Case study
A Caucasian man, 58 years of age, presented to his general practitioner with a 3 week history of acute-on-chronic lower thoracic back pain that radiated bilaterally to his lower limbs. There was no history of preceding trauma. He had a left hip arthroplasty performed 1 year before presentation, which had become infected 3 months earlier and was subsequently revised. He also suffered a dislocation of the same hip 1 month ago, which was treated conservatively in hospital. The patient smoked 20 cigarettes per day. He denied intravenous drug use.
Examination findings were unremarkable. Plain spinal X-rays revealed congenital fusion of T8–T9 vertebrae. An abdominal-pelvic computed tomography (CT) scan was unremarkable.
Several days later the patient presented to his local hospital with a 1 day history of decreased mobility, diminished lower limb sensation, as well as urinary and faecal hesitancy. He was afebrile. He had bilateral leg weakness: 4/5 power in the right leg and 1/5 power at the left knee and hip. There was an upgoing plantar reflex on the left foot and loss of anal sphincter tone.
Plain X-rays showed a burst fracture of T8 vertebral body. Magnetic resonance imaging (MRI) revealed long standing structural changes, with circumferential enhancement of the epidural tissue, discitis at T8 and T9, and a pathological fracture of T9 with posterior retropulsion causing a high grade spinal cord compression (Figure 1). His white blood cell count was 10.8 and C-reactive protein (CRP) was 220.
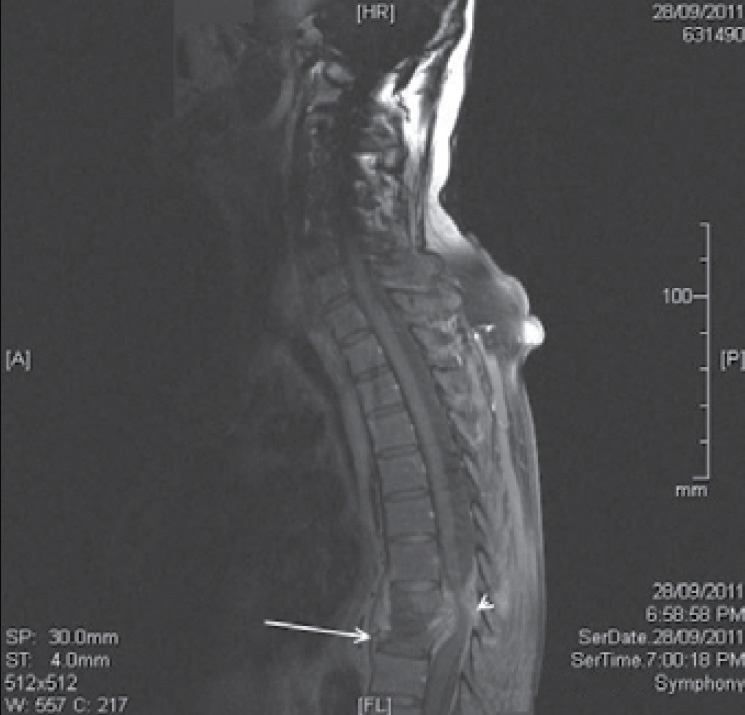
Figure 1. A sagittal section T1 weighted MRI showing pathological fracture of T8 and T9 vertebra (arrow) with spinal cord compression (arrowhead)
Question 1
What are the differential diagnoses to be considered?
Case study continued
The patient underwent T8–T9 decompression plus pedicle screw fusion (Figure 2). Histopathology showed granulomatous tissue consistent with infection. Microbiological, including mycobacterial, tissue culture was negative after 6 weeks of incubation. The patient completed a 6 week course of intravenous ceftriaxone and vancomycin. He continued to improve medically and was transferred to the spinal rehabilitation unit. At 3 months, the patient had recovered a considerable amount of motor function. However, residual left leg weakness necessitated mobilisation with a walking frame.
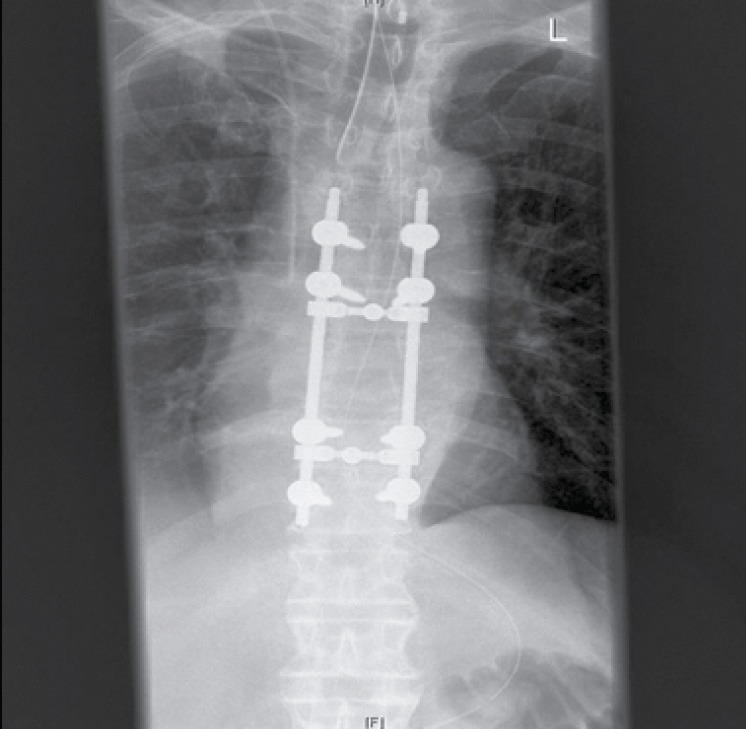
Figure 2. Plain AP X-ray after surgical decompression and pedicle fusion
Question 2
What are the key clinical features of spinal osteomyelitis?
Question 3
What are the complications of spinal osteomyelitis?
Question 4
What are the risk factors for the development of spinal osteomyelitis?
Question 5
What is the best imaging to confirm a suspicion of spinal osteomyelitis?
Answer 1
Osteomyelitis; malignancy.
Answer 2
Spinal osteomyelitis often has an indolent course, and presenting symptoms may be nonspecific, usually presenting as back pain (>80%), with or without associated fever. Only a minority of cases have neurogenic deficits on presentation (36%). The site affected is usually the lumbar (58%), followed by thoracic (30%) and cervical (11%) spine.1,2
The offending micro-organism is usually Staphylococcus aureus. Other causative microbes include Gram negative enteric bacilli, streptococci, fungi and tuberculosis.
Answer 3
The two most serious complications of spinal osteomyelitis are neurological impairment due to crush fracture and abscess formation. Urgent referral to a specialist spinal unit and administration of pathogen directed intravenous antibiotics results in the best chances of preventing these complications. Studies suggest that commencing empiric antibiotic therapy does not diminish microbial culture yield.3,4
Answer 4
It is noteworthy that this patient possessed no alarming examination findings during the first presentation to his general practitioner. The only ‘red flag’ in his history was an increased intensity of his chronic back pain.5
Spinal osteomyelitis is usually a disease of middle aged and elderly men,3 and most cases have at least one underlying medical comorbidity such as diabetes mellitus, excessive alcohol intake, renal disease, intravenous drug use, malignancy and other immunosuppressive conditions.1,3
Pathogens can reach the spine either by direct inoculation (after trauma or surgery), contiguous spread from adjacent structures, or by haematogenous seeding. The latter is the most common mechanism of infection as vertebrae possess an abundant, slowly flowing vascular supply.6
Recent surgery is a recognised risk factor in the pathogenesis of acute haematogenous osteomyelitis of the spine, especially when devices, including prosthetic joints, indwelling catheters or vascular grafts, are left in situ. A recent review found that 25% of cases of spinal acute haematogenous osteomyelitis had recently undergone an open surgical procedure within 6 months.3
Answer 5
Plain films or noncontrast CT scans may be negative or equivocal in individuals at risk for spinal osteomyelitis.7,8
Magnetic resonance imaging remains the most sensitive radiologic technique for detecting early vertebral osteomyelitis.7 Gallium bone scans are also sensitive and specific for vertebral osteomyelitis.8
Conflict of interest: none declared.