There is a range of conditions that can cause both macroscopic and microscopic haematuria. In Australia, microscopic haematuria is defined as >10 red blood cells in high field power on microscopic evaluation. This varies from >3–10 cells depending on British, European or American definitions. The prevalence of microscopic haematuria is 0.19–21.0%.1,2
Macroscopic haematuria is more concerning and warrants thorough investigation, as the prevalence of urinary tract carcinomas among patients with macroscopic haematuria has been reported to be as high as 19%, but usually ranges from 3–6%.1
One of the most common causes of haematuria is urinary tract infection, which can be investigated with a urine microscopy, culture and sensitivity (MC&S). Ureteric and renal stones are another common cause, but these typically present with pain and microscopic haematuria. In cases where macroscopic haematuria or risk factors are present, or if another cause cannot be determined, more extensive investigations to rule out underlying malignancy are recommended.
The prevalence of haematuria is no higher in patients who are anticoagulated within the therapeutic range, therefore the same investigations as those performed in patients who are not anticoagulated apply.3,4 Despite extensive investigation, studies have shown that up to 50% of patients with macroscopic haematuria and 70% with microscopic haematuria have no identifiable cause found.5 This could also be attributed to transient benign physiological conditions, including vigorous physical exercise, sexual intercourse or menstrual contamination. Common causes of haematuria are outlined in Table 1.
Table 1. Common causes of haematuria2,7,8
| Category | Cause |
|---|
| Benign |
- Renal masses (eg. angiomyolipoma, oncocytoma)
- Benign prostatic hypertrophy
- Strictures
|
| Stones |
- Staghorn calculi
- Calcium stones
- Uric acid stones
|
| Infective |
- Pyelonephritis
- Cystitis
- Urethritis
|
| Trauma |
- Pelvic trauma
- Renal injuries
- Foreign bodies
|
| Renal |
- IgA nephropathy
- Thin basement membrane diseases
- Hereditary nephritis
- Medullary sponge kidney
|
| Iatrogenic |
- Recent endoscopic procedure (eg. transurethral resection of prostate [TURP])
- Transrectal ultrasound (TRUS) guided prostate biopsy
- Traumatic catheterisation
- Radiation
- Indwelling ureteric stents
- Renal biopsies
- Extracorporeal shockwave lithotripsy
|
| Malignant |
- Renal cell carcinoma
- Transitional cell carcinoma
- Squamous cell carcinoma
- Urothelial cell carcinoma
- Prostate acinar adenocarcimona
|
Risk factors for haematuria
Risk factors can help in determining which patients are at higher risk of urinary tract and bladder cancer. Risk factors include:2,6
- age >40 years
- a history of smoking
- history of gross haematuria
- history of chronic cystitis or irritative lower urinary tract symptoms (eg. frequency, urgency, dysuria, nocturia, hesitancy, sensation of incomplete emptying)
- pelvic irradiation
- exposure to occupational chemical and dyes (eg. heavy phenacetin use, treatment with high doses of cyclophosphamide, aristolochic acid).
Investigations
There are a number of recommended investigations in the assessment of haematuria, including urine sampling, blood tests and imaging (Figure 1).
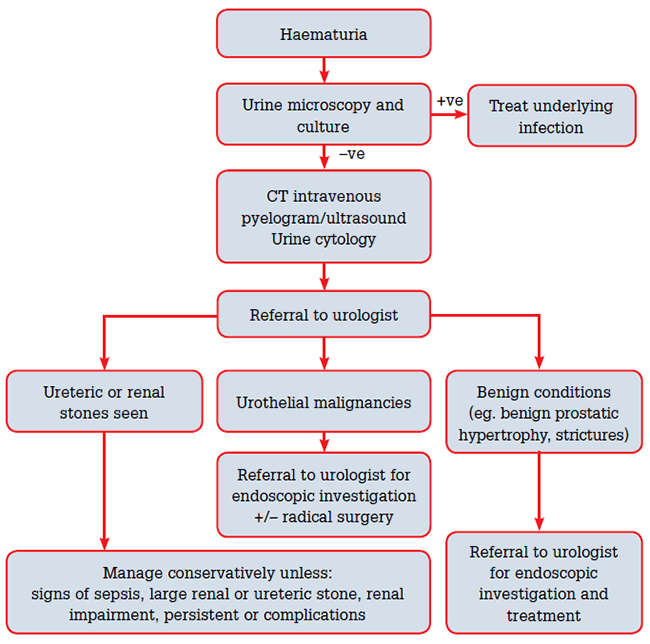
Figure 1. An approach to the investigation and initial management of macroscopic haematuria3,7,13,17
Urine dipstick
A urine dipstick can give instant results and tests for haemoglobin. It relies on Hb to catalyse the oxidation of a chromagen by organic hydrogen peroxide. Unfortunately, myoglobin, bacterial peroxidases, povidone and hypochlorite can also cause this reaction, which can result in false positives.5 Sensitivity ranges from 91–100%, specificity 65–99%, and false negatives unusual.7,8 It is advised that if a urine dipstick is positive for blood, the urine should be sent to pathology for formal testing with microscopy.9
Urine MC&S
Urine MC&S is an important investigation for the assessment of haematuria. Urinary tract infections are one of the most common causes of haematuria, and this should be ruled out before an episode of haematuria is investigated further.7 Samples should be sent to pathology and examined promptly, as delay can cause red cell lysis. Red cell morphology can also be useful in determining whether the source is glomerular or urinary tract,7 and can therefore minimise unnecessary urinary tract imaging. A urine albumin:creatinine ratio assesses albumin excretion and may be useful in cases where it is uncertain if the cause for renal impairment is renal or post-renal.10
Urine cytology
Urine cytology can be useful in the investigation of bladder and upper tract carcinomas. It has the greatest sensitivity with bladder carcinoma (specifically high grade and carcinoma in situ) but is less sensitive for upper tract transitional cell carcinomas.2,11,12 Whether the result is normal or abnormal, atypical cytology should be followed up with a cystoscopy. Cytology is useful as an adjunct to cystoscopy and can be used for the ongoing evaluation of asymptomatic microscopic haematuria post-cystoscopy and for follow up of high grade urothelial cancer and carcinoma in situ. Studies have shown the sensitivity of cystoscopy for bladder tumours is between 66% and 79%, with the specificity between 95% and 100%.8 A normal urine cytology result does not exclude malignancy. Sensitivity of urine cytology can be increased by testing mid morning or random specimens from three separate voids.13
Blood tests
Useful blood tests in the evaluation of haematuria include:2
- a full blood count can be used to assess the evidence of infection or level of blood loss
- renal function (including creatinine and estimated glomerular filtration rate [eGFR]) is important to ensure that there is no impairment of renal function
- in men, a prostate-specific antigen (PSA) test may be useful, as prostate cancer is a differential diagnosis. PSA levels can also be raised in urinary tract infections, urinary retention and benign prostatic hypertrophy.
Imaging
X-ray kidneys, ureters, bladder
X-ray of the kidneys, ureters, bladder (XR-KUB) is useful in identifying and monitoring radio opaque ureteric and renal stones, but is not useful in assessing the urinary tract. Importantly, conventional X-rays only have a sensitivity of approximately 60% in detecting renal and ureteric calculi.1
Intravenous pyelogram
Intravenous pyelogram (IVP) involves the injection of contrast with multiple XR-KUBs taken to capture the travel of contrast through the urinary tract (as contrast is renally excreted). Sensitivity for detecting urinary tract lesions is 50–60.5% with a specificity of 90.9%.14,15 IVP has been largely superseded by computed tomography (CT) evaluation.1
Ultrasound
Ultrasound can be helpful in the characterisation of renal tumours, cystic lesions and the pelvicalyceal system looking specifically for hydronephrosis and moderately large bladder lesions such as stones or tumours. It also allows for general but limited assessment of the prostate gland and the effects of bladder emptying. Ultrasound usually requires follow up with a corresponding CT scan.1 Results are operator dependant. Ultrasound has a 40% sensitivity for identifying renal tumours, but this can range from 0–57% depending on the size of the lesion.8 Ultrasound can also be useful in the investigation of haematuria in pregnant women, children and low risk patients, and does not have the risk of radiation or contrast reaction. Ultrasound has a lower diagnostic yield and is less sensitive for ureteric and bladder tumours compared to CT. Therefore it is the second investigative modality of choice if CT is unavailable.
CT kidney, ureters, bladder
Computed tomography of the kidneys, ureters, bladder (CT-KUB) is a non-contrast study – the current gold standard in identifying ureteric and renal stones, with a 94–98% sensitivity.2 However, it has a much lower sensitivity in identifying genitourinary malignancies and lesions.7
CT intravenous pyelogram
Computed tomography IVP (CT-IVP), also known as a CT urogram, is the preferred modality of imaging the renal tract for the detection of genitourinary malignancy and disease.1,2,5 It combines the benefits of CT-KUB with an IVP and can visualise the collecting system, ureteric and pelvicalyceal surfaces. Sensitivity for detecting pathology in patients with haematuria varies from 94.1–100%, with a 97.4% specificity.14,15
Cystoscopy
Cystoscopy visualises the entire bladder and is an effective way to diagnose urethral and bladder pathology and requires referral to a urologist (Figure 2). Flexible cystoscopy can be performed under local anaesthetic. Tissue can be biopsied for further analysis during the procedure, although inadequate tissue sampling is a common problem. Rigid cystoscopy is performed under a general anaesthetic and is typically indicated for lesions seen on imaging or flexible cystoscopy, and in cases where a biopsy or tumour resection is anticipated. It can also identify strictures or a source of bleeding from the bladder wall, prostate and ureters. It is recommended in patients with haematuria for those aged more than 40 years, or who have risk factors for urinary tract or bladder cancer.2
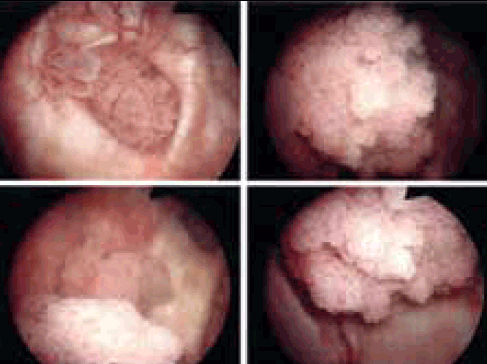
Figure 2. Endoscopic view of bladder tumours
Image courtesy Western & Central Melbourne Integrated Cancer Service
Initial management
Most episodes of haematuria will settle conservatively with no acute intervention required. A smaller percentage with more significant haematuria (including a high risk of clot retention, haemodynamic instability, a drop in haemoglobin, or cardiovascular symptoms) will require hospital admission for continuous bladder irrigation and management of symptomatic anaemia. The most common acute emergency presentation of haematuria is acute urinary retention secondary to blood clots (clot retention). This typically presents with acute abdominal pain and an inability to urinate. In these cases, patients will also need catheter insertion and continuous irrigation. Blood transfusions may also be required. In severe cases, patients may require emergency cystoscopy and diathermy if they are not responding to conservative management.16 It is also important to determine and treat the underlying cause.
Urinary tract infection
Any urinary tract infection should be treated empirically, with treatment modified if needed based on sensitivities. Once treated, repeat urine MC&S are necessary to ensure both the infection and the haematuria have resolved.
Ureteric and renal stones
Ureteric and renal stones can initially be conservatively managed. Ninety-eight percent of stones <5 mm in diameter are likely to pass spontaneously.17 This percentage decreases as the stone diameter increases. Non-steroidal anti-inflammatories are the most useful treatment for renal colic, however, stronger (opiate) analgesia is often required. The use of alpha-blockers, such as prazosin and tamsulosin, are also useful in the passage of distal ureteric stones.18 Stones may require urological referral if they are unlikely to pass, causing acute renal impairment, sepsis or a history of recurrent stones and infection.
Renal, ureteric and bladder tumours
A referral to a urological service is advised if there is evidence of genitourinary lesions on a CT-IVP or atypical cytology. Renal cancers are the fifth most common cancer in Australia, making up 2.4% of all cancers. Bladder cancer is the seventh most common cancer in Australia. Early stage, low grade disease can be treated with cystoscopy and the transurethral removal of tumours.19
Prostatomegaly
Alpha-blockers (eg. prazosin, tamsulosin) have been shown to significantly improve the symptoms of benign prostastic hypertrophy and are considered first line treatment.20 5-alpha-reductase inhibitors (eg. finasteride, dutasteride) are second line treatment for lower urinary tract symptoms and have been shown to reduce symptoms as well as decrease the risk of acute urinary retention and the need for invasive surgery. Long term combination therapy has been shown to be the most effective in reducing clinical progression of benign prostatic hypertrophy.21 5-alpha-reductase inhibitors can also be beneficial in the management of prostate-related haematuria by reducing the vascularity of the prostate. The mechanism of action for this is uncertain.22,23 5-alpha-reductase inhibitors should however, only be commenced under the recommendation of a urologist.
A less common cause of haematuria is prostate cancer. This should be investigated with PSA, a digital rectal examination and, if abnormal, referral to a urologist for transrectal ultrasound (TRUS) guided prostate biopsy.
Key points
- Urinary tract infection should be ruled out before any further investigations for haematuria are undertaken.
- Ureteric and renal stones can cause episodes of haematuria, however it is important to consider other causes if this does not settle or if there are risk factors for urinary tract malignancy.
- Initial investigations for haematuria should include CT-IVP, urine cytology, full blood examination, renal function, and PSA in men.
- A urological referral is recommended in patients presenting with macroscopic haematuria, persistent microscopic haematuria, abnormal urine cytology, irritative lower urinary tract symptoms or recurrent urinary tract infections.
Competing interests: None.
Provenance and peer review: Not commissioned; externally peer reviewed.