Adrenal incidentalomas are adrenal lesions ≥1 cm identified serendipitously on imaging performed for unrelated indications.1 The widespread use of cross-sectional imaging has highlighted this entity, which can present a management dilemma for the clinician.2 Although most lesions are benign, non-functional adrenal adenomas, further evaluation is necessary to determine whether a lesion may be hormonally active or malignant, as this would affect future management decisions.
Differential diagnosis
Overall, benign, non-functioning adrenal adenomas account for about 80% of adrenal incidentalomas. Of the tumours that are functional, 5% are pheochromocytomas, 5% cortisol-producing and 1% aldosterone-producing. Adenomas that produce sex hormones are very rare. Malignant tumours, such as primary adrenocortical carcinomas, account for <5% and metastases for <3% of adrenal incidentalomas. Tumours that commonly metastasise to the adrenal gland include lung, colon, breast, renal, stomach, melanoma and lymphoma.2
Clinical significance
Most incidentalomas are benign and non-functioning; however, patients need a systematic pathway of evaluation as outlined in Figure 1. Evaluation of incidentalomas is to determine whether there are clinical (or subclinical) manifestations and malignant potential, which may be primary or secondary. Almost 20% of patients with adrenal incidentalomas have subclinical hormonal abnormalities, which have been associated with an increased risk of metabolic, cardiovascular and bone diseases.3 Thus, identification of the incidental adrenal lesion provides the primary care physician with an opportunity to undertake early prevention strategies. Although primary malignancy of the adrenal is rare, with an incidence of 0.5–2 per 1 million, it is highly aggressive and has a poor prognosis, highlighting the importance of thorough clinical evaluation.4,5
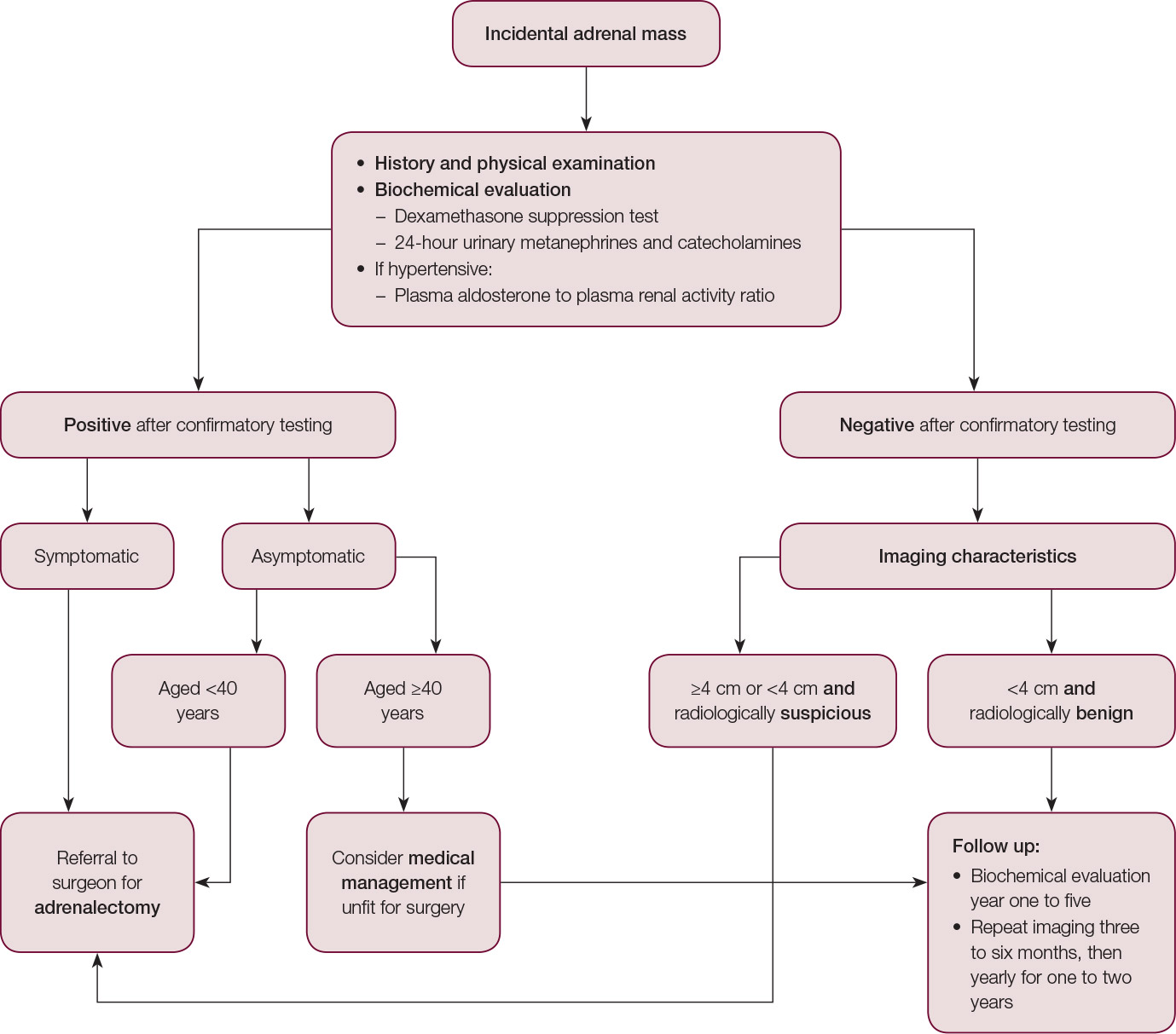
Figure 1. Algorithmic approach to the incidental adrenal lesion
Clinical evaluation
In the evaluation of adrenal lesions, the clinician must ask:
- Is the adrenal lesion functional or non‑functional?
- Is the adrenal lesion benign or malignant?
Evaluation begins with a thorough history and physical examination, focusing on signs and symptoms of adrenal hormone excess and malignant disease. This is followed by biochemical testing. Assessment should be undertaken for the following conditions, which are summarised in Table 1.
Cushing’s syndrome
Hypercortisolism can lead to a well-known array of signs and symptoms (Table 1). In some patients, hypercortisolism may be subclinical and symptoms may be absent or very mild, and easily overlooked.6,7
Metabolic evaluation
Elevated 24-hour urinary cortisol levels are often a late finding in patients with Cushing’s syndrome. Patients with subclinical Cushing’s syndrome can have normal 24-hour urinary cortisol levels; therefore, direct measurement of autonomous adrenocortical secretion is recommended.8,9 This is achieved by the 1 mg overnight dexamethasone suppression test. A serum cortisol levels of >50 nmol/L is suggestive of Cushing’s syndrome.7,10 With 91% sensitivity, confirmatory testing with a high-dose (8 mg) dexamethasone suppression test, serum adrenocorticotropin (ACTH) and cortisol in response to corticotropin‑releasing hormone, and midnight salivary cortisol should be undertaken to limit the possibility of a false-positive result.2,9,10
Pheochromocytoma
Patients with pheochromocytoma may be asymptomatic or have episodic symptoms because of catecholamine excess (Table 1). Severe hypertension is often seen; however, up to 15% of patients are normotensive. Pheochromocytomas can be associated with familial syndromes such as von Hippel–Lindau disease, multiple endocrine neoplasia type 2, familial paraganglioma syndrome and neurofibromatosis.7
Metabolic evaluation
There is currently no consensus as to the optimal biochemical test for pheochromocytoma.7 Medications such as tricyclic antidepressants and decongestants should be ceased to reduce the likelihood of a false-positive result. Fractionated plasma metanephrines have a high sensitivity of 96–100%, but a specificity of 85–89%. Measurement of 24-hour urinary metanephrines and catecholamines yields a sensitivity of 90% and specificity of 98%.7,9,11 Therefore, as a screening test in low-risk patients, minimising the likelihood of false-positive results with the 24-hour urinary measurement is preferred.7,10–13
Primary hyperaldosteronism
Primary hyperaldosteronism (Conn’s syndrome) is usually asymptomatic. Hallmarks include refractory hypertension, usually requiring more than three antihypertensive agents, and hypokalaemia or severe, diuretic-induced hypokalaemia. Most patients with primary hyperaldosteronism are normokalaemic.
Table 1. Summary of symptoms, signs and recommended screening tests for functioning adrenal lesions
|
Syndrome
|
Symptoms
|
Signs
|
Screening test
|
Confirmatory test
|
|---|
|
Cushing’s syndrome or subclinical Cushing’s syndrome
|
May be asymptomatic
Moon face, acne, buffalo hump, supraclavicular fat-pads, central obesity, striae, easy bruising, poor wound-healing, emotional and cognitive changes
|
Hypertension
Hyperglycaemia
Hyperlipidaemia
Osteoporosis
|
1 mg overnight dexamethasone suppression test
|
8 mg dexamethasone suppression test
Midnight salivary cortisol
|
|
Conn’s syndrome
|
Mostly asymptomatic
Muscle cramps, periodic paralysis, headaches, palpitations
Polydipsia, polyuria
|
Refractory hypertension
Hypokaleamia
(<3.5 mmol/L)
|
If hypertensive, plasma aldosterone to plasma renin activity ratio
|
Plasma aldosterone
24-hour urinary aldosterone, sodium, creatinine
|
|
Phaeochromocytoma
|
May be asymptomatic
Palpitations, flushing, sweating, headache, tremor, anxiety
|
Severe
hypertension
Weight loss
|
24-hour urinary catecholamines and metanephrines
|
Fractionated plasma metanephrines
|
|
Adrenocortical carcinoma
|
Mass effect
Palpitations, flushing, sweating, headache, tremor, anxiety, hirsuitism, gynaecomastia, amenorrhoea, infertility
|
Severe
hypertension
Weight loss
|
24-hour urinary catecholamines and metanephrines and serum androstenedione, testosterone, dihydroepiandrosterone sulfate, 17ß estradiol (in post-menopausal women)
|
Fractionated plasma metanephrines
|
|
Metastasis
|
Mass effect
Usually non-functional
|
Malignant disease elsewhere
|
Usually non-functional
24-hour urinary catecholamines and metanephrines and serum androstenedione, testosterone, dihydroepiandrosterone sulfate, 17ß estradiol (in post-menopausal women)
|
Fractionated plasma metanephrines
|
Metabolic evaluation
Screening for primary hyperaldosteronism is recommended for patients with an adrenal mass who are hypertensive. A plasma aldosterone concentration to plasma renin activity ratio is the recommended screening test.7,10 Patients must cease aldosterone receptor antagonists for four to six weeks prior to screening. A plasma aldosterone concentration to plasma renin activity ratio of ≥20 is abnormal and autonomous aldosterone secretion should then be confirmed with a plasma aldosterone concentration measurement while the patient maintains a high-sodium diet. A non-suppressed plasma aldosterone concentration is diagnostic.
Another important cause of hyperaldosteronism is primary adrenal hyperplasia, which may be unilateral or bilateral.7 The distinction between the two entities is critical because aldosterone excess secondary to an adrenal adenoma is successfully managed with unilateral adrenalectomy. Bilateral adrenal hyperplasia, however, is best managed medically with aldosterone receptor antagonists. Therefore, adrenal venous sampling to confirm that the unilateral adrenal mass and not bilateral adrenal hyperplasia is the source of excess aldosterone is recommended in patients aged >40 years and in those with bilateral adrenal masses.7,9,14
Adrenocortical carcinoma
Adrenocortical carcinomas are generally larger than benign incidental adrenal masses and may be heralded by symptoms of mass effect or adrenal hormone excess (Table 1).
Metabolic evaluation
Testing for all adrenal hormones is recommended as the results can be used in postoperative surveillance and monitoring of the disease (Table 1).7
Radiological evaluation
The two primary predictors of malignancy within an adrenal incidentaloma are the size of the mass and imaging characteristics on computed tomography (CT; Figures 2, 3) or magnetic resonance imaging (MRI).
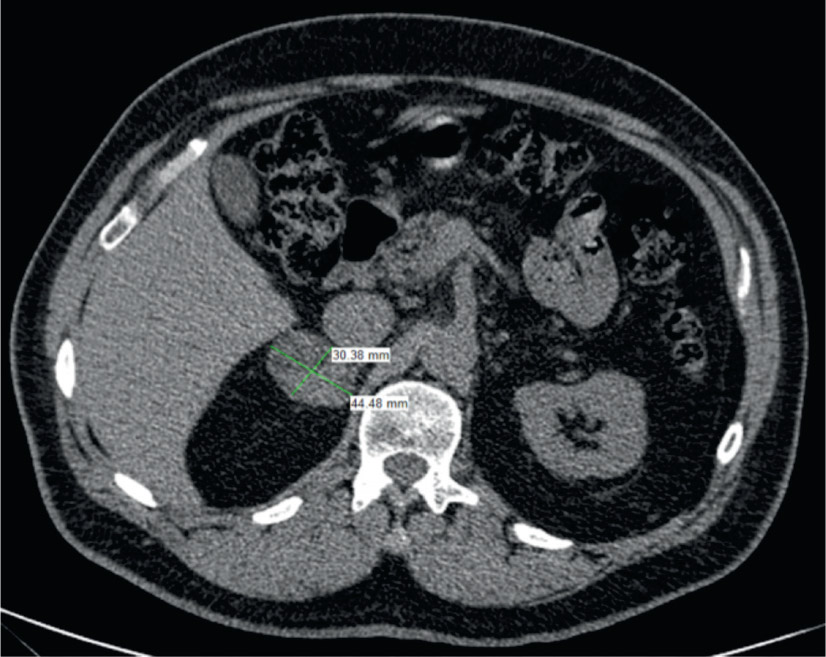
Figure 2. Adrenal adenoma
Non-contrast CT demonstrating 30 x 44 mm right adrenal lesion with well-defined borders and CT attenuation of 34 HU and delayed contrast-enhanced CT attenuation is 56 HU. Despite the lack of contrast washout, the lesion was found to be a lipid-poor adrenal adenoma
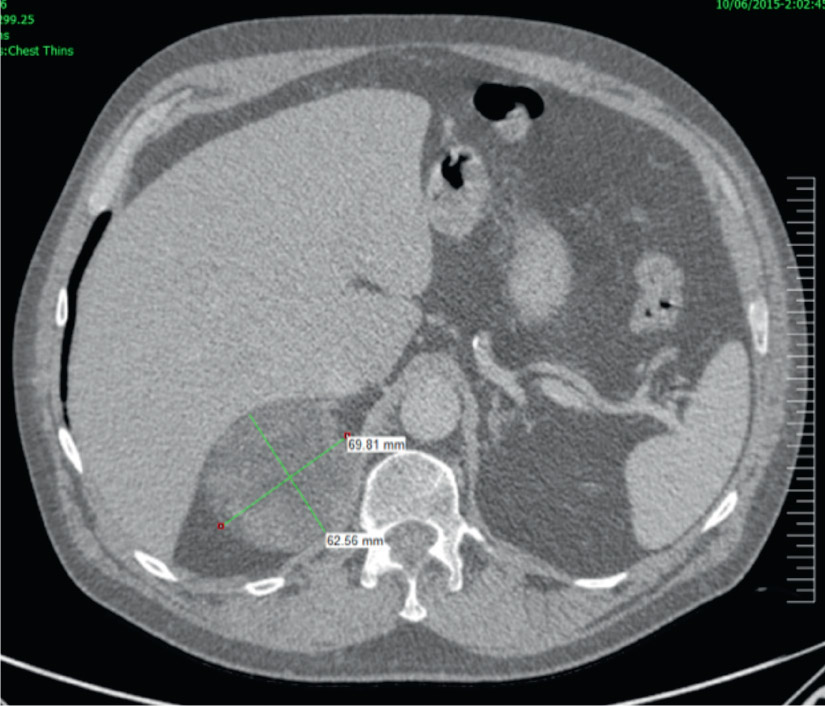
Figure 3. Adrenocortical carcinoma
Contrast CT demonstrating a large 69 x 62 mm heterogenous right adrenal lesion with
well-defined borders and CT attenuation of –33 HU. This lesion had increased in size >5 mm over six months. It was found to be an adrenocortical carcinoma
Size of mass
The risk of malignancy within an incidental adrenal mass increases with size: 2–5% of adrenal lesions <4 cm, 6–10% of tumours between 4 and 6 cm, and 25% of tumours >6 cm have been found to be malignant.4,9 Therefore, non-functioning adenomas <4 cm with benign imaging characteristics can be safely monitored (Figure 1).7
Imaging characteristics
Imaging phenotypes other than size can suggest malignancy. Irregular margins, heterogeneity in density and increased growth rate, although non-specific, are suspicious features for malignancy.15 Further radiological evaluation with pre‑contrast and post-contrast, delayed-phase CT imaging can help in characterising adrenal lesions as likely to be benign or malignant. Approximately 70% of adrenal adenomas have a high intracytoplasmic lipid content and, therefore, have a readily distinguishable phenotype as a low‑attenuation (<10 Hounsfield units) lesion on non‑contrast CT.15,16 Delayed contrast‑enhanced phase CT scans are performed to further distinguish between those 30% of benign tumours with low lipid content and malignant adrenal lesions.15,16 Both benign adenomas and carcinomas enhance rapidly on contrast imaging; however, benign adenomas tend to have a brisk washout of contrast when compared with carcinomas. It has been shown with 100% sensitivity and 98% specificity that lesions with an absolute washout of >50% of contrast medium at 10 minutes are benign adenomas as opposed to pheochromocytomas, carcinomas and metastases.9,17,18
MRI has also been used to further characterise incidental adrenal masses with comparable sensitivity and specificity to unenhanced CT in identifying lipid-rich adrenal adenomas.15 MRI is not as effective as CT washout in characterising lipid-poor adrenal lesions and, for incidental lesions measuring >30 Hounsfield units, CT adrenal protocol with washout assessment should be preferentially used.10,16
Regardless of the imaging modality used, patients with suspicious imaging characteristics should be considered for urgent surgical referral.17,19,20
Metastatic disease
Overall, 3% of incidentally identified adrenal masses are metastatic lesions; however, in patients with a previously diagnosed malignancy, 50–75% are metasteses.2,9 Metastases to the adrenal are frequently bilateral and >3 cm. A CT-guided fine-needle aspiration biopsy can be considered for patients with suspected metastases once pheochromocytoma has been biochemically excluded, to avoid a potentially fatal hypertensive crisis. However, if metastatic disease is thought to be unlikely, a biopsy is not recommended because fine-needle aspiration may not accurately distinguish between benign adenoma and adrenocortical carcinoma.2,7,9,10
Adenomas that secrete sex hormones are very rare and, therefore, routine testing for excess is not warranted in asymptomatic patients.2,4,9
Management
The two factors that affect management decisions for adrenal incidentalomas are the functional status of the lesion and malignant potential. All patients with an adrenal lesion who have signs, symptoms and biochemical evidence of glucocorticoid, sex hormone or catecholamine excess should undergo surgical intervention.4 Symptomatic patients with mineralocorticoid excess should be considered for surgical management if medical management with mineralocorticoid receptor antagonists is unsuitable (eg poor compliance). A laparoscopic adrenalectomy is the procedure of choice except in invasive adrenocortical carcinoma, where open adrenalectomy and lymphadenectomy is the standard of care.7
It is generally accepted that the clinically silent pheochromocytomas should be surgically resected, but management of a patient who is asymptomatic but has biochemical evidence of cortisol or aldosterone excess requires further consideration. While there is no current consensus, surgical intervention is generally recommended for young patients (aged <40 years) who can be spared the deleterious, long-term effects of subclinical hormone excess. However, conservative approaches are suitable for those patients who are not ideal surgical candidates.7,9,10 Ongoing clinical evaluation for the early detection of symptomatic hormonal derangements should be undertaken in the follow-up period such that treatment can be redirected to the surgical pathway if necessary.
If a lesion of any size has radiological features that are suggestive of malignancy, surgical resection is indicated. Although there is no consensus, it is broadly recommended that tumours >4 cm are resected because of the increased risk of adrenocortical carcinoma.7,10
In patients with non-functional tumours that are <4 cm, follow-up metabolic evaluation is recommended annually for five years because there is a risk of subsequent hyperfunction, which has been documented to be 17%, 29% and 47% at one, two and five years respectively.7,10 Repeat radiological imaging with an adrenal protocol CT is recommended at three to six months, then annually for one to two years to assess the growth rate. Should the tumour grow 0.5–1 cm during the follow‑up period, then adrenalectomy should be considered.7,9,10,21 Patients who remain stable on radiological imaging at two years and hormonal evaluation at five years may be discharged from follow-up.7,10 An algorithm to approach incidental adrenal masses is included in Figure 1.
Key points
- Adrenal incidentalomas are adrenal lesions ≥1 cm, identified serendipitously on cross-sectional imaging performed for unrelated reasons.
- Up to 15% of patients with pheochromocytomas are normotensive.
- In hyperaldosteronism, the serum potassium level may be normal and is not recommended as a screening test.
- Adenomas that secrete sex hormones are very rare and routine testing for excess is not recommended unless symptomatic.
- MRI is not as effective as CT in characterising lipid-poor adrenal adenomas.
- In asymptomatic patients, annual metabolic follow-up for five years is recommended.
- Repeat radiological imaging is recommended at three to six months, then annually for one to two years to assess growth rate.
Authors
Rasha Gendy, MBBS (Hons), MS (Urol), Accredited Registrar, Department of Urology, Port Macquarie Base Hospital; and Conjoint Lecturer, the University of New South Wales, NSW. rasha-gendy@hotmail.com
Prem Rashid, MBBS, FRACGP, FRACS (Urol), PhD, Urological Surgeon, Port Macquarie Base Hospital; and Conjoint Associate Professor, Rural Clinical School, the University of New South Wales, NSW
Competing interest: None.
Provenance and peer review: Not commissioned, externally peer reviewed.