Case
A personal trainer, 26 years of age, presented because of a 12-month history of secondary amenorrhoea since ceasing the oral contraceptive pill. In addition, a friend had commented on the yellow pigmentation of her hands, which had been present for an uncertain duration. Menarche occurred at 13 years of age, and menses had been regular prior to commencing the oral contraceptive pill for menorrhagia. The patient did 30–45 minutes of physical exercise five days per week and reported having a balanced diet. There were no other symptoms suggestive of pituitary hormone deficiency or mass effect. On examination, the patient had yellow hands (Figure 1) and feet, sclerae were normal and body mass index (BMI) was 24.1 kg/m2. She was clinically eupituitary, visual fields and fundi were normal, and a pregnancy test was negative. Laboratory investigations are presented in Table 1.
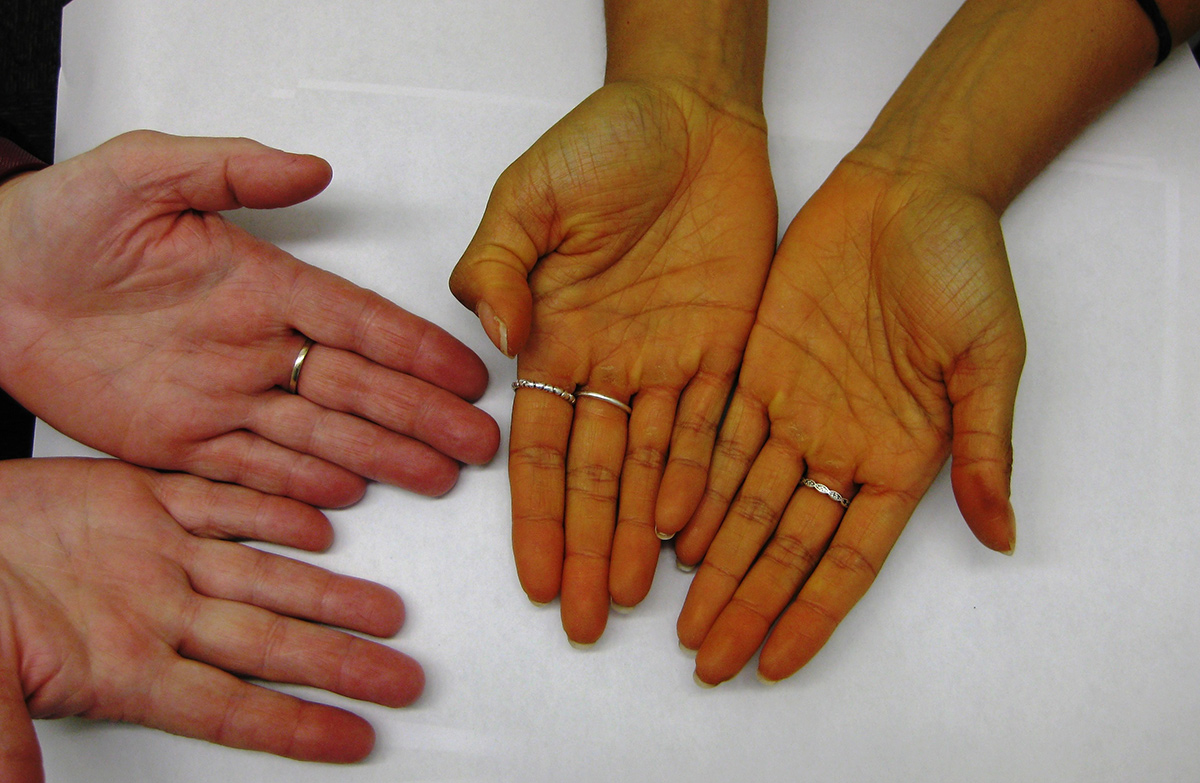
Figure 1. Patient's hands (right) and control (left)
Table 1. Laboratory investigations
|
Investigation
|
Result
|
Reference interval
|
|---|
|
β-carotene
|
12.1 μmol/L
|
0.3–1.4 μmol/L
|
|
Vitamin A
|
1.0 μmol/L
|
1.6–2.3 μmol/L
|
|
Luteinising hormone
|
4.6 U/L
|
1–8 U/L*
|
|
Follicle-stimulating hormone
|
3.2 U/L
|
1–12 U/L*
|
|
Serum oestradiol
|
42 pmol/L
|
80–450 pmol/L*
|
|
*Follicular phase
|
Question 1
What is the most likely cause of the patient’s skin discolouration?
Question 2
How would you investigate the skin discolouration further?
Question 3
What are the potential causes of the patient’s secondary amenorrhoea? How would you investigate this further?
Question 4
Is there any association between the skin discolouration and secondary amenorrhoea?
Answer 1
Carotenaemia is the most likely cause of the young woman’s skin discolouration. β-carotene is predominantly deposited in the stratum corneum, sweat and sebum; thus, pigmentation is noted in areas where sweating is marked, such as the palms and soles. The pigmentation is enhanced under artificial light. Pigmentation with jaundice is usually diffuse and most prominent in the sclerae, while the eyes are spared in carotenaemia.
Hyperbilirubinaemia was not considered in this case because the sclerae were spared. Yellow pigmentation due to lycopenaemia also typically affects the palms and soles, but has a deeper tinge than is seen in carotenaemia.1 Lycopene is a physiologically inert isomer of carotenoid found in tomatoes, rose hips and berries. Riboflavinaemia may also cause yellow skin. Several drugs and chemicals may also cause yellow pigmentation of the skin.
Answer 2
Elevated serum β-carotene levels confirm the diagnosis of carotenaemia. Together with the clinical picture, measurement of vitamin A, thyroid function, lipid profile and liver function, and exclusion of proteinuria, will assist in determining the underlying cause (Table 2).
Table 2. Differential diagnosis of carotenemia1
|
Serum levels
|
Mechanism
|
|---|
|
|
Excessive dietary intake
- Carotene-rich foods
- Dietary supplements
- Tanning pills
- Palm oil
|
Hyperlipidaemia
- Hypothyroidism
- Type 2 diabetes mellitus
- Restrictive eating disorder
- Nephrotic syndrome
|
Failure of conversion carotene to vitamin A
- Hypothyroidism
- Restrictive eating disorders, liver disease
- Enzyme deficiency
|
|
Carotene
|
Elevated
|
Elevated
|
Elevated
|
|
Vitamin A
|
Elevated
|
Elevated/normal
|
Decreased/normal
|
|
Lipids
|
Normal
|
Elevated
|
Normal
|
The most common cause of carotenaemia is excess dietary intake of carotene.1 Carotenaemia may be seen four to seven weeks after commencement of a carotenoid-rich diet. Foods that are rich in carotene include sweet potatoes, carrots, dark green leafy vegetables, squash, peppers and apricots. Raised levels of serum cryptoxanthine and zeaxanthine/lutein confirm high dietary intake of carotenoids. Carotenaemia may occur with nutritional supplements or the use of palm oil for cooking, a common practice in west Africa.
Carotenaemia seen in individuals with eating disorders may be multifactorial.2 Mechanisms may include excessive dietary intake, hypercholesterolaemia and failure of enzymatic conversion of β-carotene to vitamin A.
β-lipoprotein is the major carrier of β-carotene in plasma.3 Type IIa hyperlipidaemia may result in carotenaemia in individuals with hypothyroidism, diabetes mellitus, nephrotic syndrome and eating disorders.1
Answer 3
The ‘inappropriately’ normal luteinising hormone and follicle-stimulating hormone results, paired with low oestradiol levels, are consistent with hypogonadotropic hypogonadism (HH). Causes of secondary amenorrhoea due to HH include functional hypothalamic amenorrhoea, pituitary disease (eg hyperprolactinaemia), post-pill amenorrhoea and medications or drugs.4
The major causes of functional hypothalamic amenorrhoea include restrictive eating disorders, low body weight, excess exercise and stress.5 In addition, acute illness, chronic systemic disease (eg type 1 diabetes mellitus, coeliac disease) and nutritional deficiency may cause HH.4 Pituitary tumours, infiltrative disorders, infections, trauma and vascular insults may cause HH.4 Elevated prolactin may occur secondary to pituitary disease, hypothyroidism, medications and renal dysfunction. The prevalence of post-pill amenorrhoea in Australian women has been estimated to be one in 200 for a six-month duration, and one in 400 for longer than 12 months.6 Other medications or drugs implicated in causing HH include opioids, anabolic steroids and contraceptive progestogens.
Investigation into secondary amenorrhoea with HH should include measurements of serum prolactin, thyroid function and other anterior pituitary function tests, and imaging of the pituitary with computerised axial tomography or magnetic resonance imaging.
Answer 4
The most common link between carotenaemia and HH is restrictive eating disorders, particularly in women. Hypothalamic amenorrhoea was reported in 12 women of healthy body weight with dietary carotenaemia.7–9 Page described bright yellow ovaries with strongly positive staining for carotenoid in six women with menstrual irregularity and dietary carotenaemia.10 Carotene‑containing ‘tanning’ pills have been associated with menstrual disorders. Reversible HH has also been reported in a young male with dietary carotenaemia.11
Case continued
The patient was found to have elevated serum β-carotene levels and low vitamin A levels, which were measured by ultra−high pressure liquid chromatography (Table 1). The results were confirmed on three occasions in two laboratories. Lipids, serum bilirubin, thyroid and liver function, serum prolactin, urine protein, and anterior pituitary function were normal. Pituitary morphology was normal on imaging. The patient repeatedly denied excessive intake of carotene-rich foods or vitamin supplements, and the low serum vitamin A was not consistent with dietary carotenaemia. She had not noticed the change in her skin pigmentation until her friend brought it to her attention.
Discussion
The combination of elevated β-carotene with low vitamin A levels is most likely due to a failure in enzymatic conversion of carotene to vitamin A. A loss-of-function mutation in β-carotene-15, 15’-monooxygenase has previously been described in carotenaemia and hypovitamosis A.12,13
β-carotene levels rise linearly with age, and this may provide an explanation as to why the patient had not noted the discolouration until young adulthood.14 Additionally, a change in her dietary intake of carotenoids may have precipitated a rise in β-carotene levels.
Whether the young woman’s secondary amenorrhoea was simply related to prolonged post-pill amenorrhoea or another mechanism is unclear. Her body fat, BMI and stated exercise regime are not consistent with functional hypothalamic amenorrhoea due to excess exercise or a restrictive diet. The mechanism by which reversible HH occurs in men and women with dietary carotenaemia has not been elucidated. It is interesting to consider whether pituitary or hypothalamic deposition of carotene, as seen in the ovarian histology described by Page, could cause HH in a mechanism similar to that seen with pituitary or hypothalamic iron deposition with haemochromatosis. It has recently been shown that male mice lacking β-carotene-15, 15’-monooxygenase have HH.15
Key points
- Carotenaemia should be considered in any patient with yellow discolouration of the skin with scleral sparing.
- The most common causes of carotenaemia are excess dietary intake of carotene and restrictive eating disorders.
- The most common causes of secondary amenorrhoea with a picture of HH are eating disorders, excessive exercise, stress and hyperprolactinaemia.
- Post-pill amenorrhoea may last for longer than six months in approximately one in 200 women.
Authors
Adam Morton FRACP, Senior Staff Specialist Endocrinology and Obstetric Medicine, University of Queensland, St Lucia, Qld; Mater Health, South Brisbane, Qld. Adam.morton@mater.org.au
Sarah Morton, final-year medical student, University of Queensland, St Lucia, Qld
Competing interests: None.
Provenance and peer review: Not commissioned, externally peer reviewed.