This review will focus on ankylosing spondylitis (AS) and the recently defined disorder of non-radiographic axial SpA.1 These conditions occur in one in 200 individuals but most general practitioners have never identified a new case of AS, suggesting the need for a higher index of suspicion in primary care.
Clinical features
Back pain
Approximately 5% of chronic lower back pain is attributable to SpA. The presence of inflammatory back pain (IBP), the archetypal feature of AS, increases the likelihood of SpA to approximately 14%.2 Table 1 shows the differentiating features of inflammatory and mechanical back pain. Two very specific features of IBP are alternating buttock pain and awakening only in the second half of the night with spinal pain or stiffness. Clinicians can only assess the activity of IBP by subjective symptoms: fatigue, spinal pain, and severity and duration of morning stiffness. Identification of IBP should prompt more detailed evaluation and consideration of a multimodality treatment approach.2
Table 1. Characteristics of inflammatory and mechanical back pain
| | Inflammatory | Mechanical |
|---|
| Age at symptom onset |
<40 years old |
Any age |
| Onset |
Insidious, persists for >3 months |
Variable |
| Activity |
Improves with exercise |
Improves with rest |
| Morning stiffness |
Moderate, persists for >45 minutes |
Mild, short-lived |
| Inflammatory markers |
Elevated in 50–70% |
Normal |
Reduced spinal mobility
The most common measures of spinal mobility are listed below.
- Modified Schober’s test (Figure 1): serial measures are valuable in assessing progression of spinal restriction; may be abnormal with disc disease and degenerative lumbar disease.
- Lumbar side flexion: the best measure of overall spinal restriction and disease activity.
- Occiput-to-wall distance: should be zero in normal people.
- Chest expansion (at the fourth intercostal space): abnormal (<5 cm) in only a minority of patients during the first few years of disease
It is important to note that in early disease, spinal mobility may be normal.
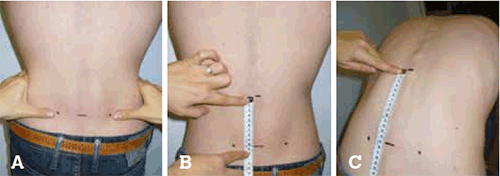
Figure 1. Spinal Mobility – Modified Schober
A) Patient standing erect. Mark an imaginary line connecting both posterior superior iliac spines (close to the dimples of Venus).
B) A mark is placed 10 cm above.
C) The patient bends forward maximally, measure the difference between the two marks. Report the increase (in cm to the nearest 0.1 cm). The best of two tries is recorded.
Reproduced with permission from ASAS handbook, Ann Rheum Dis 2009; 68 (Suppl II)
Extra-axial features
Peripheral arthritis occurs in all variants of SpA. Up to 50% of AS patients develop asymmetric oligoarthritis (≤4 joints), often targeting the lower limb joints. Enthesitis or inflammation at insertions of tendons, ligaments and joint capsules into bone can present as Achilles tendinitis, plantar fasciitis and intercostal enthesitis, which causes chest wall pain. Dactylitis (‘sausage digit’) is diffuse swelling of a finger or toe caused by tenosynovitis of the digital flexor tendon, and is more common in psoriatic and reactive arthritis.
Extra-articular features
Uniocular anterior uveitis occurs in about 40% of patients and typically presents as acute painful red eye, with blurred vision and photophobia. In suspected first episodes of anterior uveitis, ophthalmology review for slit-lamp examination is mandatory. First-line treatment is a combination of corticosteroid and mydriatic eye drops. About 60% of AS patients have mucosal inflammation on colonoscopy, while symptomatic inflammatory bowel disease occurs in 10% of patients. Osteopenia and osteoporosis commonly occur, particularly in long-standing persistently active disease, and the risk of vertebral fracture is increased approximately sevenfold. Importantly, bone densitometry in the spine may be spuriously elevated by the syndesmophytes of AS. Patients with AS are at risk of developing apical pulmonary fibrosis (up to 15%) and aortic valve incompetence (up to 10%). These features usually develop late in the disease and are often asymptomatic.
Juvenile SpA
It is important to be aware of juvenile SpA and 50% of cases will evolve into typical AS. Asymmetric oligoarthritis, often associated with severe midfoot pain (tarsitis), is a common presentation. HLA-B27, tarsitis and hip involvement are predictors of the persistence of disease as AS into adulthood.
Investigations
Imaging
X-ray
A single anteroposterior pelvis X-ray (rather than dedicated sacroiliac joint views) should be adequate to assess the sacroiliac joints. AS is defined by IBP and at least bilateral grade 2 or unilateral grade 3 changes (Figure 2).4
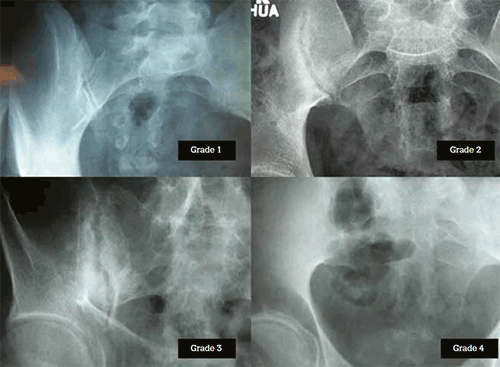
Figure 2. Radiographic grading of sacroiliac joints. Grade 1: Suspicious changes, not definitive. Grade 2: Small localised areas of erosions or sclerosis; normal joint space width. Grade 3: Definite changes of erosions, sclerosis, narrowing or partial fusion. Grade 4: Complete fusion
The radiographic findings represent reparative changes in response to earlier acute inflammation. As such, the appearance of radiographic abnormalities is typically delayed. In the spine, there is progression from reactive sclerosis (‘shiny corner’ or Romanus lesion), squaring and erosions at the edge of the vertebral bodies to syndesmophyte formation and bony bridging (Figure 3). The classic ‘bamboo spine’ is characteristic of advanced AS.
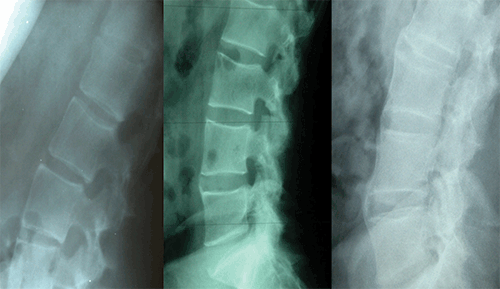
Figure 3. Lumbar spine changes in AS. A) Shiny corners and erosions. B) Early syndesmophytes C) Spinal fusion or ankylosis
Magnetic resonance imaging (MRI)
MRI is often considered when X-rays are normal but there is a clinical suspicion of SpA. MRI may be particularly helpful in early disease, although it can be normal even in active SpA and the chronic changes of sacroiliitis are better seen on plain X-rays. Fat-suppressed short T1 inversion recovery (STIR) sequences demonstrate marrow oedema at the sacroiliac joints and/or vertebral corners. Marrow oedema is associated with later development of bony sclerosis or fusion, either of the sacroiliac joints or spine.5
Ultrasonography
Ultrasonography can be useful in detecting enthesitis, such as Achilles tendonitis. Its role in the detection of sacroiliitis remains to be determined as this modality is largely operator-dependent. There are, however, some suggestions that colour Doppler ultrasound may be used to diagnose early sacroiliitis and to monitor response to therapy.
Laboratory tests
Acute phase reactants such as erythrocyte sedimentation rate (ESR) and C-reactive protein are useful markers of inflammation but are elevated in only 50–70% of AS patients. They are more likely to be elevated in patients with concurrent peripheral arthritis and inflammatory bowel disease.
The role of HLA-B27 and other genes
The association of HLA-B27 with AS is well established and occurs in 85–90% of patients. HLA-B27 is seen in 5–15% of the general population, with some variability associated with ethnic background, but only 5% of HLA-B27 positive people develop AS. As such, HLA-B27 in the patient with clinical features of SpA can support the diagnosis but has no role as a general screening test for spinal pain.
Delay in diagnosis
The delay between onset of symptoms and physician diagnosis of AS is 5–7 years on average.6 Chronic back pain is common and recognition of early disease requires clinical experience and a high index of suspicion. Further, inflammatory markers are not invariably elevated and radiographic changes are often a late finding. The delay in diagnosis is longer among women, who comprise one third of AS patients, perhaps related to a misconception that AS is rare among women.
Features of SpA are important to identify early at presentation and include alternating buttock pain, asymmetric peripheral arthritis, heel pain, uveitis, positive family history and good response to non-steroidal anti-inflammatory drugs (NSAIDs). The presence of one or two of these features increases the likelihood of SpA to 35–70%, and three or more features increases the likelihood to 80–95%.3
Non-radiographic axial SpA
Even in the modern era of MRI, X-ray changes at the sacroiliac joints must be present in order to establish a diagnosis of AS. The delay from symptom onset to X-ray changes, however, can be 10 years or more. Many but not all patients with early presentations have active sacroiliitis on MRI scan. If MRI abnormalities are identified, a diagnosis of non-radiographic axial SpA can be established if at least one clinical feature of SpA is present. If the MRI is normal, then HLA-B27 and two or more SpA features are required. The Assessment of SpondyloArthritis International Society (ASAS) has developed and validated these classification criteria (Figure 4).7
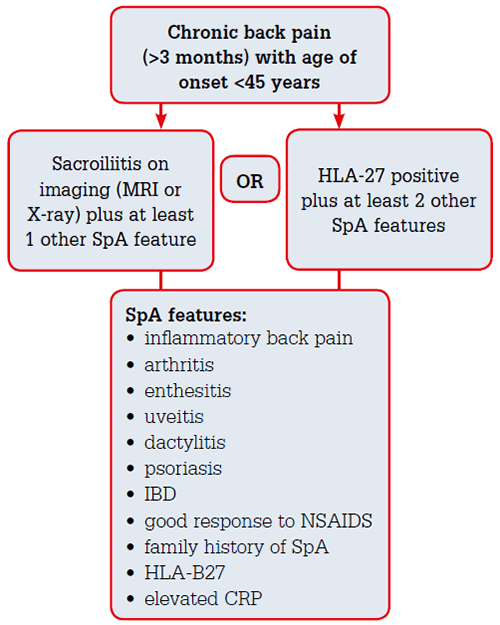
Figure 4. ASAS classification criteria for axial spondyloarthritis.7 These classification criteria have a sensitivity of 83% and specificity of 84% for detecting all axial SpA. (IBD = inflammatory bowel disease, CRP = C-reactive protein, NSAID = non-steroidal antiinflammatory drug)
Although patients with non-radiographic axial SpA patients have fewer intense inflammatory changes on MRI and have lower levels of inflammatory markers, they have similar levels of disease activity and pain as those with established AS. Therefore patients with non-radiographic axial SpA warrant early evaluation and treatment.
Predictors of outcome
Several factors associated with poor prognosis are identified in AS, most importantly juvenile-onset and hip arthritis. Other factors include persistent active disease, cigarette smoking, inadequate response to NSAIDs and baseline spinal radiographic changes.
Management
Symptom management
Education and exercise
As with any chronic condition, patient education and support is vital in the management of AS. Patient disease and medication information, as well as access to support groups, can be found on the Arthritis Australia website (www.arthritisaustralia.com.au). A tailored exercise and stretching program is recommended for AS patients and there are several useful online resources (eg. www.nass.co.uk/exercise) and mobile apps (eg. iAnkSpond).
NSAIDs
NSAIDs are first-line therapy for symptomatic AS patients. Recent studies suggest that regular NSAID use in AS slows radiographic progression more than on-demand use.8 An individualised assessment of risk of long-term NSAID use should be made in consultation with the rheumatologist before long-term daily NSAID use is recommended.
Tumour necrosis factor (TNF) inhibitors
TNF inhibitor therapy has strikingly improved the quality of life for the more than two thirds of AS patients with an inadequate response to NSAIDs.9 These agents are listed on the Pharmaceutical Benefits Scheme (PBS) for active AS not adequately responsive to exercise and NSAIDs, but not for non-radiographic axial SpA. As such, X-ray changes of sacroiliitis are required. Randomised controlled trials, however, suggest better clinical responses in early non-radiographic disease.
For IBP, all TNF inhibitors have similar efficacy and the choice of agent is often determined by extra-articular features and patient preference. Approximately 94% of AS patients commenced on treatment with a TNF inhibitor at Austin Health in Melbourne remain on long-term treatment (personal communication). To date, there is little evidence to suggest slowing of spinal fusion with TNF blockers.
Other DMARDs
Traditional DMARDs such as methotrexate and sulfasalazine have little effect in spinal disease, but can be useful for an associated peripheral arthritis. Other biologic agents available for rheumatoid arthritis in Australia have not been effective in AS.
Assessment and monitoring
In concert with the treating rheumatologist, the general practitioner has an important part to play in monitoring of AS patients. The goal of AS treatment in 2013 should be to achieve a state of minimal morning stiffness and gel phenomenon, stable measures of spinal mobility and optimal functional status. A significant change in any of these parameters should prompt semi-urgent review by a rheumatologist. General practitioners should familiarise themselves with the risks of TNF inhibitors, particularly infection. Among Australian patients, risk of melanoma and other skin cancers is important to recognise. Before major surgery, TNF inhibitors are generally withheld for 1–2 half-lives. The treating rheumatologist should be notified of any possible complications of treatment, to consider whether to withhold or cease treatment.
Key points
- Inflammatory back pain can be identified only by a targeted medical history.
- In early disease, spinal mobility may be normal.
- Identification of IBP should prompt more detailed evaluation and consideration of a multimodality treatment approach.
- The diagnosis of AS is established by IBP and the presence of definite X-ray changes of sacroiliitis.
- Non-radiographic axial SpA, with or without MRI changes, is often an early presentation of AS.
- Patient education and a tailored exercise routine are essential aspects of management.
- NSAIDs are first-line therapy in SpA.
- TNF inhibitors are indicated in AS patients with inadequate response to exercise and NSAIDs.
- Regular review of IBP symptoms and spinal mobility is important to determine the effectiveness of treatment.
Competing interests: None.
Provenance and peer review: Commissioned; externally peer reviewed.