Table 1. Common causes of goitre
- Hashimoto thyroiditis
- Graves disease
- Familial or sporadic multinodular goitre
- Iodine deficiency
- Follicular adenoma
- Colloid nodule or cyst
- Thyroid cancer
|
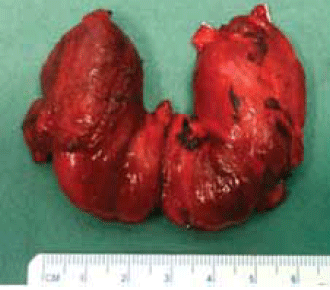
Figure 1. Macroscopic appearance of a thyroid gland removed from a patient with diffuse goitre secondary to Graves disease
When diffuse enlargement of the thyroid occurs in the absence of nodules and hyperthyroidism, it is referred to as a diffuse nontoxic goitre. This is sometimes called 'simple goitre' because of the absence of nodules, or 'colloid goitre' because of the presence of uniform follicles that are filled with colloid. Worldwide, diffuse goitre is most commonly caused by iodine deficiency and is termed 'endemic goitre' when it affects >5% of the population in a given geographic area (Figure 2).
The prevalence of goitre, diffuse and nodular, is dependent on the status of iodine intake of the population. In general, in iodine sufficient countries the prevalence of clinically palpable goitre is less than 4%. In countries with a previous deficiency corrected by universal salt iodination, elderly subjects may have a prevalence of approximately 10%. This can be attributed to lack of nutritional iodine in early adult life, as longstanding endemic goitres usually do not regress with iodine supplementation.1
Smoking is also known to be goitrogenic, with cigarette smoke containing goitrogens including thiocyanates. Smoking may also exacerbate autoimmune thyroid disease (and possibly autoimmunity in general). The goitrogenic effect of smoking is most pronounced in people with intercurrent iodine or selenium deficiency.2 Certain raw foods, such as soybeans, cassava and cruciferous vegetables, are lightly goitrogenic but their typical impact is only modest.3
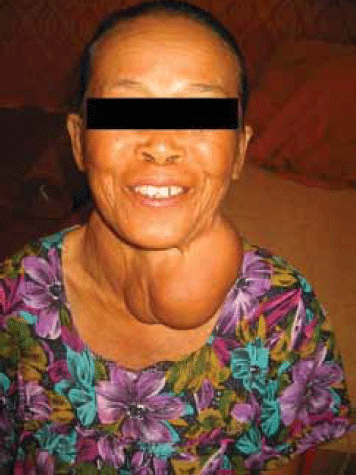
Figure 2. Woman from Northern Thailand with endemic goitre secondary to iodine deficiency
Iodine deficiency
Iodine deficiency has re-emerged in Australia over recent decades.4 Urinary iodine concentration (UIC) is an excellent proxy marker for current iodine intake and is a useful screening tool for iodine deficiency at a population level.5,6 However, day-to-day variations in UIC preclude the use of UIC as a diagnostic tool to assess the iodine nutritional status of an individual, unless multiple samples are collected over a period of weeks to derive an average level.7 As this is neither practical nor a prudent use of resources, the public health response has instead focused on supplementation. Since October 2009, all bread produced in Australia and New Zealand (with the exception of organic bread) must contain added iodine from iodised salt.8 Consequently, adults and children who eat bread regularly should receive sufficient quantities of iodine. However, the iodised salt in bread alone does not meet the increased daily requirements of the majority of pregnant and breastfeeding women. The National Health and Medical Research Council recommends that all women who are pregnant, breastfeeding or considering pregnancy, take an iodine supplement of 150 micrograms each day.9 Women with preexisting thyroid conditions should seek advice from their medical practitioner before commencing a supplement.
Clinical presentation
Nodular goitre is most often detected simply as a mass in the neck, but an enlarging gland may also produce pressure symptoms on the trachea and the oesophagus. As well as discomfort, there may be difficulty in breathing, dysphagia, cough and hoarseness. Paralysis of the recurrent laryngeal nerve may occur when the nerve is stretched taut across the surface of an expanding goitre, but this event is rare. Symptoms suggesting obstruction of the trachea may occur. These include cough, stridor and shortness of breath. Occasionally tenderness and a sudden increase in goitre size arise due to haemorrhage into a nodule. Symptoms of hyperthyroidism or hypothyroidism may be evident at presentation.
Thyroid examination
The thyroid gland is located superficially and, while usually not visible, is easily palpated. A careful examination should record the size, shape and consistency of the gland. The number, dimensions and consistency of any nodules should also be recorded. Importantly, in patients with long, thin necks, a prominent but normal thyroid gland may be perceived by an examiner as abnormal and in patients with short, fat necks, nodules may be extremely difficult to detect. The accuracy of thyroid palpation depends greatly on the experience of the examiner.
Substernal goitre may obstruct the thoracic inlet. Pemberton sign refers to faintness with evidence of facial congestion and cyanosis due to external jugular venous obstruction when the arms are raised above the head, a manoeuvre that draws the thyroid into the thoracic inlet.
Laboratory investigations
Patients with goitre or thyroid nodules should have a serum thyroid stimulating hormone (TSH) to determine the current functional status of the thyroid. If the TSH is abnormal, a free T4 and free T3 should also be checked. Checking for antibodies against thyroperoxidase (anti-TPO) and thyroglobulin (anti-TG) is recommended as thyroid autoimmunity may coexist with goitre. If the patient is hyperthyroid, a thyroid receptor antibody (TRAb) can help to confirm Graves disease.
Imaging
Due to the limitations of the physical examination, ultrasound has become an invaluable tool and an extension of clinical examination in specialist endocrinology practice. An ultrasound performed at the time of the initial clinical assessment is not only useful for diagnosis, but also as a baseline for monitoring the progress of thyroid volume or the size of nodules. Thyroid ultrasound should be performed in all patients with known or suspected thyroid nodules.10
A radionuclide thyroid scan is indicated in patients with a subnormal TSH to determine the functional status of any nodules within the thyroid and the underlying cause of clinical or subclinical hyperthyroidism. Since hyperfunctioning ('hot') nodules rarely harbour malignancy, if one is found that corresponds to the nodule in question no cytological evaluation is necessary.10
A computed tomography (CT) scan of the neck is not a routine part of the investigation of a thyroid nodule, but may be performed in patients with significant compressive symptoms, or to evaluate the degree of retrosternal extension or tracheal compression. In the presence of a nodule or goitre, CT scans should be ordered as 'noncontrast' due to risk of contrast induced hyperthyroidism or hypothyroidism in patients with nodular thyroid disease. Contrast should also be routinely avoided when imaging other regions in the presence of goitre unless essential for medical care.
Thyroid nodules
The normal thyroid gland is a fairly homogenous structure, but nodules often form within its substance. These nodules may be the result of growth and fusion of localised colloid filled follicles, or discrete adenomas or cysts. Thyroid carcinoma also typically presents as a nodule. Between 4% and 7% of the general population have a palpable thyroid nodule.11 Around 30–50% of adults have a thyroid nodule visible on ultrasound.12 The major challenge for the clinician and patient is determining which of these nodules are clinically important; a structured diagnostic approach is required (Figure 3).
Ultrasound enables characterisation of nodules and detection of other clinically significant nodules that are not palpable (Figure 4). Importantly, the risk of malignancy is the same for incidental impalpable lesions as for nodules of the same size that are palpable.13
Ultrasound is essential to determine those nodules, if any, which should be subjected to fine needle aspiration (FNA) cytology (Table 2).
Increasingly, thyroid ultrasound is being performed by endocrinologists and endocrine surgeons at the time of their initial assessment. For patients who require a biopsy, ultrasound is useful to guide the biopsy and essential when the nodule is not readily palpable, as ultrasound increases the likelihood of obtaining a diagnostic sample.14
There has been a move toward greater uniformity in thyroid FNA cytology reporting to help provide more clinically useful results.15 Any patient who doesn't have benign cytology should be referred for specialist opinion – this includes referring patients with results of undetermined significance on histology. It is recommended that all benign thyroid nodules be followed with serial ultrasound examination 6–18 months after the initial FNA to assess for interval change. If the nodule has grown significantly (>20% in two dimensions with a minimal increase of 2 mm), the FNA should be repeated.
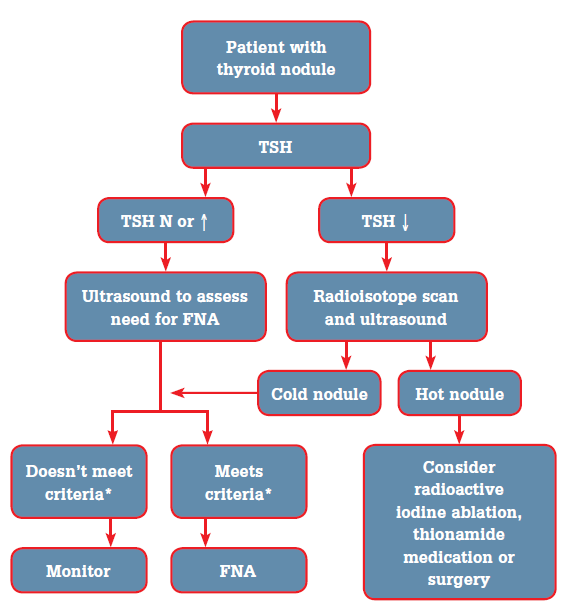
Figure 3. Diagnostic approach to a patient with a thyroid nodule
* Criteria refers to clinical and ultrasound features associated with an increased risk of malignancy and therefore warrant FNA (see Table 2)
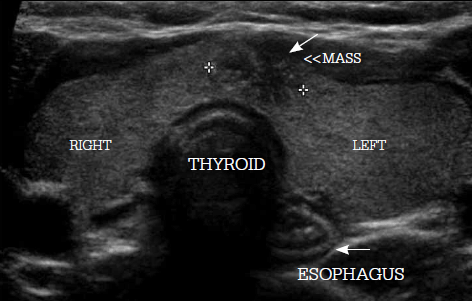
Figure 4. Thyroid ultrasound of a patient with an impalpable nodule incidentally discovered on ultrasound. Fine needle aspiration cytology of the lesion revealed a papillary carcinoma
Table 2. Which thyroid nodules to biopsy
|
|
>5 mm |
- Abnormal cervical lymph node
|
All nodules† |
|
|
>1 cm |
|
|
>1 cm |
|
|
>1.5–2 cm |
|
|
>2 cm |
|
|
FNA not indicated |
* High risk history includes head and neck irradiation, thyroid cancer in a first degree relative, radiotherapy or radiation exposure as a child, uptake on F–18 fluorodeoxyglucose positron emission tomography, multiple endocrine neoplasia type 2, elevated calcitonin
† In the case of cervical lymphadenopathy the cervical node itself is usually biopsied and, if thyroid cancer is present, a total thyroidectomy performed with the entire specimen subjected to histological examination |
Treatment of goitre
Potential treatment options for goitre will depend on the cause and the clinical picture. Options include observation, iodine supplementation, thyroxine suppression, thionamides (carbimazole or propylthiouracil), radioactive iodine (I131) ablation and surgery. Patients with an asymptomatic euthyroid goitre can usually be observed without specific treatment. Growth preventing intervention is usually unnecessary, as benign nodules usually grow quite slowly.16
Iodine supplementation will usually reduce thyroid volume in children and adolescents living in iodine deficient environments.17 However, for the general population and nonpregnant, nonlactating women in Australia, iodine supplementation over what is obtained from iodine fortified bread is not necessary. Iodine supplementation is unlikely to be beneficial for other forms of goitre.17 High dose iodine supplements, such as kelp, should be avoided as they have the potential to trigger hypothyroidism or hyperthyroidism.
Controlled trials have shown a beneficial effect of thyroxine treatment for both diffuse goitres and thyroid nodules. A goitre reduction of 20–40% can be achieved, but results are variable and potential long term harms of TSH suppression warrant consideration.18 The most difficult challenge for the clinician is to obtain suppression of the serum TSH level to between 0.5 and 0.1 mIU/L without going beyond this limit. A recent trial demonstrated the combination of thyroxine and iodine was more effective than either agent alone. Thyroid nodule reduction was achieved with TSH being kept in the lower part of the normal range to minimise potential side effects.19
Thionamides (carbimazole and propylthiouracil) are used in patients with thyrotoxicosis and a goitre due to Graves disease. Patients with multinodular goitre will also respond to thionamide medication, but definitive treatment with surgery is generally preferred. Radioactive iodine is another option and reduces goitre size by about 50% in the majority of patients over 6–12 months.20 However, the required dose is usually high and may need to be fractionated to ensure radiation safety. In patients with toxic adenoma, goitre is rarely a significant problem in its own right, but radioactive iodine can be helpful to treat the hyperthyroidism and could also shrink the adenoma. Radioactive iodine carries a significant risk of causing hypothyroidism over time, and so patients require annual TSH follow up. Occasionally, hyperthyroidism can also occur with radioactive iodine treatment.
Surgery is appropriate in patients who have troubling compressive symptoms and/or fail to respond to medical therapy. Thyroid surgery requires meticulous care to avoid damage to surrounding structures but is now a low risk procedure in experienced hands.21
Key points
- Goitre is common and is usually due to autoimmune disease, thyroid nodules or iodine deficiency.
- Thyroid stimulating hormone is the appropriate first test to assess the functional status of the thyroid.
- Patients who have a subnormal TSH should have a radionuclide thyroid scan and specialist advice should be sought on the most appropriate treatment.
- Thyroid ultrasound is an essential part of the evaluation of patients with goitre and will help determine the need for a FNA biopsy in patients with thyroid nodules.
- Patients with euthyroid goitre or benign nodules can generally be observed clinically with repeat ultrasound in 6–18 months. In some cases patients may be offered a therapeutic trial of thyroxine suppressive therapy. Other treatment options include thionamides (carbimazole or propylthiouracil), radioactive iodine ablation and surgery.
- Patients with suspicious nodules or atypical cytology should be referred for specialist evaluation.
Conflict of interest: none declared.