Discussion
The prevalence of heterophile antibodies is 0.17–40% in the general population.1 In eight automated tumour marker immunoassays, the prevalence of heterophile antibodies was 0.2–3.7%.2 Sources proposed for the induction of heterophile antibodies include exposure to mice and mouse products, immunisation, blood transfusion, autoimmune diseases, dialysis and maternal transfer. Two-site immunometric or sandwich assays are particularly susceptible to analytical error in the setting of heterophile antibodies. These assays use at least two antibodies directed against different epitopes of an antigen: one bound to a solid-phase, the other in solution and tagged with a signal moiety. The antigen in the sample bridges the two antibodies so that the amount of labelled antibody that binds to the solid-phase is proportional to the antigen concentration in the sample (Figure 1). Heterophile antibodies can also bridge the two antibodies independently of antigen, resulting in an increase in labelled antibody concentration (Figure 2).
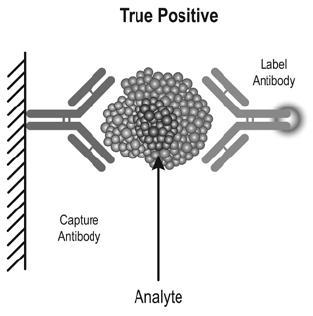
Figure 1. 2-site immunometric (sandwich) assays
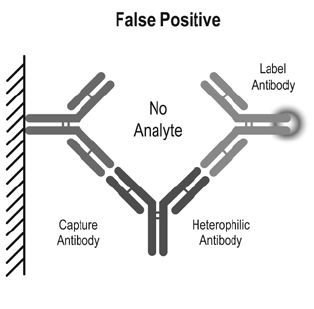
Figure 2. Interference by heterophile antibodies
Heterophile antibodies may affect a wide array of laboratory tests, resulting in false elevation of tumour markers, endocrine tests, cardiac injury markers and drug levels (Table 1). Heterophile antibodies may also cause false depression of serum cortisol levels, resulting in incorrect diagnosis of hypothalamic-pituitary-adrenal axis insufficiency and inappropriate and potentially harmful replacement with glucocorticoids. False lowering of thyroglobulin levels may also occur. Where suspected, the presence of heterophile antibodies interfering with the accuracy of investigations can be addressed through analysis by an alternative analytical platform, non-linearity of results on serial dilution or the use of heterophile-blocking reagents or immunoglobulin-blocking reagents.
Table 1. Immunoassays potentially affected by heterophile antibodies
Cardiac markers
- Troponin, creatine kinase-MB isoenzyme, B-type natriuretic peptide (BNP)
Tumour markers
- Prostate-specific antigen (PSA)
- Carcinoembryonic antigen (CEA), cancer antigen 19-9 (CA 19-9), cancer antigen 125 (CA 125)
- Alpha-fetoprotein (AFP), beta human chorionic gonadotropin (ßhCG)
- Calcitonin
- Thyroglobulin
- Chromogranin A
Endocrine tests
- Follicle-stimulating hormone (FSH), luteinizing hormone (LH), prolactin, oestradiol, progesterone, testosterone
- Thyroid-stimulating hormone (TSH), free thyroxine (FT4), Free triiodothyronine (FT3)
- Adrenocorticotropic hormone ( ACTH), cortisol
- Parathyroid hormone (PTH)
- Inhibin A
Drug levels
- Digoxin
- Cyclosporine, tacrolimus
Other
- Tryptase
- Myoglobin
- Antiphospholipid antibodies
- Erythropoietin
- Human immunodeficiency virus (HIV)
|
A study of patients investigated for cTnI, because myocardial infarction was suspected, found heterophile antibodies caused false positives in 5.5% of those with raised cTnI and 14% of those with raised cTnI and normal creatine kinase.3 The universal international definition of myocardial infarction requires the detection of a rise or fall of cardiac biomarker values together with symptoms of ischaemia, new changes on electrocardiography, evidence on imaging of new loss of viable myocardium or identification of an intracoronary thrombus.4 Acute elevation of cTnI may also occur with sepsis, pulmonary embolism, myopericarditis, stroke, chemotherapy, strenuous exercise and acute heart failure.5 In addition to heterophile antibodies, chronic persistent elevation of cTnI may occur with renal dysfunction, chronic obstructive pulmonary disease, pulmonary hypertension, left ventricular hypertrophy and obstructive sleep apnoea.
Heterophile antibody interference with tumour markers is particularly important as it may lead to a false diagnosis of malignancy (either primary or residual post-treatment) and unnecessary surgery and/or chemotherapy. Rotsmensch and Cole described 12 women incorrectly diagnosed with postgestational choriocarcinoma on the basis of persistently positive human chorionic gonadotropin (hCG) levels. Most of the women underwent extirpative surgery or chemotherapy without diminution in hCG titre and were found to have false-positive hCG values due to heterophile antibodies.6 The authors concluded that protocols for the diagnosis and treatment for choriocarcinoma should include a compulsory test for hCG in urine as the immunoglobulin-hCG complex is not cleared by the kidney. It has been suggested in clinical scenarios where there is a low rate of true positives and a higher likelihood of interference, adding a heterophilic antibody blocker to all positive tests may be a more cost-effective than retesting positive samples.7 An example of this is measuring serum calcitonin levels in patients with thyroid nodules. A study of 378 subjects found 5 patients (1.3%) with falsely elevated calcitonin levels as a result of heterophile antibodies, whereas none of the patients had medullary thyroid cancer.8 The use of tumour markers in screening should be discouraged not only because of interference by heterophile antibodies, but because of the significant rate of false positive and false negative results with these tests due to tumour biology.
Conclusion
Heterophile antibodies are a cause of falsely elevated or depressed laboratory values and should be considered and sought whenever there is incongruity between a clinical scenario and the results of pathology investigations.
Competing interests: None.
Provenance and peer review: Not commissioned; externally peer reviewed.