During this period, the sex-specific proportion of CVA managed (% encounters) was significantly lower in women (0.16%; 95% CI: 0.15–0.17) than in men (0.24%; 0.23–0.26). The age-specific proportions of CVA managed were 0.68% (0.63–0.73) for patients aged ≥75 years and 0.35% (0.32–0.38) for those aged 65–74 years, compared with 0.19% (0.18–0.20) for the whole sample. Comorbidities most often managed with CVA were hypertension (17.6% of other problems), DM (7.4%), LD (4.7%), depression (4.0%) and AF (2.7%).
On average, for every 100 CVA problems managed at an encounter, 60.2 medications (56.2 prescribed, including 9.0 vitamin K antagonists), 18.2 clinical treatments (including counselling), 5.1 medical specialist referrals (including 2.2 to neurologists) and 4.7 allied health referrals were recorded. Figure 1 shows that the percentage of encounters at which a CVA was recorded was constant: 0.2% for each year throughout the period 2000–2013, despite the significant increase in the proportion of older patients in the population over the past decade.3 Over the same period there were significant increases in the proportion of encounters where AF or DM were managed, whereas the proportion with LD or hypertension was variable. These findings are consistent with the effective prevention and management of CVA in Australian general practice.
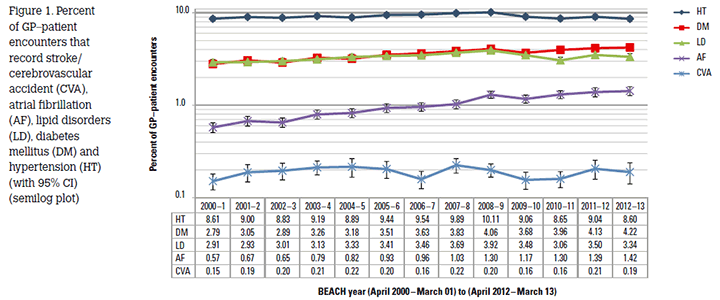
Figure 1. Percent of GP–patient encounters that record stroke/ cerebrovascular accident (CVA), atrial fibrillation (AF), lipid disorders (LD), diabetes mellitus (DM) and hypertension (HT) (with 95% CI) (semilog plot)
Acknowledgements
The authors thank the GP participants in the BEACH program and all members of the BEACH team. Funding contributors to BEACH from April 2000 to March 2013: Australian Government Department of Health and Ageing; AstraZeneca Pty Ltd (Australia); CSL Biotherapies Pty Ltd; Merck, Sharp and Dohme (Australia) Pty Ltd; National Prescribing Service; Novartis Pharmaceuticals Australia Pty Ltd; Pfizer Australia Pty Ltd; Abbott Australasia Pty Ltd; Janssen-Cilag Pty Ltd; Sanofi-Aventis Australia Pty Ltd; GlaxoSmithKline Australia Pty Ltd; Bayer Australia Ltd; Wyeth Australia Pty Ltd; and Roche Products Pty Ltd. BEACH is approved by the Human Research Ethics Committee of the University of Sydney.