Studies show that 40–60% of patients have compliance problems with their glaucoma medications.2,3 Difficulty squeezing or opening the bottle and lack of a partner are factors that may increase problems.2 There is large variability in the force required to deliver a single drop of various ophthalmic preparations.4
Eye drop instillation difficulties are also seen in patients who are unable to tilt their head back adequately and in those with tremor.5
Cataract and glaucoma are prevalent in the elderly, and both conditions are amenable to surgical intervention. With the advent of sutureless clear corneal incisions and antimetabolite enhanced glaucoma drainage blebs, contact of the eye drop bottle with the eye can have untoward consequences.2 Preservative-free eye drops such as lubricants are frequently packaged in containers which, when opened, have relatively sharp edges.6 The bottle tips of these containers have been shown to cause ocular surface trauma.7 There may also be an increased risk of infection as a result of surface trauma.
Furthermore, eye drop instillation may be particularly problematic in unstable environments such as on aeroplanes, where the dry atmosphere often necessitates the use of topical lubricants.
Given these problems of poor patient compliance and risk of injury, there is a need for an alternative method of administration of eye drops that addresses these issues.
A novel method
We describe a novel method for the administration of eye drops, which overcomes the main problems of conventional eye drop administration.
The method was first used while on an aeroplane. It was noted that an eye drop applied to the fingertip (conveniently the index finger of the dominant hand) held its shape, presumably because of surface tension (Figure 1). It was also noted that eye drop size could be varied and that for all but the largest volume eye drops, could be transported to the medial canthus of either eye without loss of the eye drop or spillage (Figure 2). We recommend that patients aim to place the drop in the region of the caruncle. Importantly, given the potential for contamination, prior hand washing is mandatory.
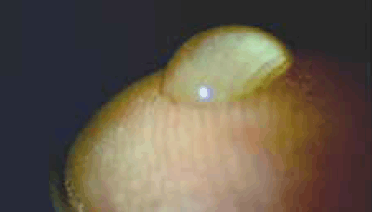
Figure 1. Eye drop applied to the fingertip, illustrating the holding of its shape
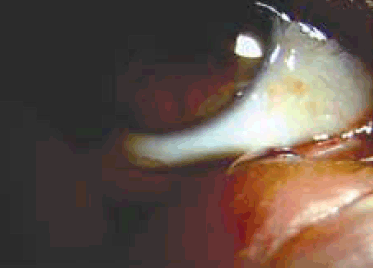
Figure 2. Eye drop being transferred to the medial canthus of the eye without loss of the eye drop or spillage
Discussion
In the authors’ experience, this method of eye drop instillation has been particularly useful in patients with arthritis who are unable to lift their hands (and the eye drop container) above eye level. It has also been useful in patients who are unable to hold or squeeze containers hard enough to extract an eye drop. To date, no episodes of conjunctivitis have been reported by our patients using this method.
As described, difficulties with eye drop application appear to be common, particularly in elderly populations. It is the authors’ experience that this difficulty is often not reported and is best elicited by direct questioning and observing a patient attempting to instil eye drops during a consultation.
While this issue has been recognised1–3,5,8 there have been only limited attempts at providing an adequate solution. A method of stabilising the bottle against the forehead with the thumb of one hand, positioning the dropper tip over the conjunctival sac while the other hand retracts the inferior lid has been described9 but does not appear to be in widespread use. in addition, a number of aids to facilitate eye drop self administration have been developed and patented.8 Such aids aim to address issues of augmenting the patient’s grip, squeeze, aim and ability to keep the eye open. Most are made of lightweight plastic with a cone configuration that fits over the eye, allowing separation of the dropper from the eye so that contamination risk is minimised. These devices do address some of the issues, particularly that of the inability to raise the arm above eye level if used with patient lying down. This may be convenient for glaucoma medications, but not so for ocular lubricants which may need to be instilled frequently during the day. The devices carry a cost and while supplied free with some medications, they are usually designed to take one manufacturer’s bottle type rather than being universal. If used with the patient in the erect position, they do not address issues of neck extension, arm raising difficulties, or tremor. For these reasons, their use appears to be limited.
An important consideration in the use of the technique we describe is the risk of infection. Contamination of eye drop containers occurs in as many as 35% of bottles opened for 2 weeks.10 While this has been in part addressed by the introduction of ‘single use’ dispensers (with the added advantage of allowing preservative-free medication), issues of cost and convenience can ultimately result in multiple usage with an increased risk of contamination.11 In the digital installation method, it is possible to deposit a drop on the fingertip without the container coming in contact with the finger.
Adequacy of hand washing remains a concern with our technique. There is, however, a fairly good safety record of hand washing in contact lens handling,12 and applying eye drops that have only a temporary residence time on the ocular surface is likely to be even safer than applying a contact lens using a clean hands technique. Admittedly, there is a place for determining more precise guidelines as to how hand washing should be carried out.
Summary
Our pragmatic method of eye drop instillation potentially provides a way of improving compliance and reducing the rise of ocular surface injury. With appropriate care, we believe that the relative risk of infection is small and this should be balanced against the risk of infection from conventional eye drop application, as well as the other adverse outcomes that may be associated with poor compliance. Further studies examining hand and ocular surface contamination using this method are required. Our experience to date, however, is that the technique is at least as safe as that used for insertion of contact lenses.
Competing interests: None.
Provenance and peer review: Not commissioned; externally peer reviewed.