Case
A woman aged 78 years was admitted to hospital with gallstone-induced acute pancreatitis. She had a five-hour history of epigastric pain, high lipase 7040 U/L, bilirubin 66 µmol/L, deranged liver function tests and leukocytosis (white cell count of 12.1 x 109/L). Multiple gallstones were visualised on abdominal ultrasound. Her heart rate, blood pressure, temperature and respiratory rate were within normal limits and she was managed conservatively after she declined a cholecystectomy. On day 2, the patient developed redness and pain over both of her anterior shins, but these were worse on the right shin. There was circumferential erythema with a purpuric patch above the right medial malleolus and two 1 x 2 cm erythematous nodules on the left pretibial region (Figure 1). Her medical history included a high body mass index (BMI) and hypothyroidism, which was controlled with maintenance thyroxine 50 µg daily. The patient had a chronic cough and had been diagnosed with eosinophilic pneumonia one month earlier. Since then, she had been on a tapering dose of prednisone and is currently taking 12.5 mg per day.
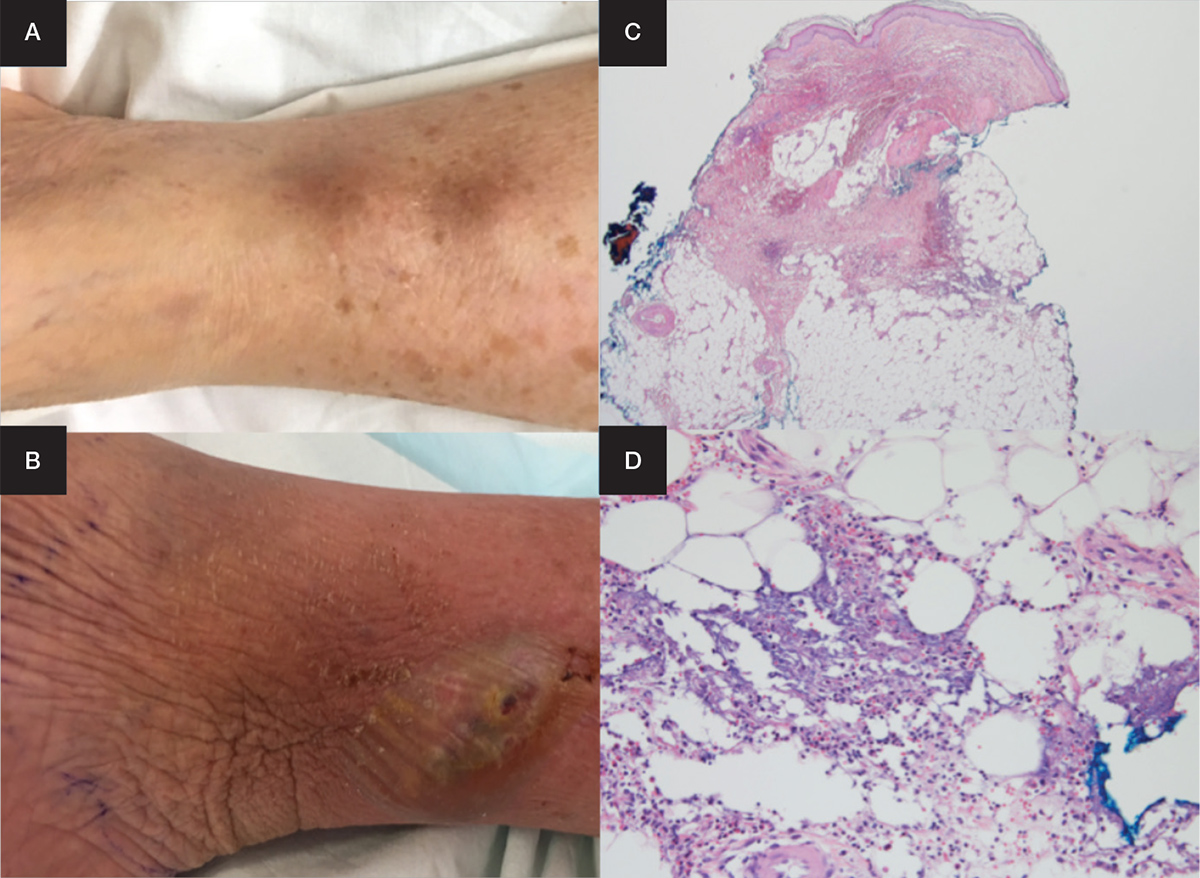
Figure 1. Lesions on the right and left shin
Images shown are of two nodules on the left shin (A); liquefaction of a single large nodule on the right shin (B); incisional biopsy at magnification x20 (C) and x400 (D)
Question 1
What are the differential diagnosis in this case?
Answer 1
The differential diagnoses and key cutaneous and histological features are summarised in Table 1.
Table 1. Differential diagnoses, and clinical and histological features
|
Differential diagnoses
|
Clinical features
|
Histological features
|
|---|
|
Pancreatic panniculitis
|
- Painful, erythematous nodules that often ulcerate and drain oily, brown fluid (liquefaction)
- Pretibial area, knees and ankle
- Associated with visceral fat necrosis in omentum and peritoneum – abdominal pain, elevated pancreatic enzymes7,8
|
- Changes in a lobular distribution
- Fat necrosis, ‘ghost cells’ (anucleate adipocytes)
- Neutrophils and haemorrhage adjacent to fat necrosis
- Stippled basophilia representing calcification
- Acute and chronic inflammation in subcutaneous adipose tissue away from fat necrosis7,9
|
|
Traumatic panniculitis
|
- History of physical or chemical trauma, cold and electricity exposure
- Intensity of trauma is not related to the severity of cutaneous lesion
- Lesions are indurated, warm, red subcutaneous plaques or nodules that can have oily, brown discharge
- Occurs in all age groups7,8
|
- Acute lobular inflammation in acute cases but mononuclear infiltrate in longstanding lesions
- Usual fat necrosis of nodulocystic fat necrosis, calcification or foreign body type giant cell reaction around foreign material
- Organising haematoma in blunt trauma cases7,9
|
|
Acute erythema nodosum
|
- Tender, erythematous nodules
- Anterior shin
- Ulceration is rare
- Common in young women
- Systemic features are common: fever, arthralgia, headache, abdominal discomfort and ocular symptoms7
|
- Septal panniculitis in early lesions but may involve part or whole lobule in chronic lesions
- Inflammatory cells comprise neutrophils, lymphocytes and histiocytes, but no vasculitis
- Septal fibrosis and granulomatous inflammation in chronic lesions
- Meischer microganulomas – small groups of macrophages within septa9
|
|
Nodular vasculitis
|
- Tender, dusky subcutaneous nodules
- Posterolateral aspects of legs
- Often ulcerative and suppurative – oily fluid
- Young–middle aged women with venous stasis changes
- Chronic, relapsing course7,8
|
- Septal and lobular panniculitis, often nodules of inflammation scattered throughout lobule
- Acute vasculitis involving small veins and venules are present, and inflammatory cells comprise neutrophils, lymphocytes and histiocytes
- Fat necrosis often present and granulomas may be seen
- Fibrosis of septa and lobules in chronic lesions, and granulomas with giant cells may still be present9
|
|
Infectious causes:
- Erysipelas
- Cellulitis
- Necrotising fasciitis
|
- Acute painful red swelling in elderly patient (often cannot recall minor trauma/insect bite)7
- Reduced immune defenses as patient on prednisone
- Usually unilateral, and fever is common
|
- Mixed septal and lobular panniculitis
- Extensive neutrophilic infiltrate, haemorrhage, basophilic necrosis, and vascular proliferation
- Granulomatous inflammation with giant cells
- Special stains (eg PAS, GMS and Gram stain) may be helpful8,9
|
|
Lipodermatosclerosis
|
- Indurated plaques, with erythema and oedema
- Medial aspects of legs and ankle
- Secondary to venous insufficiency
- Female predilection, aged 40s, obesity common7,8
|
- Overlying dermis shows vascular stasis changes
- Early lesions show fat infarction, centrilobular ischaemia and vascular congestion
- Late stage lesions have microcystic changes, hyalinised fat lobule. Lipophagic changes, septal scarring leading to subcutaneous atrophy8,9
|
Case continued
Bilateral venous Doppler ultrasound scans were negative for deep vein thrombosis and the patient had no acropathy or ocular symptoms. Initially, the patient was commenced on oral flucloxacillin and then changed to benzylpenicillin for suspected Streptococcus pyogenes infection. However, there was no bacterial growth in the aspirated bulla fluid and the patient’s symptoms did not improve. At this point, an incisional biopsy was obtained and histopathological examination showed fat necrosis with surrounding inflammatory infiltrate. A review of the abdominal ultrasound and magnetic resonance cholangiography excluded the possibility of a pancreatic malignancy. The patient improved with an increase of prednisone to 20 mg per day plus wet dressings (betamethasone + 50% emulsifying ointment) and she was discharged with a weaning prednisone regimen. On follow‑up two weeks later, the patient’s pain, swelling and erythema had significantly improved. However, an ulcer developed over the biopsy site, which is now managed with dressings.
Question 2
What is the most likely diagnosis?
Question 3
How is this condition diagnosed? What is the aetiology?
Question 4
What is the management for the diagnosed condition?
Question 5
What is the prognosis for the diagnosed condition?
Answer 2
Given the temporal relationship and histological findings, the most likely diagnosis is pancreatic panniculitis. Subcutaneous fat necrosis affects 2–3% of all patients with pancreatic disease.1,2 This is usually associated with malignancy, but can be a complication in acute and chronic pancreatitis.3,4 Male gender (2:1) and heavy alcohol consumption are key risk factors.4,5 Cutaneous lesions usually precede underlying pancreatic disease, but may occur at any point.2 Hughes et al reported that panniculitis was the presenting symptom in 65% of patients with pancreatic carcinoma.4 There are three subtypes of pancreatic panniculitis:6
- type 1: erythematous plaques on trunks and buttocks
- type 2: scattered nodules on lower extremities
- type 3: a mixture of types 1 and 2.
This case is consistent with type 2, which is the most common subtype (46% of cases).
Answer 3
The diagnosis is based on history, cutaneous morphology and histology. Characteristic clinical features are painful, oedematous, erythematous nodules that ulcerate spontaneously. There is usually a sterile discharge consisting of byproducts of fat necrosis. Patients may report associated monoarthralgia or oligoarthralgia. Infection should be clinically excluded. Other differentials are excluded via histological tissue examination. A skin biopsy is best obtained through an elliptical incisional biopsy down to subcuits.3,6 Classical pathological findings are fat necrosis and adipocytes without nuclei.7
The pathogenesis has not been clearly delineated. However, it is postulated that pancreatic enzymes induce inflammation and vessel permeability, which gives lipase access to adipocytes.5,6 Lipase then hydrolyses the adipocytes, resulting in necrosis.2–4,6 Dhawan et al demonstrated intracellular staining of adipocytes with monoclonal antibodies to lipase.5 Arthritis is a result of high levels of fatty acids in joint spaces from sinus tracts between cutaneous lesions and nearby joints.1,3
Answer 4
Treatment is supportive and focused on diagnosis and management of underlying pancreatic disease.8 This could be cholecystectomy for gallstone pancreatitis, pancreatic resection for malignancy or pancreatic duct stent for chronic pancreatitis.1,4 Evidence for oral prednisone, topical prednisone and nonsteroidal anti-inflammatory medication is inconclusive.
Answer 5
Mortality can be as high as 24% in those with panniculitis due to acute pancreatitis. Patients with type 1 panniculitis tend to have more severe acute pancreatitis, with 80% of cases being severe.6 The mortality rate rises to 74% when the underlying cause is carcinoma.4 Poor prognostic features are polyarthritis, serum eosinophilia and subcutaneous nodules. Together, these features are known as Schmidt’s triad.1 For those who survive their underlying pancreatic disease, the cutaneous disease usually resolves concomitantly.3 The average duration of symptoms is 15–30 days in acute pancreatitis and more prolonged if it is malignancy-induced.6,7
Key points
- Pancreatic panniculitis can precede pancreatic pathology, more than half of which is pancreatic carcinoma.
- Consider pancreatic panniculitis as a differential diagnosis in patients with painful lower leg lumps after common causes have been ruled out, as such lumps can be a harbinger of indolent pancreatic carcinoma, a difficult condition to detect early.
- Diagnosis is achieved through clinical and histological correlation. A deep incisional biopsy that includes sufficient subcutaneous tissue is essential.
- The mainstay treatment is thorough investigation for underlying pancreatic disease, timely referral for definitive treatment and supportive care.
Authors
Linda Chan MBBS, Senior Medical Resident Officer, Concord Repatriation General Hospital, Concord, NSW. lindachan.health@gmail.com
Kenneth Lee MBBS, FRCPA, Senior Staff Specialist, Head of Department and Clinical Director of Anatomical Pathology, Concord Repatriation General Hospital, Concord, NSW; Clinical Senior Lecturer, the University of Sydney, Concord Clinical School, NSW
David Cook MBBS, FACD, Consultant Dermatologist, Visiting Medical Officer, Concord Repatriation General Hospital, Concord, NSW; Clinical Senior Lecturer, the University of Sydney, Concord Clinical School, NSW
Competing interests: None.
Provenance and peer review: Not commissioned, externally peer reviewed.