Defining an optimal level of vitamin D drives clinical treatment decision making: should treatment begin at 25(OH)D levels <30, <50 or <75 nmol/L, and what is the target level? Advice, including that from reputable sources, is highly variable. Here we give a brief review of the vitamin D metabolic pathway and examine some challenges in interpreting the large body of available literature.
Vitamin D metabolic pathway
For most Australians, the main source of vitamin D is cutaneous synthesis after sun exposure of the skin.5 Levels of ultraviolet radiation vary throughout the year (higher in summer than winter) and this is reflected in 25(OH)D levels.6 Diet and supplements also provide vitamin D, but the vitamin D content of most foods in Australia is quite low.
Vitamin D from the skin or diet is carried to the liver where it is converted to the inactive metabolite, 25(OH)D. This is converted into the active form, 1,25 dihydroxyvitamin D (1,25(OH)D), primarily in the kidney. Other cells, including those of the immune system7 and the skin,8 also have the required enzymes so 1,25(OH)D can be generated to have local (cellular level) effects.
The primary role of 1,25(OH)D in the body is the maintenance of calcium homeostasis. Low serum calcium concentrations are sensed in the parathyroid gland by a calcium-sensing receptor and this causes increased secretion of parathyroid hormone (PTH). This stimulates renal production of 1,25(OH)D, which increases calcium uptake in the intestine and, in a negative-feedback loop, suppresses PTH secretion.
Measuring vitamin D status
Most commercial pathology laboratories use an automated immunoassay, such as Diasorin Liaison, to measure the 25(OH)D concentration. More specialised laboratories may use a liquid chromatography tandem mass spectrometry (LC-MS/MS) assay, which provides superior performance but is less amenable to high throughput.9 Although most laboratories participate in an external quality assurance scheme this does not guarantee quality. Figure 1 shows the results from the Vitamin D External Quality Assurance Scheme (DEQAS) for a single sample tested in 1090 different laboratories around the world. The solid line is at 50 nmol/L; the US Institute of Medicine (IOM) recommends maintaining 25(OH)D concentration at this level or higher. After excluding the top and bottom 5% of values, the mean of the remaining results (the all laboratory trimmed mean, ALTM) for this sample was 47 nmol/L. However, results from individual laboratories ranged from <20 nmol/L to >100 nmol/L. Here there is no ’true‘ value – quality assurance is measured against the ALTM.
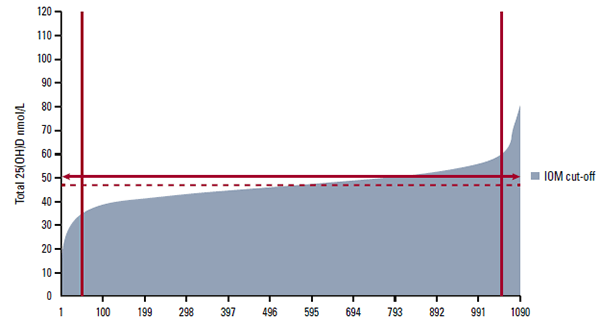
Figure 1. DEQAS sample 417 (July 2012): results for a single sample tested in multiple different laboratories (personal communication, G Carter, DEQAS)
IOM = Institute of Medicine of the USA
Figure 2 shows the 25(OH)D results for 25 samples where aliquots of the same samples were measured in different assays. Results from the University of Wisconsin Liquid Chromatography (UWLC) assay are considered to provide the true value.10 For low 25(OH)D levels, there is fairly close agreement between the different assays (eg. samples 10 and 23). However, for several samples within the range where there is a question about treatment or not, there is wide variation. For example, the true value for sample 9 is about 80 nmol/L, but assays report values of about 60 nmol/L and 100 nmol/L. The true value for sample 14 is about 70 nmol/L, but one Liaison assay reports a value of about 45 nmol/L, whereas an LC-MS/MS reports about 80 nmol/L. We have reported similar variability across three laboratories and two assays – Liaison and LC-MS/MS – in Australia.11 Two Liaison assays had a mean difference of about 11 nmol/L; Liaison differed from LC-MS/MS by about 26 nmol/L.11
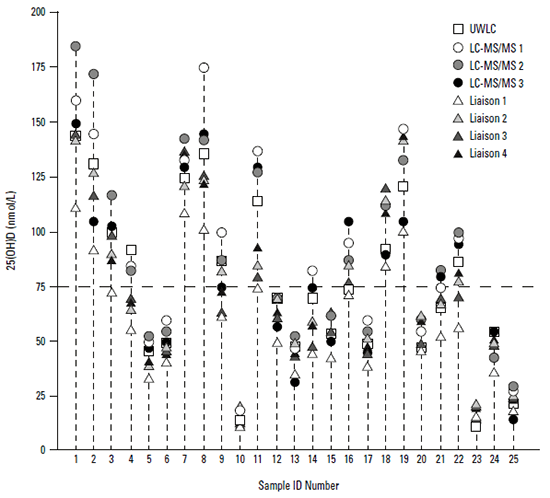
Figure 2. Results for 25(OH)D tested by multiple different assays
Adapted with permission from Elsevier from Binkley N, Krueger DC, Morgan S, Wiebe. D Clin Chim Acta 2010;411.10
What does this mean for the clinician? The types of 25(OH)D assays commonly used in Australia tend to read low when compared with reference measurement procedures (true values).10 For any individual result this means that a level of 40 nmol/L could really be 60 nmol/L or higher. Similarly, a level of 60 nmol/L – which will be treated with vitamin D supplementation by some practitioners – could be more than 80–90 nmol/L. A consistently low reading against the true value is not necessarily a problem, provided the evidence around optimal levels arises from the same low-reading assays. Unfortunately this is not the case. Variability across different assays in generating the evidence and interpreting an individual result in light of this evidence leads to considerable uncertainty.
A single 25(OH)D measurement provides a snapshot of current vitamin D status. This typically varies slowly during the year, paralleling changes in the amount of sun that our skin receives.6 There are no data to indicate whether seasonal variation in 25(OH)D is good or bad for health or has no effect. However, vitamin D deficiency (<30 nmol/L) in summer is of concern, as this implies a much lower level is likely in winter, and should certainly prompt treatment and checking of the winter level.
What do the epidemiological studies, including meta-analyses, show?
There are a number of factors to keep in mind when evaluating the evidence from epidemiological studies and meta-analyses of vitamin D status in relation to various diseases. First, while some studies examine risk of disease in categories according to pre-selected absolute values (eg. <50 nmol/L, compared with ≥ 50nmol/L) others use a data-driven approach, comparing risks across quantiles of the distribution. Decisions about the cut-off point for optimum health rely on the former, but the latter are most often used. Second, there needs to be careful consideration of how the quantiles are defined. In most studies showing an association between vitamin D status and a health outcome, risk is related to vitamin D deficiency (i.e. very low 25(OH)D levels). If the cut-off point for the lowest quantile is not set at a sufficiently low level, then this category will be heterogeneous. That is, the category will include some people at high risk and others at low or negligible risk, diluting risk estimates. For example, in one study the cut-off for the low (reference) category was 57 nmol/L and the risk estimates for colon cancer were non-significant.12 Yet there is considerable evidence linking vitamin D deficiency to increased colorectal cancer risk. The upper quantile may also be heterogeneous for risk. In 2007, Freedman and colleagues13 reported that 25(OH)D levels of 80 nmol/L or more were associated with a 72% lower risk of colorectal cancer mortality, compared with levels of <50 nmol/L. Yet in their follow-up analysis with a larger sample size, there was an increased overall cancer mortality in men with 25(OH)D levels >100 nmol/L, and the protective effect of higher levels for colorectal cancer risk was no longer significant. An increasing risk of all-cause mortality in association with 25(OH)D levels >100 nmol/L in the same population has recently been published.14
This consideration of the effect of different definitions of quantiles is also important when interpreting meta-analyses. Here, data from smaller studies are combined to provide a larger overall sample size and thus more precise estimates. For vitamin D meta-analyses, the relative risk estimates for the highest 25(OH)D quantile, compared with the lowest are generally combined no matter how these quantiles are defined. For example, in a recent meta-analysis of 25(OH)D level in relation to colorectal cancer,15 cut-off points to define the highest quantile varied from >48 to >105 nmol/L in the studies. Similarly cut-offs for the lowest quantile varied from ≤25 to ≤57 nmol/L. Is it valid to combine risk estimates from such disparate groupings? And, how should the results be interpreted? The combined analysis shows that higher 25(OH)D levels are protective, compared with ‘lower’ levels but the analysis provides no information on ’optimal‘ levels.
What is the evidence for cut-off points of 50 nmol/L, 75 nmol/L or higher levels?
Keeping in mind the longstanding measurement issues, this question is almost impossible to answer. In most studies, the at-risk group is that with the lowest 25(OH)D levels. The highest quantile commonly has reduced risk of disease, compared with the lowest quantile. However, the difference in risk between the highest quantile and the middle quantiles is often not significant. That is, if the reference group was changed to be a middle quantile (eg. 50–75 nmol/L), there would be no significantly increased benefit for levels >75 nmol/L.16
In 2010, the IOM concluded that bone health is the only condition for which a causal association with vitamin D has been established, and that a level of 50 nmol/L is sufficient to optimise bone health in the majority of the population.17 Two recent publications have extended the range of diseases for which it was considered there was reasonable evidence of a causal association. Yet these publications also conclude that concentrations of around 50 nmol/L are sufficient to minimise the risk of these conditions and only marginal gains are achieved above this level.16,18
Many studies have sought to define optimal vitamin D status as the level of 25(OH)D at which PTH levels are at a minimum plateau. The results, however, have been highly variable. Some studies have found no plateau or no relationship between 25(OH)D and PTH levels. In other studies, the plateau occurs at 25(OH)D levels ranging from <30 nmol/L to >100 nmol/L17. Much of this variability may be due to differences in the assays used, the population under consideration and even the statistical methodology. Nevertheless, at this stage there is little consistent evidence to indicate an optimal 25(OH)D level on the basis of a plateau in PTH concentration.
Are there risks of (moderately) higher 25(OH)D levels?
The first reports of a U-shaped or reverse J-shaped relationship were for prostate cancer,19 where the point of inflexion was at a 25(OH)D level of approximately 50 nmol/L. An Australian study showed an increased risk of prostate cancer in association with higher levels of sun exposure, which is consistent with an upswing in risk at higher 25(OH)D levels.20 There are similar findings of increased risk at higher 25(OH)D levels for other outcomes, including tuberculosis (turning point = 76–140 nmol/L, three categories only),21 premature ageing (studies in mice),22 schizophrenia in relation to neonatal vitamin D status (turning point = 47 nmol/L),23 small-for-gestational-age births among white women (turning point = 60–70 nmol/L),24 cardiovascular disease (turning point = 50–90 nmol/L),25 total cancer mortality26 and all-cause mortality (turning point = 80–100 nmol/L).14,27
Problems with interpretation of observational studies
An additional challenge in evaluating the evidence is the need to rely mostly on observational data. Serum 25(OH)D levels are affected by body mass index, physical activity and diet, all of which may influence disease outcomes. While most studies do control for these confounding factors, complete control of confounding is notoriously difficult.28 Where prospective data are not available, it may be impossible to determine whether the low 25(OH)D levels preceded the disease or were a consequence of it. In a recent meta-analysis, the strength of the evidence for an association between 25(OH)D levels and breast cancer decreased as the quality of the study design increased.29 The literature is replete with examples of exposures shown to be associated with disease in observational studies but where supplementation trials have generated null or even the opposite findings (antioxidants, beta-carotene, hormone replacement therapy, homocysteine). With the exception of bone health and falls, data from randomised controlled trials provide only sparse and inconsistent support for a causal association between vitamin D and disease outcomes. We need to be cautious about vitamin D supplementation (other than for frank deficiency) given the weak evidence currently available.
If we can get all of the protective effect at 25(OH)D levels of 40–50 nmol/L, then why go higher?
Studies that focus only on the decreased risks of the highest category of 25(OH)D concentration versus the lowest do not provide evidence to support specific cut-off levels. There are well-demonstrated health risks of very low 25(OH)D levels, less than approximately 30 nmol/L, and treatment through supplementation and/or increased sun exposure is clearly required. Most of the decrease in risk is achieved by levels >40–50 nmol/L; only small further gains are achieved at higher levels.16 Yet we know that there are measurement issues: the most common assays typically return lower concentrations than the gold standard. This indicates a need to err on accepting a ’low‘ result, assuming that the true 25(OH)D concentration is likely to be higher. This is preferable to supplementation to reach a high 25(OH)D level and possibly the upswing of the reverse J-shape (ie. increased disease risk). Many vitamin D researchers advocate maintaining 25(OH)D levels at >75 nmol/L; others advocate even higher upper limits of ’normal‘ (eg. 80 ng/ml or 200 nmol/L).30 We contend that this is not well supported by the current evidence.
Concluding remarks
Vitamin D testing is the fastest growing Medicare item3 and testing has increased exponentially over the past 15 years. Yet we know that assays return highly variable results, there is dispute about optimal levels, and there is recent evidence of risks with high levels. Managing patients requires assessment of individual risks. For patients at high risk of poor bone health or colorectal cancer, accepting the potential risks of higher 25(OH)D levels may be warranted to gain the possible benefits. This is a judgement call that physicians must make in consultation with an informed patient. Apart from these high risk groups, there is no strong evidence to support health benefits of vitamin D supplementation for those with apparent mild insufficiency (40–50 nmol/L) on routine testing.
Competing interests: Robyn Lucas has received a speaker fee from the Danish European Movement, the European Policy Centre and Leo Pharma. She has also received payment for conference travel, accommodation and meeting expenses from the European Skin Cancer Foundation, ECTRIMS, MS Research Australia, the United Kingdom MS Society and the 1st World Conference on Skin Cancer Prevention and the World Conference on Science and Technology Education.
Provenance and peer review: Not commissioned; externally peer reviewed.