The impact of nocturia on quality of life
- Nocturia has a profound impact on quality of life, being comparable to gout, hypertension, diabetes and angina in terms of disease burden2
- Nocturia typically becomes bothersome to patients when it occurs more than two times per night3
- The severity of sleep impairment increases with the number of nocturnal voids3
- Decreased sleep can cause daytime fatigue, poor concentration, memory impairment, mood alterations and affects work performance4
- Fall and hip fracture risk is increased in elderly patients with nocturia5
- Nocturia is a predictor of mortality, with higher mortality risk with increased number of nightly voids6
- Nocturia affects bed partners and carers and can cause relationship disturbances.7
Epidemiology
The prevalence of nocturia increases with age. More than 70% of people aged 70 years and over are affected.8 A substantial proportion of younger adults are affected with up to 30% of those aged 20–40 years voiding at least once per night.6 Younger sufferers are more likely to be female, however, prevalence is similar in both genders in older patients.8,9
Circadian control of urine production
Antidiuretic hormone (ADH) is released by the posterior pituitary and increases water reabsorption at the renal collecting tubule. Normally, ADH increases during sleep resulting in smaller volumes of concentrated urine.10 In elderly patients with nocturia this rhythm is often blunted with reduced nocturnal levels of ADH. This may be partly responsible for the increased prevalence that occurs with advancing age.11
Classification of nocturia
Nocturia can be categorised into three clinical entities based on the pattern of 24 hour urine (Figure 1):
- nocturnal polyuria
- global polyuria
- bladder storage disorders.
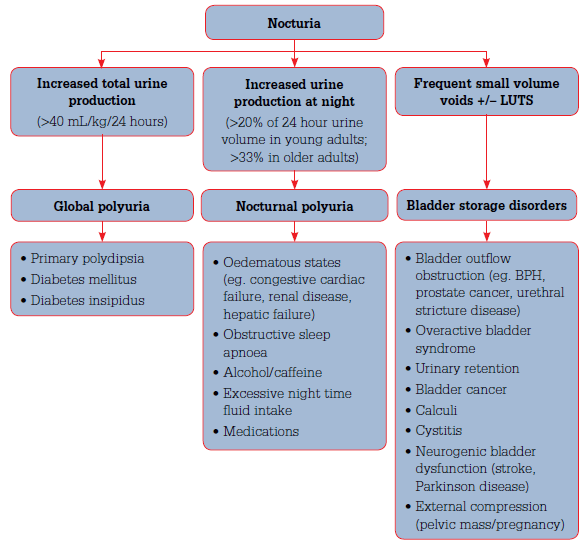
Figure 1. Classification of nocturia
The frequency-volume chart
A frequency-volume chart (FVC) – or 'voiding diary' – distinguishes between the three categories of nocturia.
Charts should include:
- volumes and times of urination for a 24–72 hour period1
- beginning and end of sleep periods
- time, type and volume of fluids ingested.12
Giving clear instructions before patients complete the chart improves compliance and eliminates the need for multiple diaries. It is usually more convenient for patients to choose a time when they are mostly at home to complete their voiding diary. The period recorded needs to be representative of the patient's typical symptoms. Once the diary has been completed by the patient, important values can be calculated (Table 1).
Table 1. Definitions of terms derived from the frequency-volume chart
| Term |
Definition |
| Nocturia |
Number of voids recorded during a night's sleep, each void is preceded and followed by sleep |
| Night time |
Time spent asleep |
| 24 hour voided volume |
Total volume of urine voided in a 24 hour period |
| First morning void |
The first void after waking with the intention of rising |
| Nocturnal urine volume |
Total volume urine passed during the night:
- includes the first morning void
- excludes the void before going to bed
|
| Maximum voided volume |
Largest single volume voided (usually between 300–600 mL) |
Pathophysiology
Global polyuria
Global polyuria is continuously raised urine output defined as >40 mL/kg/24 hours.1 The most common cause is primary polydipsia. Polydipsia can also be a compensatory mechanism for fluid loss, such as the osmotic diuresis of uncontrolled diabetes mellitus. Global polyuria can also result from diabetes insipidus.13
Nocturnal polyuria
Nocturnal polyuria (NP) is increased urine production at night. Nocturnal polyuria exists when the nocturnal urine volume represents >20% of the 24 hour voided volume in younger adults and >33% in patients aged 65 years and over. The 24 hour voided volume is normal.13 Nocturnal polyuria is present in up to 80% of patients with nocturia and can be easily overlooked if a FVC is not completed.7
Nocturnal polyuria can be caused by low nocturnal ADH levels. This can occur from central nervous system lesions that affect the hypothalamicpituitary axis, but it can also occur in elderly patients without specific central nervous system pathology.
Peripheral oedema can result in NP as the accumulated fluid is redistributed to the intravascular compartment once recumbent in bed. This excess fluid is then excreted by the kidneys.11
Obstructive sleep apnoea (OSA) causes NP due to increased secretion of atrial natriuretic peptide, which induces a natriuresis.10,11
Bladder storage disorders
Lower urinary tract pathology may present with frequent small volume voids often in association with other lower urinary tract symptoms (LUTS). Any structural or functional pathology that affects the reservoir capacity of the bladder can increase voiding frequency. Potential pathologies are listed in Figure 1.
Bladder storage disorders tend to predominate among younger patients while NP predominates in older patients.14 The presence of other LUTS suggests lower urinary tract pathology, however many patients (especially the elderly) have a mixed pattern of disease with multiple contributing factors.15 Nocturia in males should not be assumed to be due to benign prostatic hypertrophy (BPH). Nocturia is the least specific symptom of BPH and prostatic pathology is unlikely to exist in the absence of voiding symptoms such as poor stream and hesitancy.7
Sleep disorders
Patients who constantly wake at night for other reasons may feel the need to void. These patients pass small volumes of urine and have a similar FVC to a bladder storage disorder.1 It is important to determine whether the primary reason for waking was to void.10 If a sleep disorder is suspected, referral to a sleep physician should be considered.
Clinical evaluation
It is important to enquire about nocturia as it is often under reported. Many patients will not present with the complaint of nocturia, but with fatigue related symptoms. Some may not raise the issue as they feel embarrassed, do not realise treatment is available or may perceive it as a normal part of ageing.9
Given the wide range of possible aetiologies, the key diagnostic tool is the FVC. History and examination give important clues to the underlying pathological processes (Table 2).
Table 2. Recommended clinical assessment of patients with nocturia
| History |
- Daily fluid consumption
- Fluid consumption before going to bed
- Alcohol and caffeine consumption
- Presence of other LUTS
- Voiding – hesitancy, weak stream, incomplete emptying
- Storage – frequency, urgency
- Incontinence
- Typical sleeping habits
- Medical history – especially heart failure, renal failure, diabetes, obstructive sleep apnoea, gastro-oesophageal reflux, lung disease, endocrine evaluation (eg. thyroid disease)
- Symptoms and signs of the above conditions
- Symptoms of obstructive sleep apnoea – history of snoring, collaborative history from partner
- Medications
|
| Examination |
- Blood pressure
- Waist circumference and weight assessment
- Cardiovascular examination – signs of congestive cardiac failure
- Neurological examination of sacral integrity – perineal sensation/ anal wink and other neurological evaluation (where indicated)
- Digital rectal examination
|
| Investigations |
- Frequency-volume chart (voiding diary)
- Urinalysis +/– midstream urine
- Urea, electrolytes and creatinine
- Serum glucose
- Lipids
- Prostate specific antigen (if clinically relevant)
- Urinary tract ultrasound (if clinically relevant)
|
Medications can precipitate nocturia through several mechanisms, hence medication review is warranted in all patients (Table 3).10 Investigations include screening for renal failure, urinary tract infection and diabetes (Table 2).10
Table 3. Medications implicated in nocturia
| Effect |
Medication |
| Increased urine output |
- Diuretics
- SSRIs
- Calcium channel blockers
- Lithium (nephrogenic diabetes insipidus in 40% of users)
- Tetracylines
|
| Precipitate insomnia and other central nervous system effects |
- Stimulants
- Centrally active antihypertensives (α-blockers, β-blockers, methyldopa)
- Decongestants
- Psychotropic medications
- Parkinsonian medications
- Phenytoin
|
| Precipitate LUTS |
- Ketamine
- Cyclophosphamide
|
Management
Management should be directed to correctly identifying the underlying aetiology. Most individual treatments reduce nocturia by less than one episode per night compared to placebo. A combined approach consisting of behavioural, medical and surgical interventions, where appropriate, has greater benefit.15
Lifestyle modifications
Appropriate lifestyle modification should be recommended (Table 4).
Table 4. Recommended lifestyle modifications to manage nocturia
- Avoid drinking caffeine and alcohol in the evening
- Limit excessive fluid and food intake 3 hours before bedtime
- Evening leg elevation
- Pre-emptive voiding
- Medication timing (moving diuretic doses to the mid-afternoon. Calcium channel blockers, especially hydropyridines, to be administered in the morning rather than evening for symptomatic patients)
- Optimisation of the sleep environment
- Exercise program
- Attention to psychological, financial and family concerns
|
The sleep environment should be optimised with attention to room temperature, noise, lighting and consistent times of going to bed. Regular exercise can lead to deeper sleep and increases the bladder volume arousal threshold.10 Dealing with psychological factors, financial and family issues have been shown to improve sleep quality.16,17 Lower limb elevation before going to bed is useful in patients with peripheral oedema.15 Dosage times for diuretics should be moved from evening to mid afternoon.
Sedatives are best reserved for sleep disorders rather than nocturia, although a short acting hypnotic may be beneficial for patients who wake early in the night and find it difficult to return to sleep.10
Specific treatment
Global polyuria
Primary polydipsia can be effectively managed with fluid restriction. Patients with poorly controlled diabetes mellitus or diabetes insipidus will benefit from an endocrinology assessment.
Nocturnal polyuria
Underlying medical causes (eg. congestive cardiac failure, chronic kidney disease and obstructive sleep apnoea [OSA]) should first be identified and treated. If patients already have these conditions, new onset nocturia should prompt a management review. Patients suspected of OSA will benefit from referral to a sleep physician for a sleep study and tailored treatment.
Nasal desmopressin (an antidiuretic hormone [ADH] analogue) is currently used in Europe for the treatment of NP. It is hypothesised to rectify defects in circadian secretion of ADH and has been shown to reduce the number of nocturnal voids in patients with NP.18 However, elderly patients are at risk of hyponatraemia and use is contraindicated in congestive cardiac failure and renal failure.19 Desmopressin is not currently listed for use in Australia for treating nocturia, except where associated with diabetes insipidus.
Bladder storage disorders
These patients require urological assessment. The urologist may perform further investigations such as uroflowmetry/postvoid residual urine measurement, urodynamics or cystoscopy, depending on the clinical scenario.
In male patients with bladder outlet obstruction, treatments are less effective at improving nocturia than for obstructive LUTS such as slow stream and hesitancy.20,21 α-blockers (eg. prazosin, tamsulosin, alfuzosin) have shown a modest improvement in the number of nocturic episodes.20 Gains tend to be noticed almost immediately and correlate with the severity of nocturia.20 α-blockers must be prescribed with caution, especially in the elderly due to associated postural hypotension.
Prostatic surgery (eg. transurethral resection of prostate [TuRP], laser prostatectomy) in appropriately selected patients with bladder outlet obstruction reduces episodes of nocturia.22 Surgery appears to be more effective in younger patients and in those with more severe obstruction.10 Surgical results are less successful in mixed nocturia, especially those with NP.
A trial of antimuscarinic therapy (eg. oxybutynin, tolterodine, darifenacin, solifenacin) can be of benefit to patients with symptoms suggestive of overactive bladder. Patients who derive most benefit are those whose nocturic episodes are associated with urgency.7
Key points
- Nocturia is common over the age of 40 years and its prevalence is similar in both males and females.
- Nocturia can have a profound impact on quality of life and has been shown to be a predictor of mortality.
- Prostatic disease should not be assumed to be the cause of nocturia in men.
- Nocturia may signify the presence of an undiagnosed medical condition or suboptimal management of a known medical condition. Cardiovascular, respiratory, renal, neurological and endocrine causes may be relevant.
- A multimodal approach to treatment yields the most effective results because aetiology is often multifactorial.
- Appropriate behavioural modifications should be recommended to all patients.
Conflict of interest: A/Prof Prem Rashid has acted as a consultant for Coloplast, AstraZeneca, Abbott, Ipsen and Sanofi Aventis pharmaceuticals, as well as, the Neotract Corporation. He was a Preceptor in Advanced Laparoscopic Urology with Professor Inderbir S. Gill, (then) Head of the Section of Laparoscopic and Robotic Surgery and Chairman, Glickman Urological Institute, Cleveland Clinic Foundation via a 2006 grant from the Australasian Urological Foundation.
Acknowledgements
Thanks to Dr Mohan Vattekad, Director of Nephrology, Port Macquarie Base Hospital, for his helpful comments on the content of this article.