Case
A man, 30 years of age, presented with an intensely pruritic eruption that had been evolving for approximately two months. The rash was characterised by clusters of translucent, tense vesicles located symmetrically on the extensor surfaces of the arms and legs (Figure 1A, B), and by erythematous papules and small plaques on the buttocks (Figure 1C). Some of the lesions were excoriated with bloodstained crusting. The patient reported a loss of approximately 5 kg in weight, and several episodes of diarrhoea and abdominal discomfort in the previous year. The patient was otherwise healthy and denied any history of fever, photosensitivity or symptoms in other family members. Four punch biopsies of lesional and perilesional skin from the arms and buttocks were taken for routine histopathology and direct immunofluorescence (DIF).
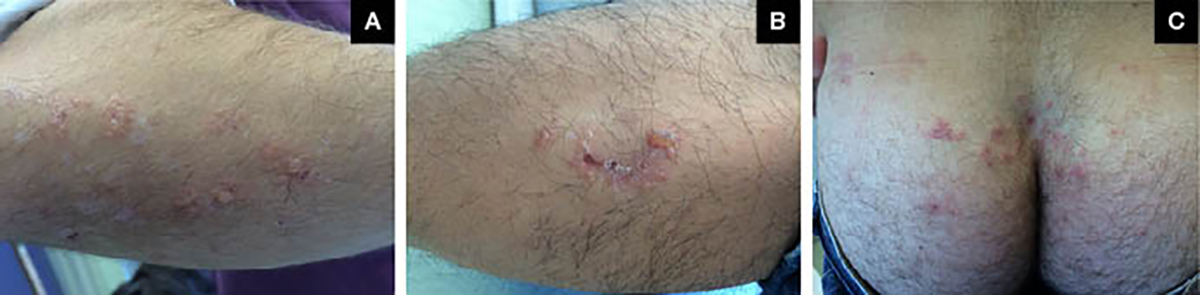
Figure 1. Clinical appearance of the lesions
A. Right forearm; B. Right shin; C. Buttocks.
Question 1
What differential diagnoses should be considered in this case?
Question 2
What is the most likely diagnosis and aetiology of this condition?
Question 3
How is this condition diagnosed?
Question 4
What is the appropriate management of this condition and prognosis?
Answer 1
The main diagnoses to be taken into account in this patient are listed in Table 1.1–3
Answer 2
The most likely diagnosis is dermatitis herpetiformis, which is the result of an immunological response of the gut mucosa to persistent stimulation by dietary gluten.2 The majority of patients with dermatitis herpetiformis have evidence of coeliac disease, but less than 10% have symptoms or signs of malabsorption.4 Among patients with coeliac disease, 15–25% will develop dermatitis herpetiformis.3 The pathophysiology of dermatitis herpetiformis involves gluten as an environmental trigger, genetic factors and dysregulation of the immune system.5 A diet containing gluten causes the development of immunoglobulin A (IgA) antibodies against gluten-tissue transglutaminase, which is found in the gut, leading to a cross-reaction between these antibodies and epidermal transglutaminase.6 There is an association with human leukocyte antigens (HLA) DQ2 and DQ8.5 Some patients also describe a personal or family history of other autoimmune disorders such as Hashimoto’s thyroiditis, pernicious anaemia, type 1 diabetes mellitus and alopecia areata.1
Answer 3
The diagnosis of dermatitis herpetiformis relies on skin biopsy results. Routine histopathology of a dermatitis herpetiformis skin lesion showing an inflammatory infiltrate mainly composed of neutrophils is not diagnostic, as other bullous diseases may show similar findings.1,5 Thus, to make a diagnosis, biopsies in two different locations – one from lesional skin for routine histopathology and another from perilesional skin for DIF – should be taken: DIF will show deposits of IgA with epidermal transglutaminase complexes in the papillary dermis.6 Serological tests, such as anti-transglutaminase and anti‑endomysial antibodies, have high specificity and sensitivity for dermatitis herpetiformis, and are a useful adjunct for diagnosis.7,8
Answer 4
Treatment of dermatitis herpetiformis is based on a gluten-free diet for life, which leads to healing of the rash and enteropathy.7 However, in the first months after the diagnosis, several drugs can be used.1 Dapsone is the initial treatment of choice, but requires monitoring for side effects, especially haemolytic anemia and methaemoglobinaemia.10 Before starting the patient on dapsone, a full blood count, glucose-6-phosphate dehydrogenase (G6PD) enzyme levels, and renal and liver function tests should be performed, followed by a full blood count every month.7 Dapsone cannot be used in patients with G6PD deficiency because of the high-risk of haemolysis.10 In our opinion, dapsone should only be used by physicians who have experience with this drug. Sulfasalazine is an alternative in patients who cannot tolerate, or are unresponsive to, dapsone. Potent topical steroids are also helpful in decreasing the pruritus and preventing the appearance of new lesions.1
Patients require follow-up with a multidisciplinary team that includes a dermatologist, gastroenterologist and dietitian.7 Patients who adhere and respond well to a gluten-free diet and medication have a good prognosis.3,7 Progression to intestinal lymphoma was seen in several studies, although it tends to occur predominantly in individuals who do not follow a gluten-free diet.2,10
Table 1. Main differential diagnoses to be considered with usual clinical manifestations1–3
|
Disease
|
Differentiating features
|
|
Dermatitis herpetiformis
|
- Pruritic, polymorphic, grouped and symmetrical lesions consisting of erythema, urticarial plaques, papules, vesicles and blisters, followed by erosions, abrasions and hyperpigmentation
- Extensor surfaces of knees/elbows, shoulders, buttocks, sacral region and scalp
- Gluten-sensitive enteropathy on endoscopic biopsy
- More frequent in males and adults aged 15–40 years
|
|
Pompholyx eczema
|
- Abrupt onset of small, clear vesicles or bullae on the palms and/or soles
- Prodromal itching and burning sensation on palms/soles
- Vesicles and bullae dry out and resolve without rupturing; desquamation two to three weeks after
|
|
Infections
|
Scabies
- Pruritus worse at night
- Burrows are pathognomonic
Viral
- Herpes zoster: Unilateral vesicular eruption of one to three dermatomes, painful or pruritic lesions, rarely bilateral or disseminated
- Herpes simplex virus (HSV) infection: Clusters of sometimes painful fluid-filled blisters or sores, classic sites are lower back, buttocks and thighs in HSV-2 infection, and face in HSV-1
Bullous impetigo
- Caused by coagulase positive Staphylococcus aureus
- Blisters on face (around mouth, nose) or site of trauma that rupture easily leaving crusted edges and honey-coloured crusts
|
|
Erythema multiforme
|
- Associated with HSV, infections or drugs
- Concentric ‘target’ lesions
- Palms, neck and face; mucosal involvement in up to 70% of cases
|
|
Rare
|
Bullous pemphigoid
- More prevalent in elderly patients with neurological disease or psoriasis
- Large, tense bullae that rupture, forming crusted erosions
- Any part of the skin surface, predilection for flexural areas
Bullous systemic lupus erythematosus
- Widespread vesiculobullous eruption that develops very fast, ranging from large, tense bullae to small, grouped vesicles
- Usually in the superior part of the trunk, proximal upper extremities, neck and face
- Pruritus of variable intensity
- Can present with systemic lupus symptoms
Linear IgA dermatosis
- May have prodromal or transitory pruritus
- Clear or haemorrhagic vesicles or bullae on normal, erythematous, or urticarial skin. Arranged in rings or a ‘cluster of jewels’
- Ocular symptoms (eg grittiness, burning, discharge) are common
|
Case follow-up
In our patient, histopathological examination and DIF were suggestive of dermatitis herpetiformis. Anti-endomysial and anti-transglutaminase levels were elevated, and G6PD was within the normal range. A small bowel biopsy was performed, which confirmed that the patient had coeliac disease. The patient started a gluten-free diet and oral dapsone (100 mg/day), and at the three-week visit, we observed a complete regression of the skin lesions. The patient maintained the oral dapsone dose for 10 months, and it was then slowly tapered off over the next six months without relapse of the dermatosis.
Key point
Authors
Ana Baptista MD, Family Medicine Trainee, Unidade de Saúde Familiar (USF) Viseu-Cidade, Viseu, Portugal. nibaptista@gmail.com
Sofia Madanelo MD, Family Medicine Trainee, USF Santa Joana, Aveiro, Portugal
Paulo Morais MD, Consultant Dermatologist, Department of Dermatovenereology, Centro Hospitalar Tondela-Viseu, Viseu, Portugal
Competing interests: None.
Provenance and peer review: Not commissioned, externally peer reviewed.