Laryngopharyngeal reflux (LPR) is a distinct entity to gastro-oesophageal reflux disease (GORD). It is defined by the retrograde passage of gastric contents beyond the upper oesophageal sphincter, with contamination of the larynx, pharynx and lungs. In susceptible patients, this exposure causes mucosal injury, damage to ciliated respiratory epithelium and mucus stasis, which result in a troublesome array of symptoms and signs termed LPR.
Prevalence
Half of the laryngeal complaints referred to ear, nose and throat (ENT) services are ultimately diagnosed as LPR.1 Meta-analysis of pH studies reveals reflux in 63% of patients with LPR, compared with 30% in controls,2 and reflux is seven times more frequent in this group.3 Changes in pH suggesting reflux occur in 50% of patients with hoarseness, 64% with globus, 55% with chronic cough and 35% with dysphagia.1,4
Diagnostic significance
LPR is one of the most common and important disorders of upper airway inflammation5 and predicts oesophageal adenocarcinoma, laryngeal granuloma, polyps, Reinke’s oedema, stenosis and chronic laryngitis.6–8 LPR is correlated with laryngeal cancer, although causation is unconfirmed. Despite this, LPR remains under-diagnosed and under-treated.9 Its diagnosis is plagued by non-specific symptoms and signs, and by overlap with differential diagnoses such as upper respiratory infection, rhinitis, asthma, smoking, vocal abuse and allergy.
LPR without heartburn: A pathophysiological and conceptual dilemma
The exact mechanism for LPR is unclear. It is hypothesised that the injury occurs directly (via exposure to gastric acid, pepsin and bile salts) or indirectly (via repetitive trauma from vagally mediated cough and throat clearing).4,10 What is clear, however, is that GORD and LPR share only limited overlap in symptoms, signs and patient characteristics (Table 1). This causes great consternation for patients and clinicians, who are frequently confronted with ‘heartburn denial’ when proposing the diagnosis. The ‘LPR without heartburn’ disconnect thwarts patients’ understanding, limits diagnosis acceptance, and lessens compliance with treatment recommendations.
When counselling patients, it is essential to note that less than half of patients with pH-proven LPR report ‘heartburn’.11 This paradox is explained by the fact that the oesophagus has a range of mucosal protections that are absent within the laryngopharynx. These include lower oesophageal sphincter tone, peristaltic propulsion, mucosal tissue resistance and active extracellular bicarbonate.12 These mechanisms do not exist in the larynx. Some protection is afforded by carbonic anhydrase isoenzyme III (CAI III), which aids bicarbonate production. However, laryngeal CAH III expression is suppressed reversibly by acid and irreversibly by pepsin.13 Furthermore, CAI III is absent in 64% of biopsies taken from patients with LPR.14 Patients with LPR are also deficient in salivary epidermal growth factor (EGF), compared with healthy controls.15
Thus, differences between sites and between patients create a range of susceptibilities to LPR. The larynx is relatively more vulnerable to caustic injury and has a lower threshold at which ‘physiological reflux’ causes symptoms. In turn, LPR often resolves more slowly than GORD, even with appropriate treatment.
Table 1. LPR and GORD: Distinguishing characteristics
|
Laryngopharyngeal reflux
|
Gastro-oesophageal reflux disease
|
|---|
|
Breach upper oesophageal sphincter
|
Breach lower oesophageal sphincter
|
|
Hoarseness, globus, cough, ‘thick mucus/postnasal drip’, throat pain
|
Acid reflux, heartburn, chest pain
|
|
Symptoms worse while upright
|
Symptoms worse while recumbent
|
|
No association with obesity/high body mass index (BMI)
|
Associated with obesity/high BMI
|
|
Patients usually deny heartburn, reflux
|
Patients report heartburn and reflux
|
Diagnosis
Controversy exists around the best diagnostic approach for LPR. In primary care, the diagnosis may be reached clinically according to symptoms and empirical treatment response, following the judicious exclusion of red flags (Box 1).
In the absence of red flags (Box 1), LPR warrants consideration in patients presenting with non-specific laryngopharyngeal symptoms not explained by alternative diagnoses, even if a patient vehemently denies ‘reflux’.
Box 1. Red flags: Seek early specialist review for laryngopharyngeal visualisation
- Significant risk factors for head and neck malignancy (eg heavy smoking/alcohol)
- Prior history of head and neck malignancy
- New onset, constant hoarseness in smoking patient
- Unexplained weight loss
- Haemoptysis
- Highly lateralised symptoms
- Severe pain
- Referred otalgia
- ‘Obstructive’ dysphagia +/– regurgitation
|
Symptoms
Globus pharyngeus and hoarseness are the most common LPR symptoms. Globus may be described as throat tightness, discomfort or ‘something stuck’, whereas hoarseness tends to be relapsing-remitting and manifests as reduced vocal quality, power, clarity or stamina.
Throat discomfort and ‘mucus that cannot be cleared away’ are commonly reported and may be erroneously attributed to postnasal drip. Where postnasal drip is reported in the absence of other sinonasal symptoms (especially if a trial of nasal saline and corticosteroid has been ineffective), LPR warrants consideration.
Dysphagia or a mildly ‘troublesome swallow’ may be described as food residue left behind or new difficulties swallowing bread or tablets. It is essential to distinguish this from true obstructive dysphagia – with impaction and regurgitation – which may indicate pouch, tumour or stricture.
Less commonly, patients present with lower respiratory chest tightness or wheeze, or with laryngospasm – a sudden and alarming closure of the airway with temporary inability to inspire.
These primary symptoms often cause maladaptive compensatory manoeuvres, resulting in the oft-seen ‘secondary symptoms’ of habitual dry cough, throat clearing and repetitive swallow, which propagate mucosal trauma.
Signs
Above the larynx, signs of LPR include halitosis, dished-out dental erosions and pharyngeal ‘cobblestoning’. Patients with recurrent pharyngitis, otitis media and chronic rhinosinusitis also warrant consideration of LPR as a unifying diagnosis, as these conditions are suspected (although uncertain) disease associations.
Investigation
Laryngoscopy is the mainstay of ENT assessment. Accuracy is hindered by variable inter-rater reliability, however, as the more commonly available flexible nasendoscopy is more sensitive but less specific than its rigid counterpart.16 Laryngoscopic findings may be quantified using the ‘Reflux finding score’ (Table 2), although this is not used routinely in Australian practice. The most convincing laryngeal finding is posterior laryngitis, or pachyderma. Translated literally to mean ‘like an elephant’, pachyderma denotes rough, thickened inter-arytenoid mucosa. Pseudosulcus vocalis, a linear indentation along the medial edge of the vocal cord, carries 70% sensitivity and 77% specificity for diagnosing LPR.17
Barium swallow may reveal a hiatus hernia or pharyngeal pouch, and should be ordered if a patient reports food impaction with regurgitation, halitosis, aspiration, recurrent lower respiratory tract infection, cervical borborygmi or a compressible neck mass. It may also reveal stenosis caused by an oesophageal web, bar or tumour, or extrinsic compression caused by a cervical or thoracic lesion. Importantly, a barium swallow that is ‘non-diagnostic for reflux’ does not exclude LPR, given its inherently low diagnostic yield.
Oesophageal endoscopy has limited utility in the workup of LPR – while finding oesophagitis may lend support to the diagnosis, <30% of LPR patients have histological oesophagitis.12
Oesophageal pH and manometry testing are usually reserved for refractory or complicated cases, and for those patients under consideration for surgery. pH monitoring is the gold standard for diagnosing GORD, but is less sensitive for LPR, and may not predict response to therapy reliably. Worn over 24 hours, with results interpreted in conjunction with diarised meal and sleep times, pH monitoring localises the site, duration and frequency of reflux events.
Management
Lifestyle modifications
Vigorous lifestyle counselling is essential as isolated medical therapy often fails. Patients should abstain from eating for three hours before bedtime and allow the stomach to fully empty before lying flat. A ‘tipping teapot’ diagram depicting the relationship between a full stomach and oesophagus can be illuminating.
Evening meals should be the smallest of the day, and ‘trigger foods’, including fried foods, tomato, citrus, mint, chocolate, acidic dressings, juices, coffee, carbonated drinks and alcohol, as well as smoking, should be avoided at this time. Patients should avoid ‘gulping and lounging’. They should eat their evening meal slowly, then remain upright until bedtime. A careful history will tease out any contributory ‘late night cuppa’ or ‘midnight snack’.
Tight clothing should be avoided, and patients who are overweight should reduce their waistline. Central obesity causes fat deposition between the distal oesophagus and diaphragm, compromising lower sphincter function. Patients should elevate their bedhead by 15 cm (paired house bricks or phonebooks are useful). Importantly, stacked pillows are often inadequate as they may result in neck flexion, rather than true chest elevation.
Table 2. Reflux finding score27
|
Pseudosulcus vocalis (infraglottic oedema)
|
0 = Absent
2 = Present
|
|
Ventricular obliteration
|
0 = None
2 = Partial
4 = Complete
|
|
Erythema/hyperaemia
|
0 = None
2 = Arytenoids only
4 = Diffuse
|
|
Vocal fold oedema
|
0 = None
1 = Mild
2 = Moderate
3 = Severe
4 = Polypoid
|
|
Diffuse laryngeal oedema
|
0 = None
1 = Mild
2 = Moderate
3 = Severe
4 = Obstructing
|
|
Posterior commissure hypertrophy
‘Pachyderma’
|
0 = None
1 = Mild
2 = Moderate
3 = Severe
4 = Obstructing
|
|
Granuloma / granulation
|
0 = Absent
2 = Present
|
|
Thick endolaryngeal mucous
|
0 = Absent
2 = Present
|
|
|
Total score: /26
|
|
|
A score exceeding 13 is considered abnormal
|
|
Reproduced from Belfasky PC, Postma GN, Koufman JA. The validity and reliability of the reflux finding score (RFS). Laryngoscope 2001;111:1313–17, with permission from John Wiley and Sons.
|
Table 3. Reflux symptom index28
|
Within the last month, how did the following problems affect you? 0, no problem; 5, severe problem
|
|
|
Hoarseness or a problem with your voice
|
0 – 1 – 2 – 3 – 4 – 5
|
|
Clearing your throat
|
0 – 1 – 2 – 3 – 4 – 5
|
|
Excess throat mucus or post-nasal drip
|
0 – 1 – 2 – 3 – 4 – 5
|
|
Difficulty swallowing food, liquids or pills
|
0 – 1 – 2 – 3 – 4 – 5
|
|
Coughing after you ate or after lying down
|
0 – 1 – 2 – 3 – 4 – 5
|
|
Breathing difficulties or choking episodes
|
0 – 1 – 2 – 3 – 4 – 5
|
|
Troublesome or annoying cough
|
0 – 1 – 2 – 3 – 4 – 5
|
|
Sensation of something sticking in your throat or a lump in your throat
|
0 – 1 – 2 – 3 – 4 – 5
|
|
Heartburn, chest pain, indigestion, or stomach acid coming up
|
0 – 1 – 2 – 3 – 4 – 5
|
|
|
Total
|
|
The Reflux Symptom Index provides a useful score-card for establishing the diagnosis, counselling patients over time, and tracking gradual symptom resolution. Scores exceeding 13 are considered abnormal
Reproduced from Belfasky PC, Postma GN, Koufman JA. Validity and reliability of the reflux symptom index (RSI). J Voice 2002;16(2):274–77, with permission from Elsevier.
|
Anxiety and depression
Given the historical origin of globus hystericus, several studies have attempted to elucidate the role of psychiatric disease among patients with LPR. Many have shown no relationship, and thus, it has become unfashionable to enquire about patients’ mental health when discussing aerodigestive symptoms. However, it is important to remember that patients with LPR report an overall lower quality of life and greater psychological disturbance than healthy controls; 30% of patients with LPR-like symptoms report anxiety, compared with 6% of healthy controls. Furthermore, this discrepancy – across all parameters of mental health – was shown to improve after successful treatment of LPR.18
Patients with significant psychiatric stressors experience greater perceived heartburn without a measurable increase in objective reflux,19 and it seems reasonable to postulate a similar phenomenon in LPR. Critically, psychiatric comorbidity reduces the validity of the ‘reflux symptom index’ (Table 3) when making an LPR diagnosis and monitoring the response to treatment. As diagnostic tools, the reflux symptom index and reflux finding score are more valid when patients with psychiatric disease are excluded.20 Thus, although the exact nature of the relationship is unclear, patients with LPR present the astute GP with a valuable opportunity to discuss both mental and laryngopharyngeal health.
Medications
Medications include histamine H2 receptor antagonists, proton pump inhibitors, prokinetics (for patients with known oesophageal dyskinesia), mucosal cytoprotectants and tricyclic antidepressants (for those in whom laryngeal neuropathy is suspected). Proton pump inhibitors show greater control than drugs in other classes.21,22 Of the proton pump inhibitors, omeprazole is the most widely studied, but newer agents such as rabeprazole and pantoprazole offer once-daily dosing and have shown additional efficacy.23,24
Although controversy exists around proton pump inhibitor dose, frequency and duration of therapy, a recent meta‑analysis of 13 randomised controlled trials confirmed that the reflux symptom index for patients prescribed proton pump inhibitors was significantly improved, compared with those receiving placebo.25 It should be noted that proton pump inhibitors, while extremely effective for GORD, where they reduce reflux by 80% and resolve oesophagitis in 80–90% of patients, are less reliable in reducing LPR. It is generally accepted among ENT specialists that higher doses of proton pump inhibitors and a longer treatment duration are indicated prior to accepting treatment failure.26
Commencing medical therapy
In the primary care sector, it is reasonable to commence a low-dose proton pump inhibitor regime (eg omeprazole 20 mg once daily) 30 minutes before meals, in conjunction with strict lifestyle modifications, and continue this over an eight-week empirical trial (Figure 1). While proton pump inhibitors are safe and well tolerated, common side effects, including headache, abdominal pain and bloating, diarrhoea, and nausea, affect up to 2% of patients. These should be discussed at the commencement of medical therapy. Completing a reflux symptom index with the patient at the initial review aids surveillance, and completing a second reflux symptom index at eight weeks allows tracking of symptom resolution (which may be present but incomplete at this point). The proton pump inhibitor dose may then be maintained, or increased to either 20 mg BD or 40 mg OD for a further eight weeks.
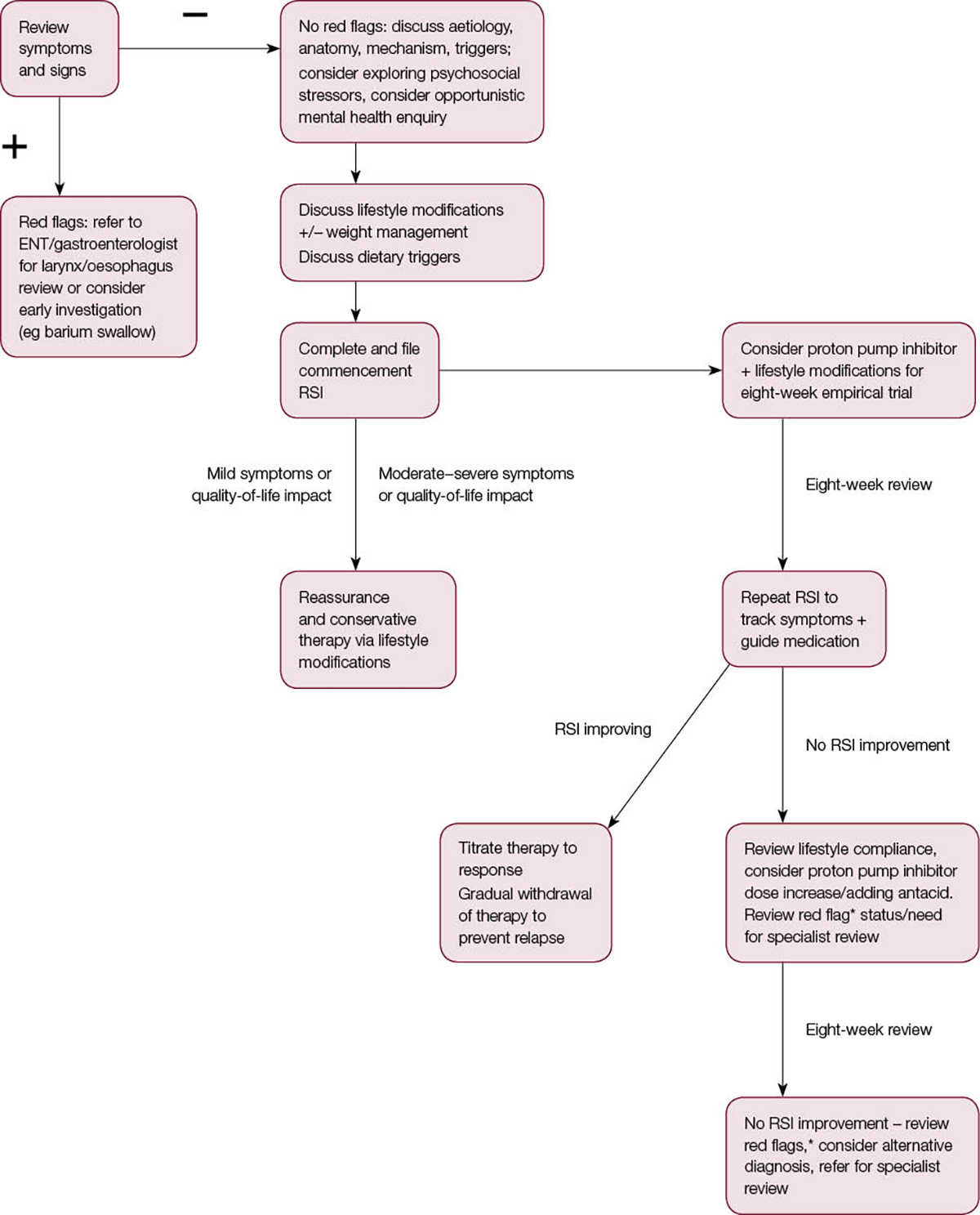
Figure 1. Suggested algorithm for suspected laryngopharyngeal reflux
*Red flags: refer to ENT specialist or gastroenterologist for larynx/oesophagus review or consider early investigation (eg barium swallow) ENT, ear, nose and throat; RSI, reflux sympton index
During the second eight-week period, it is the author’s practice to add an antacid 30 minutes after meals. Internationally, a proton pump inhibitor regime of 40 mg twice daily is commonplace, but as this meets with Pharmaceutical Benefits Scheme (PBS) limitations in Australia, local ENT specialists occasionally add an evening dose of a histamine H2 receptor antagonist (eg ranitidine), while accepting there is limited evidence for this.
Treatment failures
Patients who fail to improve may warrant a trial of a tricyclic antidepressant to address laryngeal neuropathy, or benefit from dual-probe pH/impedance testing, which quantifies reflux status and may identify patients with non-acid or bile salt reflux.
Non-responders should be referred for ENT and/or general surgical review. In carefully selected patients, Nissen fundoplication yields 80–90% cure, with 91% control maintained at 10 years.12 Surgery may be indicated in patients with inadequate response to maximal medical therapy, young patients not wishing to commit to lifelong continuous or high‑dose proton pump inhibitor therapy, or in response to patient preference where the diagnosis is clear.
Conclusion
LPR is one of the most common and important disorders of aerodigestive dysfunction, yet is under-diagnosed and under-treated in primary care and specialist sectors. The disorder has a significant impact on quality-of-life , yet diagnosis is plagued by variable symptoms and signs, and a lack of definitive diagnostic indicators. In particular, limited overlap between GORD and LPR causes confusion among clinicians and non-compliance among patients.
LPR diagnosis requires a high index of suspicion and careful history-taking, but in the absence of red flags, can often be arrived at clinically. Although not life‑threatening, aerodigestive dysfunction – particularly globus – is disproportionately troubling to patients, and a clinician well schooled in LPR is well positioned to offer reassurance, identify contributory lifestyle factors, and educate and engage patients in a multifaceted treatment plan.
Author
Kristy Fraser-Kirk BA (Psych), MBBS (Hons), FRACS (OHNS), Acting Director, ENT Department, Sunshine Coast University Hospital and Health Service, Brisbane, Qld. kristyfraserkirk@me.com
Competing interests: None.
Provenance and peer review: Not commissioned, externally peer reviewed.