Case study
A woman, 82 years of age, presented to the emergency department with an 8 day history of worsening generalised abdominal pain, nonfaecal emesis and abdominal distension associated with a background history of 20 kg weight loss over the past few months. She described significant emotional distress since the sudden death of her eldest son 6 months previously.
Over the preceding months she had presented several times to her general practitioner complaining of epigastric pain, anxiety and weight loss. The GP provided counselling and prescriptions for a benzodiazepine (for episodes of acute distress) and omeprazole (for the epigastric pain). Her past medical history included incidentally detected gallstones, trigeminal neuralgia, anxiety and hypertension. She had no past history of abdominal surgery, diabetes or peptic ulcer disease. A gastroscopy performed 2 months prior was normal.
On examination in the emergency department, she was obviously distressed and had a tense distended abdomen, which was tympanic to percussion. A succussion splash-on was heard on auscultation.
Question 1
What is the differential diagnosis?
Case study continued
A plain abdominal X-ray showed a distended stomach (Figure 1). An abdominal computed tomography (CT) scan revealed a markedly distended stomach extending to the pelvic brim with compression of the small bowel (Figure 2).
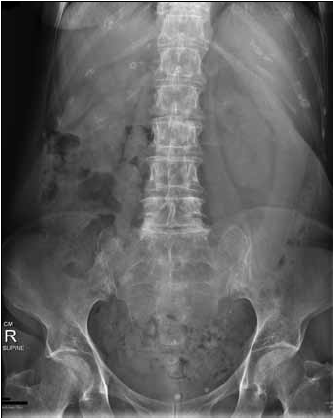
Figure 1. Plain X-ray of the patient's abdomen
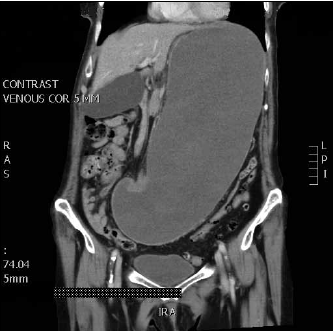
Figure 2. CT scan of the patient's abdomen
Question 2
What is the aetiology and pathophysiology of gastric distension?
Question 3
How is gastric distension diagnosed?
Question 4
What are the key elements in the initial management of this patient?
Question 5
What are the potential complications of gastric distension?
Answer 1
The history and examination findings suggest proximal gastrointestinal tract obstruction either at the level of the stomach (secondary to malignancy or a scarred benign prepyloric or duodenal ulcer) or small bowel (commonly adhesions, volvulus, hernias or neoplasm).
Answer 2
First described by Duplay1 in 1833, gastric distension most commonly occurs as a postoperative complication. Benign aetiologies include diabetes mellitus, trauma, resuscitation, electrolyte derangements, emotional stress, superior mesenteric artery syndrome, gallstone (duodenal bulb obstruction, known as Bouveret syndrome) and others (Table 1).2–4 In patients with an eating disorder, the stomach may distend to extremely high volumes.
Ischaemia and perforation as a result of gastric distension is extremely rare, as the stomach is well protected against ischaemic events due to a generous collateral circulation. Chronicity in gastric distension may also play a protective role. Ischaemia and perforation occur as a result of venous insufficiency, with the point of tension needed to cause mucosal ischaemia documented as 14 mmHg.2
Septic shock and multi-organ failure as a result of gastric perforation is invariably fatal.5
Table 1. Aetiology of gastric distension8
| Obstruction |
|---|
| Luminal |
Bezoar, ingested foreign body, hyperphagia, gallstone |
| Mural |
Gastric tumours, Crohn stricture, tuberculosis, iatrogenic (slipped gastric banding, anastomotic stricture) |
| Extrinsic |
Lymphoma, other intraperitoneal tumours or metastases, annular pancreas, superior mesenteric artery syndrome |
| Dysmotility |
|---|
| Autonomic neuropathy (diabetes mellitus, Parkinson disease), ileus, eating disorders |
Answer 3
Diagnosis of gastric distension from clinical signs can be a challenge in the clinical setting, as symptoms may overlap those of several other conditions including bowel perforation and blood or fluid collections. Common presenting symptoms include progressive abdominal distension and increasing abdominal pain accompanied by nonbilous vomiting. Hypotension, metabolic alkalosis and respiratory compromise may occur.6 If the stomach perforates, signs of generalised peritonitis may be obvious. A case has been reported of a patient with a massively distended stomach causing compromised blood flow in the abdominal aorta and leading to absent pulses in the lower limbs.7
Answer 4
The most important step in the initial management of this patient is to promptly decompress the stomach with a nasogastric tube (NGT). This will halt venous congestion and potential ischaemia and prevent aspiration. Fluid resuscitation with crystalloid should be initiated and titrated according to the patient's volume status, degree of renal impairment and electrolyte depletion. An urgent referral should be made to a surgical unit. Consultation with a gastroenterologist may also be required and the potential for the need for a referral to an intensive care unit should be considered early.
Answer 5
Complications of gastric distension include massive dehydration with hypochloraemic metabolic alkalosis and prerenal failure, and perforation with resultant sepsis, multi-organ failure and death. Complications are more likely in the elderly and already frail patients.
Case study continued
The patient had an initial potassium level of 4.2 mmol/L (reference range: 3.5–5.0). This dropped to 2.4 mmol/L after further vomiting and massive fluid losses from the NGT. Despite aggressive resuscitative efforts, the patient developed gastric necrosis, perforation and multi-organ failure. Combined anaesthetic, intensive care and surgical opinion concurred that she was not a surgical candidate and palliative management was initiated. The NGT was left in situ as a comfort measure.
Summary
This case illustrates the importance of early recognition and prompt referral and intervention in cases of progressive gastric distension in order to prevent serious adverse outcomes.
Conflict of interest: none declared.