How does the test work?
Regional cerebral blood flow (rCBF) is closely linked to regional cerebral metabolism under conditions of normal brain autoregulation. Cerebral perfusion studies with single photon emission computed tomography (SPECT) image the distribution of one or other of the technetium-99m labelled compounds, Tc-99m-hexamethylpropylene amine oxime (HMPAO) or Tc-99m-ethinyl cysteinate dimer (ECD).
HMPAO and ECD are both lipophilic agents that passively cross the blood-brain barrier, but are then trapped within neural tissue with high avidity. In a normal cerebral perfusion scan (Figure 1), there is up to a fourfold higher radiotracer uptake in the cortical grey matter relative to the white matter.1 Activity is also high in the subcortical grey matter, such as in the basal ganglia, and is highest in the cerebellum. The Society of Nuclear Medicine Brain Imaging Council has developed a publicly available database which provides a quantitative atlas of normal rCBF maps using these agents, recognising that there are minor differences in the distributions of HMPAO and ECD.2
Positron emission tomography (PET) scanning is another type of imaging study, which instead uses radiolabelled glucose as a marker. As neural tissue uses glucose as its fuel, regional cerebral metabolic rate of glucose can be imaged directly using fluorine-18 fluorodeoxyglucose (F-18 FDG). Although FDG-PET has superior resolution to SPECT, its availability in Australia is currently limited. In the future, PET imaging of b-amyloid protein may become available to directly visualise the pathological process of dementia of the Alzheimer type (DAT).
With newer devices, a low dose, non-contrast computed tomography (CT) is acquired in conjunction with the SPECT study to produce a fused SPECT/CT image that permits precise coregistration of rCBF with regional cerebral anatomy. This low dose CT scan is not a substitute for diagnostic quality CT or magnetic resonance imaging (MRI).
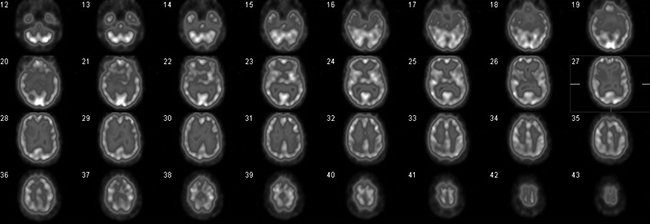
Figure 1. Normal cerebral perfusion, transaxial slices. Radiotracer uptake is consistent (and symmetrical) throughout the cortical and subcortical grey matter
When might it be helpful?
The most common use of cerebral perfusion studies is in the differential diagnosis of suspected dementia in patients presenting with cognitive impairment. Certain perfusion patterns are characteristic of different types of dementia (Figures 2–4). Broadly speaking, these patterns can be categorised as posterior (temporoparietal), frontotemporal or vascular. Recognising these differences can influence the management strategy as each entails a different treatment approach.
The posterior dementias include DAT, dementia with Lewy bodies (DLB) and dementia of Parkinson disease. Subtle differences can refine the diagnosis further. DLB, for example, might be differentiated from DAT by more involvement (manifest as hypoperfusion) of the occipital cortex.
It is most useful in patients aged less than 70 years, but may be less useful in older patients as at times the effects of atrophy and multiple disease processes can make the patterns of change less distinct. Diagnostic accuracy has been improved by FDG-PET and may be preferred in some centres, but it is less widely available and is not Medicare reimbursed for dementia.
A much less common use of cerebral perfusion studies is to localise the epileptogenic focus in patients with intractable or medically refractory epilepsy for whom surgery is contemplated. It is an adjunct to electroencephalography (EEG) and MRI in preprocedural planning. The procedure is technically demanding as optimal localisation of the focus requires the cerebral perfusion agent to be injected during a seizure (proven by continuous EEG and video monitoring): because radiotracer distribution in the brain is stable for several hours after injection, image acquisition can then be delayed until the patient has been stabilised.
Cerebrovascular disease is another approved use of cerebral perfusion studies. Parts of the brain affected by stroke will demonstrate reduced tracer uptake long before the development of anatomical changes on CT. However, the narrow time window available for the effective management of stroke with thrombolytic agents prohibits the use of cerebral perfusion studies in the acute setting.
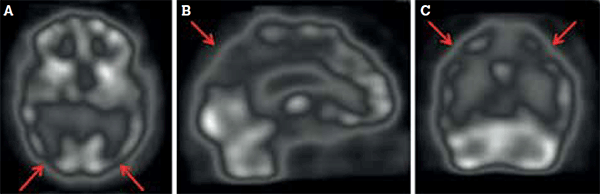
Figure 2. Dementia of the Alzheimer type is characterised by bilateral parietotemporal hypoperfusion (arrows), evident in (A) transaxial, (B) sagittal and (C) coronal views
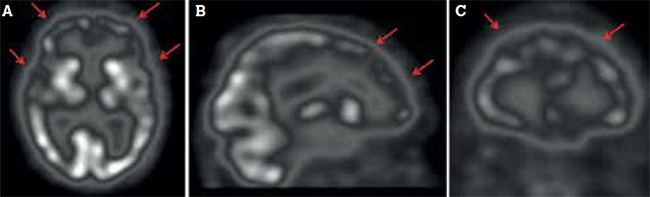
Figure 3. Frontal and temporal hypoperfusion (arrows), paralleling neuronal degeneration in frontotemporal dementia, shown in (A) transaxial, (B) sagittal and (C) coronal views
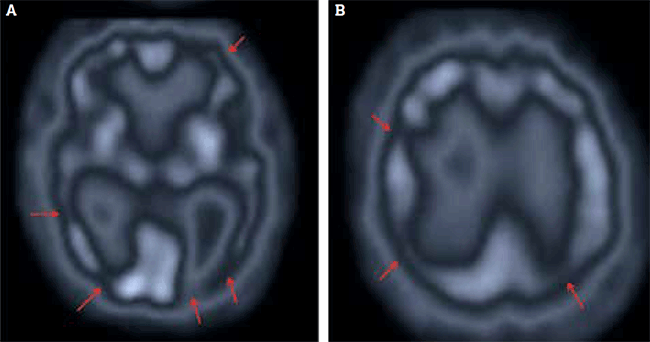
Figure 4. Cerebral perfusion studies in multi-infarct dementia typically show bilateral scattered areas of hypoperfusion throughout the cortex (arrows) in (A) inferior and (B) superior sections
Are there any contraindications?
Apart from a definite previous reaction to the radiotracer (a very rare event), there are no absolute contraindications. Claustrophobia, pregnancy and the uncooperative patient are relative contraindications. As the radiotracers are not nephrotoxic, renal impairment is not a contraindication. Interruption of breastfeeding is not required following this investigation.3
What is the radiation dose?
Radiation dose is measured in units of millisieverts (mSv). For typical activities of Tc-99m compounds used in cerebral perfusion scans, the effective radiation dose (an estimate of the dose to the whole body) averages 9.4 mSv.2 In Table 1, this is compared to the background radiation level in Australia (average annual exposure with usual activities) and to the effective doses from some other commonly performed diagnostic imaging tests.
Table 1. Radiation dose for a typical cerebral perfusion study and other common diagnostic imaging tests
| Test/situation | Total effective dose (mSv) |
|---|
| Chest X-ray4 |
0.1 |
| Annual Australian background5 |
1.5–2.0 |
| Bone scan1 |
6.0 |
| Cerebral perfusion study2 |
9.4 |
| CT pulmonary angiogram6 |
13.7 |
| CT abdomen + pelvis4 |
15.0 |
What does the patient need to know (before the test)?
No special requirements – in terms of diet, change of medications – are required in preparation for a cerebral perfusion study.
Scanning commences approximately 60 minutes following administration of the radiotracer. For at least the first 20 minutes, the patient will be resting in a quiet, dimly lit room. The patient should not speak or read during this time, as it will activate the corresponding cortical area, which will be overrepresented in the images.
During imaging, the head is lightly restrained to minimise motion. The whole process (injection and imaging) may take up to 2 hours. Patients are safe to travel home after the scan has been completed, as anaesthesia or sedation are not required for the procedure.
The radiation dose is so low as not to affect bystanders.
Case study
A man, 68 years of age, with treated hypertension, osteoarthritis and mild renal impairment was persuaded by his wife to see his general practitioner about changes in his short-term memory. On further questioning, she described personality changes (irritability and apathy) and said her husband could no longer manage the family finances. His Mini-Mental State Examination (MMSE) score was 22/30. The remainder of history and examination was normal. Specifically, he did not have a past history of stroke or recent visual symptoms.
A CT scan of the brain and blood tests were unremarkable. It was difficult clinically to distinguish between DAT and frontotemporal dementia, so a cerebral perfusion study was requested.
The study (Figure 5) showed bilateral parietotemporal hypoperfusion, consistent with early DAT. Confirmation of DAT enabled the patient to commence treatment with an anticholinesterase inhibitor. Other medications were unchanged. His MMSE score was 24/30 6 months later and his wife reported some improvement in his symptoms of irritability and apathy.
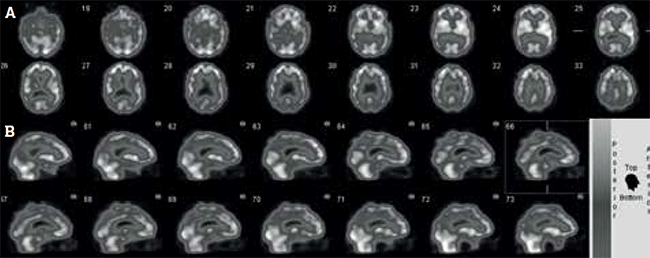
Figure 5. Cerebral perfusion study of patient with worsening short-term memory and mild cognitive impairment, shown in (A) transaxial and (B) sagittal views
Competing interests: None.
Provenance and peer review: Commissioned; externally peer reviewed.