Initial assessment2
A thorough history should be taken, with specific attention to:
- Risk factors:
- family history of breast, colon, uterine or ovarian cancer, hereditary ovarian cancer syndrome (BRCA gene mutation/Lynch syndrome)
- Protective factors
- parity and breastfeeding (50% reduced risk)
- combined oral contraceptive pill
- Menopause status
- Symptoms, including those of endometriosis or malignancy (persistent abdominal distension, change in appetite, pelvic pain, urinary urgency).
A careful examination, including an abdominal and vaginal examination, should be undertaken and the presence of lymphadenopathy assessed.
Investigations
Imaging
Ultrasonography
Transvaginal and transabdominal ultrasound views should be obtained.1,3 This allows better differentiation and characterisation of the mass. The only definitive diagnosis of an ovarian mass is through histology; however, there are typical characteristics of certain structures seen on an ultrasound. Although ultrasonography is the best mode of imaging we have for assessment of ovarian pathology, its sensitivity and specificity for the diagnosis of ovarian cancer is only 86–91% and 68–81% respectively.3
The International Ovarian Tumor Analysis (IOTA) Group has developed a list of characteristics for benign and malignant masses.1,4,5 These rules are used in premenopausal women; however, similar characteristics are also used in the risk of malignancy index (RMI), which is discussed later. The IOTA Group rules are defined as benign or B-rules and malignant or M-rules (Table 1). Any patient with an M-rule should be referred to a gynaecologist.4,5 The presence of ascites has a positive predictive value of 95% for ovarian cancer.6
Table 1. IOTA Group ultrasound rules to classify masses as benign or malignant1,4,5
|
Benign (B-rules)
|
Malignant (M-rules)
|
|
Unilocular cysts
|
Irregular solid tumour
|
|
Presence of solid components where the largest solid component <0.7 cm
|
Ascites
|
|
Presence of acoustic shadowing
|
At least four papillary structures
|
|
Smooth multilocular tumour with largest diameter <10 cm
|
Irregular multilocular solid tumour with largest diameter >10 cm
|
|
No blood flow
|
Very good blood flow
|
Computed tomography and magnetic resonance imaging
Ovarian masses may be seen on computed tomography (CT) and magnetic resonance imaging (MRI). These are typically incidental findings. Assessment with ultrasonography is required to further assess the character of the mass. The use of CT or MRI in the assessment of an ovarian mass does not improve the sensitivity or specificity obtained through ultrasonography in the detection of ovarian cancer. MRI may be useful in assessment of large cysts that are difficult to assess on an ultrasound.1
Tumour markers
Serum Ca125
Serum Ca125 is a glycoprotein antigen and is the most widely used tumour marker in the assessment of ovarian masses. In premenopausal women, Ca125 should be measured only if the ultrasound appearance of a mass raises suspicion of malignancy. It is unreliable in differentiating malignant from benign, as Ca125 >35 U/ml has a sensitivity and specificity for ovarian cancer of <80% (potentially as low as 50–60%).3 It can also be raised in conditions such as endometriosis, fibroids, adenomyosis and pelvic infection. If Ca125 is elevated, consider repeating 4–6 weeks after the initial test.7 Rapidly rising levels are more likely to be associated with malignancy rather than levels that do not change. Discussion with a gynaecological oncologist is recommended in patients with a Ca125 >250 U/ml.1,3
In postmenopausal women, Ca125 should be measured routinely. Ca125 of >35 U/ml has a sensitivity of 69–97% and specificity 81–93% for the diagnosis of ovarian cancer.3 This result should then be used in conjunction with ultrasound findings and menopause status in RMI.
Human epididymis protein 4
Human epididymis protein 4 (HE4) is another tumour marker currently available for the assessment of ovarian cancer. It has a similar sensitivity as that of Ca125 in comparing ovarian cancer to healthy controls, but is not elevated in as many common benign gynaecological conditions. It is used in conjunction with Ca125 in the Risk of Malignancy Algorithm (ROMA).8,9 HE4 can be falsely elevated in patients with impaired renal function, and can also be elevated in endometrial, primary liver and non-small cell lung cancer.10 The American, UK and Australian guidelines do not address the usefulness of HE4 or ROMA in assessing risk for ovarian cancer. An HE4 level in isolation is difficult to interpret, and its usefulness in a clinical setting is being reviewed. HE4 is currently used in the USA for monitoring recurrence or progression of epithelial ovarian cancer.3,8 HE4 is not currently covered by Medicare and costs approximately $45 for the patient.11 It is not recommended as a screening test for ovarian cancer.
Other biochemical markers
Alpha-feta protein (AFP), human chorionic gonadotropin (hCG) and lactate dehydrogenase (LDH) are also recommended in women under 40 years who have a complex mass on ultrasound, as these can be elevated in germ cell tumours.1,9
Carcinoembryonic antigen (CEA) and cancer antigen 19.9 (Ca19.9) are two other tumour markers that are commonly ordered for the investigation of an ovarian mass; however, their application to clinical practice is unclear. The usefulness of these tests is not discussed in the UK and Australian guidelines. They are non-specific and can be elevated in benign and malignant non-gynaecological conditions. Ca19.9 may be useful in the assessment of mature cystic teratomas; however, its usefulness in differentiating mature cystic teratomas from ovarian cancer is unclear.12,13 CEA seems to be an independent prognostic factor for mucinous ovarian cancer.14 Further investigation is required.
Risk of malignancy index
The risk of malignancy index (RMI) is the most widely used risk assessment for ovarian malignancy. Developed in 1990, it uses serum Ca125, menopausal status and findings on ultrasound (RMI = ultrasound findings x menopause status x Ca125 U/ml). It is particularly useful in the assessment of postmenopausal women. Moderate risk is a RMI value between 25–200, and RMI >200 is considered high risk. An RMI >200 has a sensitivity of 87% and specificity of 97% for ovarian cancer and therefore requires urgent assessment by a gynaecological oncologist (Table 2).1,6,15
Table 2. Risk of malignancy index1,6,15
|
Risk of malignancy index (RMI) = ultrasound findings x menopause status x Ca125 (U/ml)
|
|
Findings
|
Points
|
|
Ultrasound findings include:
- multilocular cyst
- solid area
- metastases
- ascites
- bilateral lesions
|
0 points: no features (unilocular)
1 point: 1 feature
3 points: 2–5 features
|
|
Menopausal status
|
1 point – premenopausal
|
|
3 points – postmenopausal*
|
|
Ca125 (U/ml)
|
Actual level
|
|
Example
For a postmenopausal* woman with a left multilocular cyst and Ca125 of 40 U/ml:
RMI = 1 point for ultrasound x 3 points for postmenopausal x 40 U/ml
RMI = 120, therefore, gynaecology referral would be recommended
*Postmenopausal = no period for 1 year, or over 50 years in women who have had a hysterectomy
|
Management
There are three main forms of management – conservative, surveillance and surgical management. Deciding which is the appropriate management is based on assessment of symptoms, ultrasound findings, menopausal status, RMI (if applicable) and risk factors. An approach to management is outlined in Figure 1.
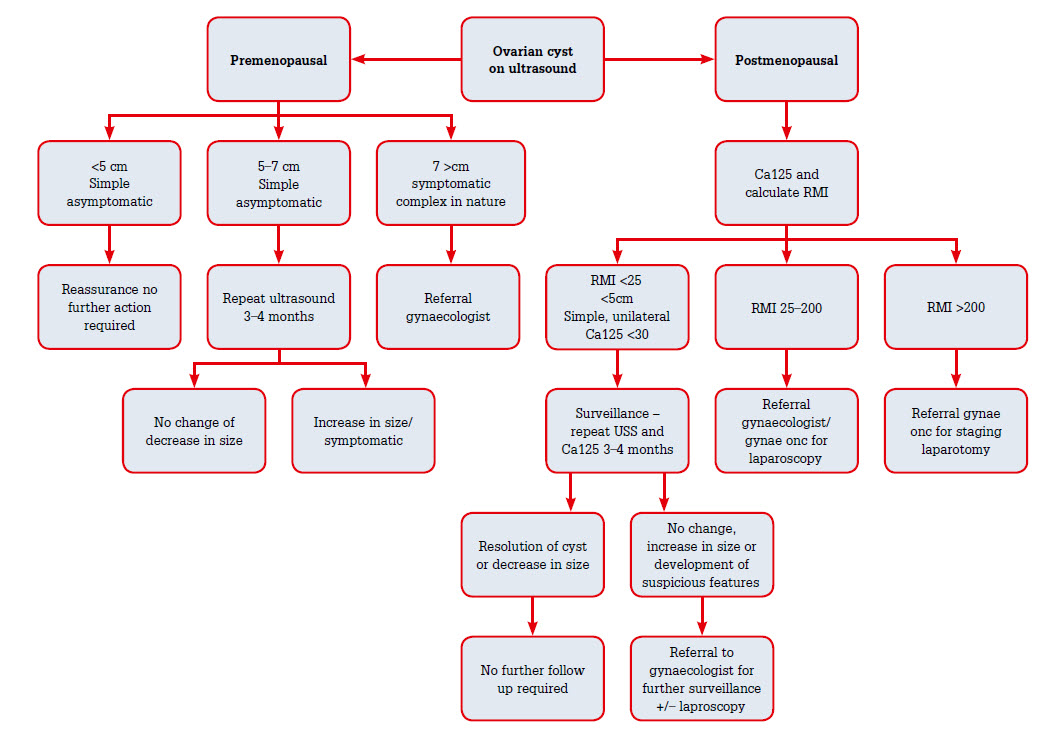 |
| Figure 1. Approach to the management of ovarian cysts1,2,15,21 |
Premenopausal womenAsymptomatic women with a simple ovarian cyst <5 cm on ultrasound do not require follow-up. These simple cysts will resolve within three menstrual cycles. For simple cysts of 5–7 cm, a repeat ultrasound should be obtained, and for cysts of >7 cm surgical intervention should be considered. If surgery is required, a laparoscopic cystectomy is the operation of choice, as aspiration can cause recurrence.16,17
Postmenopausal women
Simple unilateral, unilocular ovarian cysts of <5 cm and low risk of malignancy (normal Ca125) can be managed conservatively as the RMI would be zero and 50% of these will resolve spontaneously in 3 months. Cysts of 2–5 cm should be rescanned in 3–4 months.18–20 Women with a moderate-to-high risk RMI should be referred to a gynaecologist or gynaecological oncologist for consideration of surgical management. In addition, any woman who does not meet the criteria for conservative management should be offered surgical management. If malignancy is suspected, an oophorectomy is recommended rather than a cystectomy.1,15,20 This allows removal of the cyst intact and prevention of spillage into the peritoneal cavity. A bilateral oophorectomy may be offered for postmenopausal women because the contralateral ovary may also be affected; however, there are no studies that have assessed malignancy after unilateral versus bilateral oophorectomy.15,21
Use of the combined oral contraceptive pill
Commencing the combined oral contraceptive (COC) pill does not hasten resolution of functional ovarian cysts, but can be used to prevent formation of cysts.22,23
Complications
Cyst rupture
Patients typically present with lower abdominal pain, and an ultrasound that shows free fluid in the abdomen with a collapsed cyst. An uncomplicated cyst rupture can be managed as an outpatient procedure with oral analgesia. Symptoms usually resolve within 24–72 hours. In women with a complicated cyst rupture and a large amount of free fluid/haemoperitoneum, management in a hospital setting with fluid resuscitation is recommended. Laparoscopy may be indicated if a patient becomes unstable or has ongoing pain.23
Ovarian torsion
Ovarian torsion involves a partial or complete rotation of the ovary onto its supporting ligaments, cutting off its blood supply. Presenting symptoms usually include sudden onset lower abdominal pain, nausea and vomiting with a palpable adnexal mass. The primary risk factor for ovarian torsion is an ovarian mass >5 cm.24–26 Ovarian torsion is primarily a clinical diagnosis, but ultrasonography may be useful. One study showed a diagnostic accuracy of ultrasonography as 74.6%, with abnormal ovarian blood flow and presence of free fluid as the most diagnostic. Despite this, ultrasonography is not reliable in excluding an ovarian torsion.26 Suspected ovarian torsion requires urgent gynaecological review. Surgery usually involves laparoscopy with de-torsion and ovarian conservation, but an oophorectomy may be performed if the ovary is not viable. Torsion is most commonly associated with benign conditions.25,27
Pregnant women with ovarian cysts
Ovarian masses are usually an incidental finding. The majority of these masses are benign and can be managed expectantly, as at least 50% resolve spontaneously during pregnancy.28 The reported rate of complications with expectant management is <2%.29 If a cyst is identified early on a dating ultrasound, a repeat ultrasound at 12–14 weeks should be performed to check if it has resolved.29
Operative intervention is indicated if malignancy is suspected, if there is an acute complication (eg torsion) or if the size is likely to cause obstetric or other problems. The ideal time of operation is after the first trimester, as this decreases the miscarriage rate and teratogenicity. The risk of ovarian cancer in pregnant women who are noted to have a cyst on ultrasound is <1%.29,30
Screening for ovarian cancer
For the general population, there are currently no national or international guidelines for screening for ovarian cancer. No investigation to date has been shown to have adequate sensitivity and specificity as a suitable screening test. A precursor lesion has yet to be identified. In particular, screening with transvaginal ultrasound has a high false-positive rate because of its inability to differentiate between malignant and benign masses. Serum Ca125 can be affected in a number of benign conditions and is elevated in <50% of women with stage 1 ovarian cancer.6
Women with a very strong family history of breast and ovarian cancer should be referred for genetic counselling. Women who are carriers of the BRCA1 mutation have a lifetime risk of ovarian cancer as high as 60%, and BRCA2 as high as 40%.6
Key points
- Ultrasonography (transabdominal and transvaginal) is the main form of imaging in the assessment of ovarian masses.
- Ca125 can be unreliable in premenopausal women as it can be elevated in a number of benign conditions; however, it is useful in the assessment of postmenopausal women.
- RMI is used to assess risk of ovarian cancer and is based on menopause status, ultrasound findings and Ca125 levels.
- Unilateral, simple ovarian cysts that are <5 cm in premenopausal women are likely be functional cysts and no follow-up is required.
- Ovarian torsion is a clinical diagnosis and requires urgent gynaecological review.
- There is no routine screening for ovarian cancer for the general population.
- If concerned or unsure of management, seeking gynaecological advice is recommended.
Competing interests: None.
Provenance and peer review. Not commissioned, externally peer reviewed.