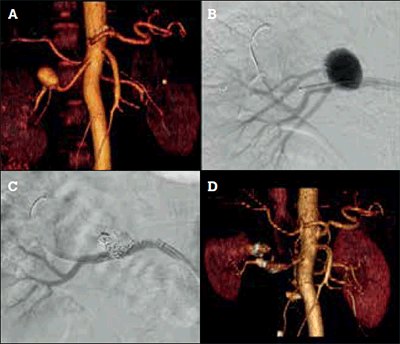
Figure 4. Endovascular treatment of a 2.5 cm right renal artery aneurysm.
A) Preoperative CT angiogram reconstruction demonstrating a right renal artery aneurysm; B) Intraoperative angiogram demonstrating the aneurysm. A right upper pole artery arises from the aneurysm and must be sacrificed; C) Intraoperative angiogram after deployment of embolisation coils (Cook Nester® 0.018”, Bloomington IN, USA) to fill the aneurysm sac and prevent retrograde filling from the upper pole artery with a covered stent (Atrium Advanta V12 RX® 0.014” stent, Hudson NH, USA) placed to contain the coils and seal the aneurysm; D) Postoperative CT angiogram reconstruction demonstrating exclusion of the aneurysm and reduced right upper pole renal parenchymal perfusion
Supra-aortic trunk and upper extremity arterial aneurysms
Upper extremity and supra-aortic trunk aneurysms are less common than other peripheral arterial aneurysms. Arch vessel aneurysms usually arise from congenital degeneration associated with connective tissue disorders or, rarely, congenital causes such as a Kommerell diverticulum with an aberrant right subclavian artery, and in 30–50% of patients are associated with aortoiliac or other peripheral aneurysms. Distal subclavian artery aneurysms can occur as a result of chronic trauma, as seen with thoracic outlet syndrome. More distal aneurysms, such as carotid, axillary, brachial, ulnar and radial artery aneurysms, may be caused by (iatrogenic) trauma. The incidence of brachial artery pseudoaneurysm after cannulation for diagnostic or therapeutic purposes is 0.3%.8 Hypothenar hammer syndrome can also lead to unusual aneurysms of the distal ulnar artery due to repetitive trauma to the palm or wrist.
The presentation of upper extremity aneurysms differs because, unlike aortoiliac and most splanchnic aneurysms, rupture is rare. Symptoms usually consist of limb or digital ischaemia from thrombosis or embolisation, resulting in ulceration, necrosis or gangrene, and potentially leading to amputation.7 Moreover, arch vessel aneurysms may present with symptoms from expansion and compression of the aneurysm against surrounding structures, such as pain, neurologic dysfunction, hoarseness, respiratory insufficiency and dysphagia.
Fit patients with symptomatic and large arch vessel aneurysms should be offered elective repair with open surgery. This treatment has substantial mortality and morbidity, but also long term durable results. In patients with high comorbidity, an endovascular approach should be considered.9 While branched stent grafts for endovascular aortic arch repair are in development, they are yet to be proven as a feasible and durable treatment strategy.
In comparison, open surgical repair of more distal upper extremity aneurysms has a significantly lower morbidity and should therefore be offered to all patients with symptomatic distal aneurysms (Figure 5). Endovascular treatment is currently rarely performed because of anatomical difficulties in the upper extremity. Brachial artery pseudoaneurysms can be repaired under local anaesthesia, or thrombosed with thrombin injections when they are small and the patient has an adequately long neck.
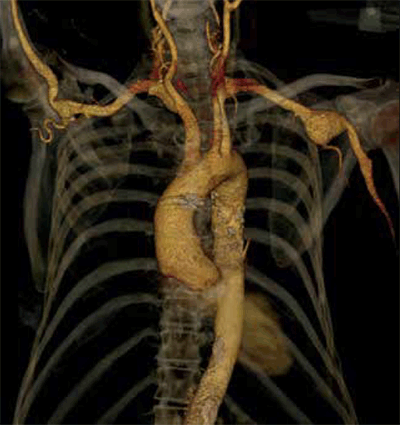
Figure 5. CT angiogram reconstruction of an axillary artery aneurysm in a patient with Marfan syndrome. This was subsequently treated by open resection and bypass
Lower extremity arterial aneurysms
Aneurysms occurring in the arteries of the lower limb are the most common peripheral aneurysms. In clinical practice, the majority of femoral or tibial artery aneurysms are (iatrogenic) false aneurysms, whereas those of the popliteal artery are true degenerative (atherosclerotic) aneurysms.
Iatrogenic femoral pseudoaneurysms can occur at the anastomotic site of previous surgery (endarterectomy or bypass) or as a complication of common femoral artery punctures for catheterisation procedures.10 More rarely, pseudoaneurysms of distal arteries, such as profunda femoris, popliteal or tibial arteries, arise from a traumatic (often penetrating) surgical or orthopaedic injury.
Pseudoaneurysms usually present as a painful pulsating mass with associated hematoma. Rupture or distal ischaemia are rare, but progressive enlargement can cause overlying skin ischaemia and necrosis. Femoral puncture pseuodaneurysms can be treated conservatively when less than 2 cm and in the absence of therapeutic anticoagulation. Larger pseudoaneurysms from a femoral puncture and anastomotic or traumatic pseudoaneurysms should be repaired. Treatment options consist of ultrasound guided compression or thrombin injection for smaller aneurysms, or open surgical repair with debridement and local repair or a bypass.11 In some patients, covered stenting from the contralateral limb has been used successfully.
The majority (approximately 70%) of true lower extremity aneurysms are popliteal artery aneurysms, with an estimated incidence of 0.4% in hospitalised patients.12 Femoral artery aneurysms are strongly associated with popliteal aneurysms and are the second most common peripheral aneurysms. Moreover, a strong association exists between the presence of true aneurysms of the femoropopliteal arteries and those of the contralateral extremity, as well as the aortoiliac segment: in patients with a femoropopliteal aneurysm, the prevalence of an aortic aneurysm is approximately 50%.7,12–14 Furthermore, both femoral and popliteal artery aneurysms are found bilaterally in 50% of patients.7 In approximately 10% of patients with an aortic aneurysm, a femoropopliteal aneurysm is found.
Risk factors for developing a true femoropopliteal aneurysm are the same as those for developing an aortic aneurysm (ie. smoking, male gender, hypertension, family history). Consequently, discovery of a femoropopliteal aneurysm mandates careful observation and evaluation for associated aneurysms in the opposite limb and abdominal aorta.
One-third of true common femoral artery aneurysms present as a painless, pulsating mass in the groin. Another 30% have local pain or tenderness, femoral nerve neuralgia or limb oedema. In reported series, 10–65% have complications of ischaemia, from claudication to critical limb ischaemia due to embolisation.12 Acute thrombosis occurs in around 15% of cases, while rupture is exceedingly uncommon. All symptomatic aneurysms and those greater than 2.5 cm should be surgically repaired by exclusion of the aneurysm with an interposition graft.13
Popliteal aneurysms may manifest as an asymptomatic, pulsating mass behind the knee or cause symptoms of either chronic or acute ischaemia. Rupture is very rare. Symptoms of ischaemia are due to thrombosis of the aneurysm itself or emboli to the distal circulation. The likelihood of symptoms developing in untreated patients is 34% at 3 years in asymptomatic patients with normal pedal pulses, but rises to 86% in asymptomatic patients with absent pulses.14 Acute limb ischaemia is the most feared complication of popliteal aneurysms, which complicates treatment and is associated with a significant risk of limb loss.
In case of acute limb ischaemia, clinical judgement should determine the type of treatment, which ranges from intravenous heparin, catheter directed thrombolysis, emergency surgical bypass or primary amputation.15 Because the results of surgical treatment are best in asymptomatic patients, and progressively worse in patients with chronic or acute limb ischaemia, detecting popliteal aneurysms before symptoms or complications occur is critical. The indications for elective treatment are diameter greater than 2.5 cm unless there is concern about mural thrombus, embolisation, or local mass effect.
The primary objective of treatment is to exclude the aneurysm from the circulation, and an open approach with surgical bypass plus ligation of the aneurysm, or with an interposition graft, is currently still the treatment of choice. With the rise in endovascular techniques, endovascular popliteal artery repair (EPAR) has become a promising and widespread treatment option.16 As with endovascular aortic aneurysm repair, EPAR is a technique that uses a covered stent to exclude the aneurysm and therefore is equally constrained, primarily by anatomy. Examples of each are illustrated in Figures 6a to 6c.
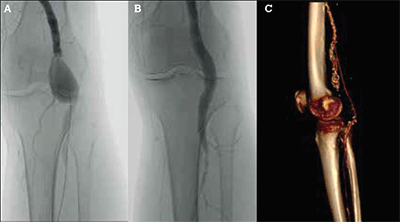
Figure 6. Open and endovascular treatment of popliteal aneurysms.
A) Unsubtracted angiogram of a 3 cm left popliteal aneurysm; B) Unsubtracted angiogram after deployment of a covered stent (Gore Viabahn®, Flagstaff Ohio, USA); C) CT angiogram reconstruction of a right popliteal bypass for aneurysm exclusion. The native popliteal aneurysm is calcified and has been ligated at surgery. The vein bypass extends from the distal superficial femoral artery to the below knee popliteal artery
In Australia’s largest reported case series of EPAR and in the only small randomised trial between open repair and EPAR, endovascular treatment resulted in decreased length of stay and recovery when compared to open surgery, with comparable patency rates but a higher number of re-interventions.17,18 Larger trials and longer follow up are needed to determine the definitive role of EPAR in the treatment of popliteal aneurysms.
Practice points
- Non-aortic aneurysms are uncommon and usually asymptomatic, but have significant morbidity and mortality when untreated.
- All patients with an arterial aneurysm should be screened for the presence of other arterial aneurysms.
- All patients with a non-aortic aneurysm should be referred to a vascular surgeon for elective assessment, counselling and potential treatment.
- Large size, pain, evidence of embolisation or (in splanchnic or renal aneurysms) young patients or women of fertile age should be referred for more urgent assessment.
- Endovascular repair has proven to be a promising minimally invasive approach for a range of aneurysms, however, open surgical repair continues to play a major role in the treatment of non-aortic aneurysms.
Competing interests: Jason Chuen has received payment for lectures from Medtronic and Cook Medical, and panel membership of Medtronic.
Provenance and peer review: Commissioned; externally peer reviewed.