Case study
A man, 69 years of age, who previously had worked in the petroleum industry, presented with a 20 year history of an episodic rash on his arms, legs and trunk. He stated that the rash started with crops of raised, itchy, nonscaly reddybrown lesions, which would ulcerate and then regress over a 6 week period. Constitutional symptoms were absent. The rash was managed at different times as eczema and psoriasis, however, there was only ever a partial response to steroids. He had not had any laboratory or radiographic testing.
Over recent months the lesions had progressed, developing first into variable sized, erythematous, pruritic and scaly patches over his thigh, trunk and back. Some of these lesions measured up to 30 cm in diameter. Other lesions gradually progressed to form annular lesions overlying these scaly patches. The annular lesions were around 2–3 cm in diameter with central ulceration and exudate and appeared on top of the pre-existing erythematous scaly lesions (Figure 1). He had a single palpable right inguinal node. Constitutional symptoms remained absent.
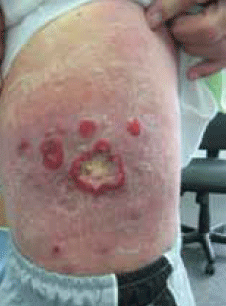
Figure 1. Lesions on the patient's trunk
Question 1
What diagnosis must be considered and excluded?
Question 2
What differential diagnoses would you consider for the rash shown in Figure 1?
Question 3
What initial investigations would you perform in this case?
Question 4
What diagnosis explains the clinical picture described above?
Case study continued
The patient's histopathological report of a punch biopsy of the rash is shown in Table 1. Immunoglobulin E was raised at 1681 kU/L. Other blood tests, including full blood examination (FBE), were normal. A computed tomography (CT) scan showed only a single enlarged right inguinal lymph node, measuring 2.2 cm in diameter.
Table 1. Histopathological report of a punch biopsy of the patient's rash
| Histopathological examination of lesional skin biopsies shows epidermal spongiosis and lymphocyte exocytosis. The dermis contains heavy interstitial lymphocytic and histiocytic infiltrate with atypical lymphocytes present. On immunofluorescence, these lymphocytes are positive for T-cell marker CD2 and CD3, CD4: CD8 ratio >1 and <1% CD30 positive cells. These findings are consistent with a peripheral T-cell lymphoma |
Question 5
What is the management of this condition?
Case study continued
The patient was commenced on oral methotrexate 40 mg weekly, a daily topical steroid application and daily wound dressings. He completed 4 weeks of rotational electron irradiation. Posttreatment there was no further evidence of disease activity.
Answer 1
A cutaneous T-cell lymphoma needs to be considered and ruled out in any skin rash initially thought to be benign that is refractory to treatment or progresses.
Answer 2
Differential diagnoses are outlined in Table 2.
Table 2. Differential diagnoses of the rash shown in Figure 17
| Condition | Features |
|---|
| Granuloma annulare |
Chronic skin condition consisting of an annular papular rash,usually affecting the posterior surface of the arms, hands or feet |
| Pityriasis lichenoides |
Spectrum of clinical presentations ranging from a chronic form involving small papular lesions, which become flat and regress over weeks, to an acute form comprising papular lesions that develop abruptly and evolve into pseudovesicles with subsequent necrosis and ulceration |
| Psoriasis |
An autoimmune disease involving red scaly patches on the scalp, elbows, knees and other aspects of the body. Also pustular and other varying forms |
| Sarcoidosis |
The spectrum of skin involvement in sarcoidosis includes lupus pernio, plaques, maculo-papular eruptions, nodular lesions, scar sarcoidosis, pruritis, erythema nodosum, nummular eczema, erythema multiforme and calcinosis cutis. Sarcoid has cutaneous involvement in one in five patients |
| Lymphomatoid papulosis |
Crops of red to red-brown papules that undergo central necrosis and spontaneously resolve with some scarring in ~2 weeks1 |
| Tinea corporis |
Has a range of clinical presentations. Commonly patchtype lesions with central clearing. Wood's lamp positive |
| Eczema |
Itchy, red scaly rash. Skin may become thickened and lichenified. Common in people with a family history of atopy |
Answer 3
A skin swab for microscopy and culture and a punch biopsy specimen for histology and immunofluorescence are both essential. Repeated punch biopsies may be required for a definitive diagnosis. Other helpful tests include:
- FBE, urea, electrolytes and creatinine (EUC), liver function tests (LFTs)
- inflammatory markers, including erythrocyte sedimentation rate (ESR) and c-reactive protein (CRP).
Answer 4
The most likely diagnosis is a cutaneous T-cell lymphoma. The initial long history of a spontaneously resolving ulcerated rash is best explained by lymphomatous papulosis, a primary cutaneous CD30+ lymphoproliferative disorder which, after mycosis fungoides, is the second most common group of primary cutaneous T-cell lymphomas.1 The recent history of progression is likely to be caused by development of patch stage mycosis fungoides, which further progressed to tumour stage disease with local node involvement (stage IVa).
Answer 5
The patient requires referral to a specialist oncologist, dermatologist and/or radiation oncologist. Often shared care between these specialties is required. Tumour, node, metastasis and blood staging forms the basis for the multidisciplinary approach to treatment and is the most important prognostic factor in mycosis fungoides.2
Management of patch/plaque stage mycosis fungoides
Patients with mycosis fungoides limited to the skin (patch/plaque stage) have a good prognosis.2 The goal of therapy is improvement in symptoms and quality of life while keeping treatment related toxicity to a minimum. This involves either skin directed therapies under the direction of a dermatologist or radiation oncologist or expectant management.2
Skin directed therapies include:
- topical corticosteroids
- topical alkylating agents
- topical bexarotene
- ultraviolet radiation (either UVA or UVB)
- total skin electron beam therapy.
Advanced stage mycosis fungoides
Patients with advanced mycosis fungoides including significant nodal, visceral or blood involvement are rarely curable3 and treatment is largely palliative. Combinations of skin directed therapies, biologic response modifiers and systemic chemotherapeutic agents are often used according to a stage based approach.2
Discussion
The slow evolution of a cutaneous T-cell lymphoma in the early phase can closely mimic many common benign chronic dermatoses.4 Mycosis fungoides is the most common form of cutaneous T-cell lymphoma.5 The aetiology of mycosis fungoides remains unknown, however, various theories implicate chronic antigenic stimulation through occupational, environmental or viral exposures. There are three classic cutaneous phases of the disease (Table 3).4
A diagnosis of mycosis fungoides can be difficult due to the variable early stage presentations and, in some cases, the coexistence of a different primary cutaneous lymphoma.5 Yen et al6 showed that the mean age at diagnosis (55 years) was significantly older than the mean age of onset of symptoms (48 years).
General practitioners are well placed to initiate the process of coming to a definitive diagnosis of a cutaneous T-cell lymphoma. Management involves referral to a specialist oncologist, dermatologist and/or radiation oncologist who will initiate accurate disease staging and risk stratification, and the selection of appropriate therapy.2
Table 3. Morphological patterns of mycosis fungoides1
| Form | Clinical features |
|---|
| Premycotic and patch phase |
Can last several years to several decades. The skin lesions can appear anywhere and often closely resemble benign inflammatory dermatoses |
| Infiltrative or plaque |
Skin lesions become thickened and develop denser, palpable, darker red or violaceous chronic inflammatory cell infiltrates |
| Tumour phase |
Lumpy tumours develop from pre-existing plaques, patches or unaffected skin. Tumours readily undergo necrotic ulceration |
Key points
- Cutaneous T-cell lymphoma is to be excluded whenever a cutaneous lesion is refractory to treatment.
- Mycosis fungoides is the most common type of cutaneous T-cell lymphoma and has three cutaneous phases.
Conflict of interest: none declared.