Obstructive sleep apnoea (OSA) is a common disease that has an estimated prevalence of 24% in men and 9% in women.1 OSA is a complex disease process that involves collapse of the upper airway during sleep and subsequent reduction or cessation of airflow.2 It is associated with a variety of medical consequences, such as excessive daytime somnolence, neurocognitive impairment, cardiovascular disease and reduced quality of life.3 The economic costs are substantial as patients with OSA use more healthcare resources, and have greater risks of motor vehicle accidents and work-related injuries.3 Interestingly, one study estimated the total yearly cost of treating OSA in Australia to be approximately $657 million.4
Continuous positive airway pressure (CPAP), generally administered through the nose, is the primary treatment for OSA and is the recommended first-line treatment for patients with moderate-to-severe forms of the disease.2,5 However, some patients are unable to tolerate CPAP because of mask discomfort, while others cannot accept sleeping throughout the night attached to a mechanical device.2 Objective data collected from patients have shown a compliance rate with CPAP of <50%.6 In these cases, surgical management aimed at addressing anatomical obstruction may be warranted.
Selection of patients for surgery by history and examination
The assessment should begin with a thorough history to identify patients with an increased risk of OSA.7 The examination should document weight, height and body mass index (BMI), as obesity is an independent risk factor.7,8 The nose should be examined anteriorly for septal deviations, spurs, perforations and hypertrophic inferior turbinates. The oral cavity should be inspected for macroglossia, enlarged uvula, hypertrophic tonsils and an elongated soft palate. The modified Mallampati classification system is a useful guide to categorise the relative crowding of the oropharynx (Figure 1).7 Finally, the neck should be palpated for any masses.
In the consulting room, otolaryngologists are able to perform flexible nasal endoscopy under local anaesthesia to assess the upper aerodigestive tract for potential sites of narrowing.7,8 Flexible nasal endoscopy enables clinicians to identify abnormalities such as nasal polyps, adenoid hypertrophy, lingual tonsillar hypertrophy, tongue-base collapse, hypopharyngeal constriction and laryngeal lesions. The use of a flexible nasoendoscopy assists the surgeon with clinical diagnosis, assessment, surgical planning and outcome assessment.9
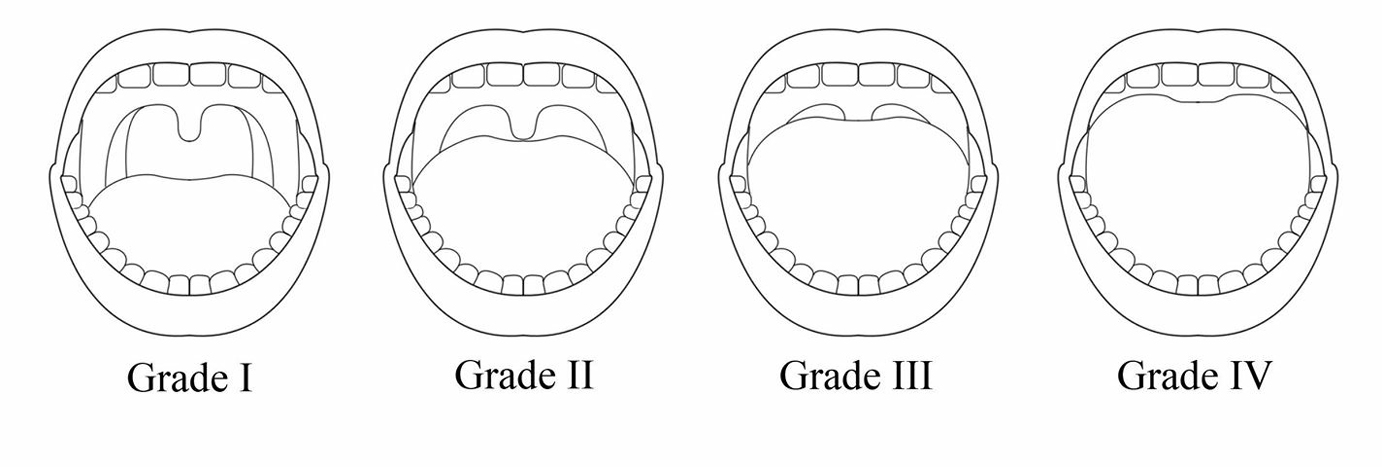 |
Figure 1. The modified Mallampati classification system
Grade I allows full visibility of the soft palate, uvula and tonsils; grade II allows visibility of the hard and soft palates, uvula and upper tonsils; grade III allows visibility of the hard and soft palates and base of uvula; grade IV allows visibility of the hard palate only |
Surgical options for OSA
For patients who are deemed suitable for CPAP but unable to tolerate it, or for patients who are unwilling to accept CPAP as a permanent form of management, surgery may be useful. These patients should be referred to an otolaryngologist for further assessment (Figure 2). Results from a study conducted in middle-aged men with severe OSA who were intolerant of CPAP show that surgery is cost-effective.10 This study was based on a model of OSA that accounted for observed increased risk of stroke, cardiovascular disease and motor vehicle accidents. OSA requires long-term, multidisciplinary management. Surgery may be offered to patients, depending on the severity of the disease and the patient’s anatomy, risk factors and preferences.11 The following section discusses the different surgical options available for OSA.
Procedures to improve nasal patency and breathing
Opening an obstructed nose will not improve OSA; however, it may improve nasal breathing, CPAP tolerance and compliance.12 Indeed, patients with reported symptoms of nasal obstruction, dryness or rhinorrhoea may benefit from nasal surgery to improve CPAP tolerance.7 Septoplasty, turbinate reduction or both may be performed to establish nasal patency and airway stability.
Procedures to improve retropalatal obstruction
Uvulopalatopharyngoplasty
Uvulopalatopharyngoplasty (UPPP) is a procedure that removes the obstructing tissues of the soft palate, lateral pharyngeal walls and tonsils in order to widen the pharyngeal airway.7,12 There are various approaches to this procedure, including relocation pharyngoplasty, lateral pharyngoplasty, Z-palatoplasty and palatal advancement. UPPP is indicated for patients with isolated retropalatal obstruction; but unlike nasal surgery, it is not recommended as a procedure to improve CPAP tolerance and compliance.7,12 On the contrary, UPPP results in large oral leaks and decreased compliance with CPAP.7 Thus, otolaryngologists must be careful to limit palatal resection in patients who may require CPAP following the procedure.
UPPP may be associated with significant complications including velopharyngeal insufficiency, dysphagia and nasopharyngeal stenosis.12 The overall success rate is around 40% with UPPP surgery alone.12
Uvulopalatal flap
The uvulopalatal flap (UPF) is a modification of the UPPP and carries similar risks.12 It is also indicated for patients with isolated retropalatal obstruction. However, it is preferred to UPPP for most cases as it provides the same anatomical results but is associated with a lower risk of nasopharyngeal incompetence and less post-operative pain.12 This is achieved by the creation of a reversible flap, whereby the uvula is pulled away from the pharyngeal wall and reflected back towards the soft palate, thus expanding the oropharyngeal space. For the management of OSA, results from UPF are comparable to those of UPPP.12
Procedures to improve tongue-base and hypopharyngeal obstruction
Tongue-base, temperature-controlled radiofrequency and coblation
Tongue-base, temperature-controlled radiofrequency is a procedure that involves the application of a temperature-controlled radiofrequency probe to multiple locations in the tongue base.7,12 Radiofrequency generates frictional heat that results in tissue injury and volume reduction. Risks of the procedure include pain, infection, swelling of the floor of the mouth, altered taste, tongue numbness and dysphagia.7 This procedure is currently performed only as an adjunct to other hypopharyngeal procedures as it rarely results in significant improvement when performed alone.12 In cases where the lingual tonsil itself is grossly enlarged, a reduction by coblation can be undertaken (Figure 3). Coblation combines radiofrequency energy and saline to create a plasma field that ablates tissue while minimising damage to surrounding areas.13
Tongue-base suture suspension
The tongue-base suture suspension procedure addresses obstruction in the tongue base and hypopharynx.12 In this procedure, a submucosal suture is anchored to the genial tubercle to prevent the tongue from collapsing and occluding the pharynx when muscle activity is reduced during sleep. The most common risks associated with this procedure are infection, haematoma and dysphagia.7 The tongue-base suture suspension procedure is generally considered to be minimally invasive, safe and effective. However, the success rate when performed alone for the management of OSA is variable.12 Thus, it is often performed in conjunction with UPPP to maximise outcomes.
Transoral robotic surgery for tongue-base reduction
While the nasal and oropharyngeal regions are easily accessible, traditional surgical approaches for OSA are limited by difficulties in accessing the tongue base and hypopharynx.14,15 Transoral robotic surgery (TORS) is a novel technique designed to overcome these limitations by improving visualisation and surgical access to the tongue-base region.15 Studies have found that tongue-base resection with TORS is feasible and well-tolerated, with less risk of neurovascular injury and intraoperative bleeding.14 Whether performed alone or as part of a multi-level strategy for OSA, TORS has been shown to result in a significant reduction in daytime somnolence and the apnoea-hypopnoea index (AHI).14,15
Posterior midline glossectomy
This procedure involves resection of the midline tongue base using carbon dioxide laser, radiofrequency ablation or robotic-assisted electrocautery.7,12 The risks of the procedure include bleeding, pain, increased secretions at the tongue base and altered taste. The success rate is variable, ranging from 25–83%.12
Genioglossus advancement
In this procedure, the geniotubercle (with the genioglossus insertion) is moved forward without moving the mandible.12 At the geniotubercle, the genioglossus muscle is attached to the lingual surface of the mandible and the hyoid complex, just above the larynx. The forward movement of these structures stabilises the tongue base and associated pharyngeal dilators. The advancement places tension on the tongue musculature and limits posterior displacement during sleep. The risks associated with the procedure include infection, haematoma, injury to the genioglossus muscle and paraesthesia of the lower teeth.12 The success rate for the procedure is variable, ranging from 23–77%. Thus, it is generally performed with UPPP in order to maximise outcomes.12
Maxillomandibular advancement
Maxillomandibular advancement is currently the most effective surgical procedure offered for OSA and has been shown to be as effective as CPAP.7,12,16 This procedure specifically addresses tongue-base and hypopharyngeal obstruction, and involves bilateral sagittal split of the ramus and Le Fort I osteotomies in order to advance the maxilla and mandible.7,12 It is an invasive procedure and the risks include bleeding, infection and facial numbness.12 Traditionally, maxillomandibular advancement is considered a second-stage procedure, following soft tissue surgery. However, it may be considered a primary procedure in patients with obvious craniofacial anomalies or multiple sites of upper airway obstruction.7
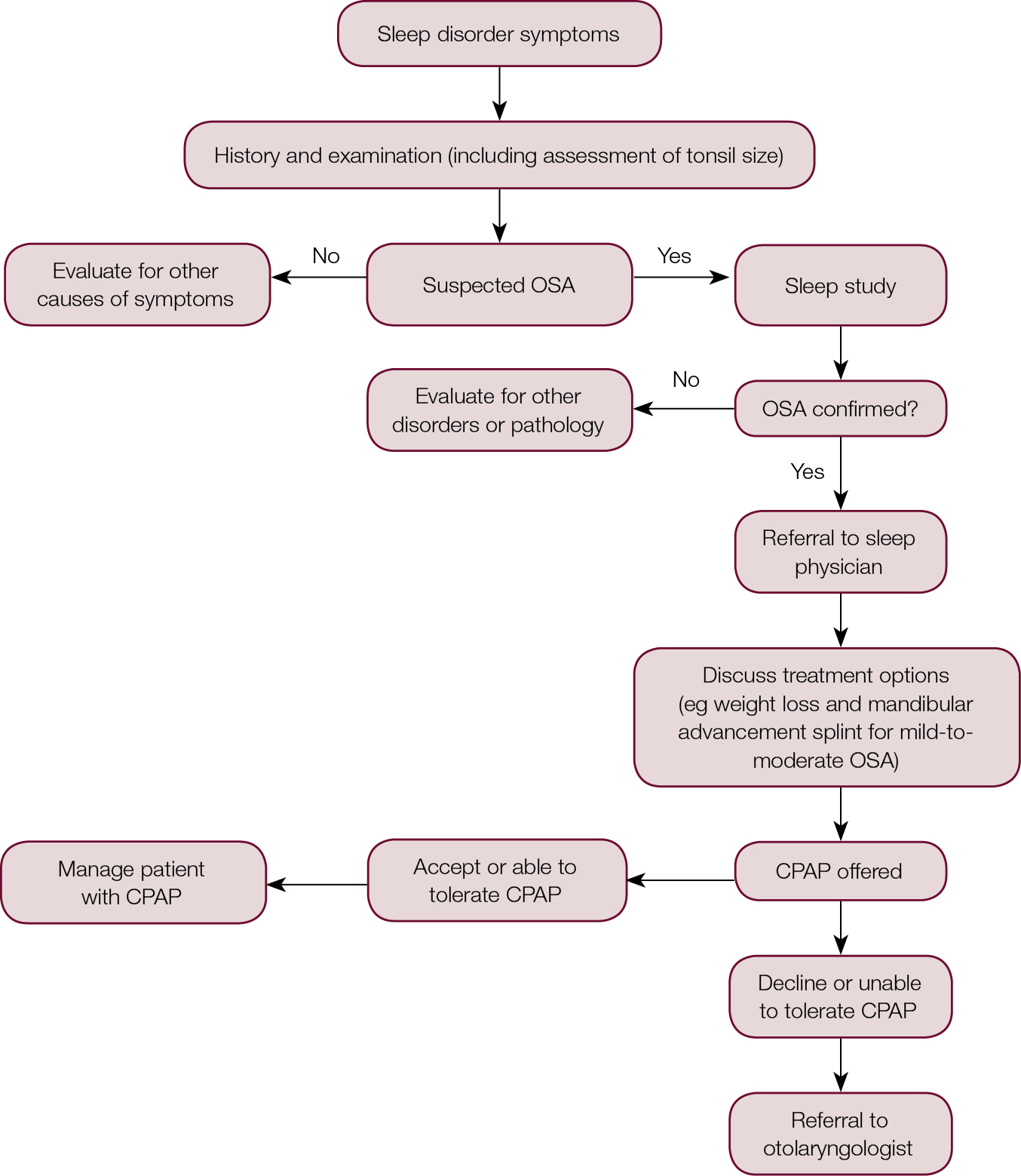 |
Figure 2. Referral algorithm for OSA
CPAP, continuous positive airway pressure; OSA, obstructive sleep apnoea |
Key messages for GPs
OSA requires long-term, multidisciplinary management. Surgery may be offered in carefully selected patients, depending on the severity of the disease, and the patient’s anatomy, risk factors and preferences. Patients who are unable to tolerate CPAP or those with craniofacial anomalies, can be referred to an otolaryngologist for further assessment. There are many surgical options available for OSA and the decision on the most appropriate intervention is based on anatomical considerations. Success rates for surgery are variable and the longevity of benefit is uncertain (Table 1). Following surgery, patients require ongoing follow-up with their sleep physician, surgeon and general practitioner. Objective measurement of success can be reported using the AHI.7 Patient-centred outcomes, such as somnolence, quality of life and improvement of comorbid conditions, are also useful.7
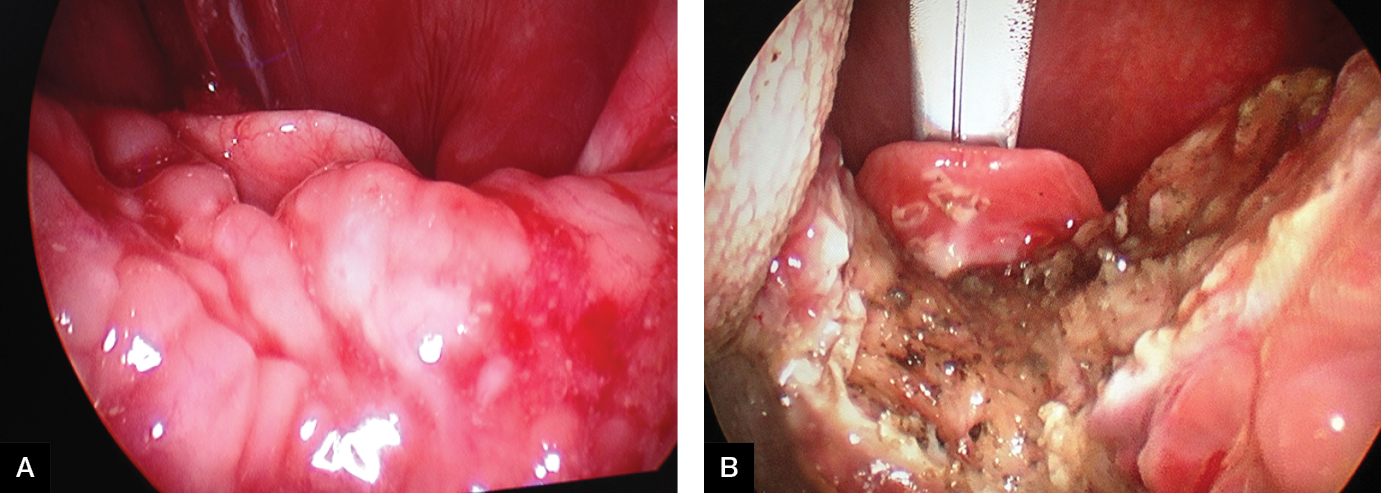 |
| Figure 3. Pre-operative (A) and intra-operative (B) views of lingual tonsil reduction by coblation |
Conclusion
CPAP is the primary treatment for OSA and is the recommended first-line treatment for patients with moderate-to-severe disease. However, for patients who are unable to tolerate CPAP or those who are unwilling to accept CPAP as a permanent form of management, surgery may be useful and warranted. These patients should be referred to an otolaryngologist for further assessment. In the past two decades, advances in sleep medicine and technology have enabled better recognition and surgical management of the disease in carefully selected patients.
Table 1. Summary of success rates for surgical treatment of OSA
|
Surgical treatment
|
Success rate
|
|---|
|
Procedures to improve nasal patency
and breathing
|
Opening an obstructed nose will not improve OSA; however, it may improve nasal breathing, CPAP tolerance and compliance
|
|
Procedures to improve retropalatal obstruction (UPPP and UPF)
|
Around 40% based on reduction of AHI and respiratory disturbance index
|
|
Procedures to improve tongue-base and hypopharyngeal obstruction
- Tongue-base, temperature-controlled radiofrequency alone
This procedure is currently performed only as an adjunct to other hypopharyngeal procedures as it rarely results in significant improvement when performed alone
- Tongue-base suture suspension alone
The success rate when performed alone is variable. The procedure is often performed in conjunction with the UPPP to maximise outcomes
- TORS for tongue-base reduction (alone or as part of multi-level strategy)
Around 54% based on reduction of AHI and resolution of daytime somnolence as measured by the Epworth Sleepiness Scale*
- Posterior midline glossectomy
Variable, 25–83%, based on reduction of AHI
Variable, 23–77%, based on reduction of AHI
- Maxillomandibular advancement
Around 75–100% based on reduction of AHI†
|
|
*Success rate based on a study conducted by Lin et al (n = 39)15
†Success rate based on a study conducted by Lee et al (n = 35) and a review by Li et al17,18
AHI, apnoea-hypopnoea index; CPAP, continuous positive airway pressure; TORS, transoral robotic surgery; UPF, uvulopalatal flap; UPPP, uvulopalatopharyngoplasty
|
Authors
Nga T Phan BPharm, MBBS, MPhil, Medical Officer, Department of Otolaryngology, Head and Neck Surgery, Princess Alexandra Hospital, Woolloongabba, Qld; Adjunct Lecturer, School of Pharmacy, The University of Queensland, Brisbane, Qld. nth_phan@yahoo.com.au
Benjamin Wallwork MBBS (Hons), FRACS, PhD, Staff Specialist, Department of Otolaryngology, Head and Neck Surgery, Princess Alexandra Hospital, Woolloongabba, Qld
Benedict Panizza MBBS, MBA, FRACS, FACS, Chairman and Director, Department of Otolaryngology, Head and Neck Surgery, Princess Alexandra Hospital, Woolloongabba, Qld; and Associate Professor, School of Medicine, The University of Queensland, Brisbane, Qld
Competing interests: None.
Provenance and peer review: Not commissioned, externally peer reviewed.
Acknowledgements
The authors thank Associate Professor Stuart MacKay for providing the images seen in Figure 3 of the article.