Epidemiology
The prevalence of NAFLD is estimated to be approximately 30% of adults in developed countries such as Australia and the United States, depending on definition and detection methods.3 However, NAFLD is also becoming increasingly common in Asia (countries previously thought to be at low risk of NAFLD), where a prevalence of up to 15% has been reported in China.4
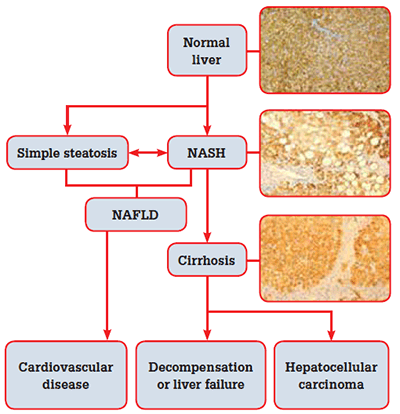
Figure 1. The natural history of non-alcoholic fatty liver disease
Natural history
Simple steatosis appears to be a relatively benign condition, although it may progress to NASH over time. Cardiovascular disease is the major cause of death in patients with NAFLD;5 NAFLD alone is associated with a slightly higher overall mortality. However, when NAFLD occurs in the presence of other features of the metabolic syndrome (MetSy), mortality doubles.6 The MetSy is a clustering of cardiovascular risk factors related to reduced insulin sensitivity. Diagnostic criteria for the syndrome include:7
- elevated serum triglycerides (TG)
- lowered serum high-density lipoprotein cholesterol (HDL-C)
- impaired glucose tolerance
- central adiposity
- hypertension.
Although NAFLD is not officially included in the definition of the MetSy, it commonly co-exists with features of this syndrome.
In addition to its association with cardiovascular complications, NAFLD can lead to liver related morbidity and mortality. The risk of developing cirrhosis is higher in the presence of NASH, which is more likely in the presence of the following features:
- type 2 diabetes mellitus (T2DM)
- obesity (body mass index [BMI] >30 kg/m2)
- age more than 50 years
- serum aminotransferases (ALT or AST) more than two times the upper limit of normal.
Non-alcoholic steato-hepatitis cirrhosis probably accounts for the vast majority of what was previously described as ‘cryptogenic cirrhosis’. In one series, 70% of patients with cryptogenic cirrhosis had risk factors for NAFLD.8 In the US, NASH is an increasingly common indication for liver transplantation.9,10 In the setting of cirrhosis from NASH, the risk of developing hepatocellular carcinoma (HCC) is between 2.4% over 7 years, and 12.8% over 3 years,11 compared to 21% over 10 years for hepatitis C cirrhosis.12 Hepatocellular carcinoma has also been reported in NASH without cirrhosis, particularly in association with the MetSy.13
Identifying NAFLD and NASH in general practice
The most common presentation of NAFLD will be incidental finding of abnormal LFTs. Typical findings in NAFLD are raised ALT and AST, with a preserved ALT: AST ratio of 1.5, raised gamma glutamyl transferase (GGT) and, occasionally, raised alkaline phosphatase (ALP). These findings commonly occur in the setting of features of the MetSy.
There are several features on examination and laboratory values that should raise suspicion of cirrhosis, such as spider naevi, low or falling platelets, low albumin or reversal in ALT:AST ratio (where AST exceeds ALT), before the features of portal hypertension and decompensation become obvious.
Apart from excluding excess alcohol consumption as a potential cause, there are several other important causes of raised ALT that need to be excluded, such as viral hepatitis, autoimmune hepatitis, haemochromatosis, thyroid disease and coeliac disease (Table 1).
Table 1. What to consider and how to exclude differential diagnoses
| Consideration | Test |
|---|
| Excess alcohol consumption |
Careful, corroborative history |
| Chronic hepatitis B |
HBsAg, anti-HBs, anti-HBc |
| Chronic hepatitis C |
Hepatitis C antibody |
| Autoimmune hepatitis |
Anti-nuclear antibody (ANA)
Anti-smooth-muscle antibody (ASMA)
Anti-mitochondrial antibody (AMA) |
| Haemochromatosis |
Iron studies |
| Thyroid disease |
Thyroid function tests |
| Coeliac disease |
Coeliac antibodies (usually deamidated antigliadin antibody, tissue transglutaminase, total lgA level) |
| Medications |
History of amiodarone, anticonvulsants, methotrexate, tamoxifen, synthetic oestrogens, corticosteroids, HIV therapy, perhexiline |
Diagnostic assessment of NAFLD
A definitive diagnosis of NAFLD depends on three factors:
- evidence of fatty infiltration from either imaging (ultrasound, magnetic resonance imaging [MRI]) or histology (liver biopsy)
- exclusion of significant alcohol consumption
- exclusion of other causes of hepatic steatosis (eg. medications, surgery, metabolic disorders).
Confirming hepatic fatty infiltration using ultrasound is important. Specificity is high (95%), but the sensitivity of ultrasound for detecting fatty infiltration is lower (85%).14 Ultrasound is also useful to look for signs of cirrhosis, such as irregular liver edge, but has a sensitivity of only 43–74% (specificity is slightly higher at 54–89%).15 Signs of cirrhotic complications are also important, eg. signs of portal hypertension (splenomegaly, increased portal vein size, varices) or other complications such as HCC, portal vein thrombosis, or ascites.
The risk of fibrosis and progressive liver disease in NAFLD increases with severity of insulin resistance. In the absence of simple available clinical measurements of insulin resistance, the number of MetSy features present can be used to estimate risk of insulin resistance. The presence of three or more features of the syndrome, especially if these include central adiposity and T2DM, are predictive of the presence of NASH rather than simple steatosis. In addition, family history plays a role: an individual with a first degree relative with T2DM has a 90% chance of developing T2DM, and therefore NASH.16 Central adiposity can be assessed using waist circumference measured at the narrowest point mid-way between the lowest rib and the iliac crest at the end of expiration with the patient standing.
Staging liver disease and detecting cirrhosis is the most important aspect of assessing fatty liver disease. However, it is also difficult and error prone. The traditional gold standard in assessing liver disease is liver biopsy. However, biopsy has unfavourable cost, safety, availability, sampling error, inter-observer variability and patient acceptance. Liver biopsy may be considered where cirrhosis is suspected, or where an alternative diagnosis is considered. At this stage, referral to a gastroenterologist is also suggested.
Non-invasive tools for estimating the degree of fibrosis include transient elastography (FibroScan®), acoustic radiation force impulse (ARFI), and non-invasive biomarker algorithms such as NAFLD Fibrosis Score, FibroTest and Hepascore. However, their use in clinical practice is still being evaluated.
Management of NAFLD
Cardiovascular risk factors and lifestyle modification
The cornerstone to managing NAFLD is achieving weight control and reduction in cardiovascular risk factors such as smoking, diabetes, hypertension and dyslipidaemia. This may include referral to a dietician, endocrinologist or cardiologist. Treatment of associated dyslipidaemia is appropriate with either HMgCo-A reductase inhibitors (‘statins’) or fibrates where required. Moderate elevations in liver enzymes due to the use of statins should be tolerated and treatment continued.17 However, severe elevations more than 10 times the upper limit of normal should prompt cessation of the medication and reassessment.
Dietary manipulation, such as the adoption of a Mediterranean-type diet, has shown promise. The most important feature appears to be caloric reduction. Where diet and exercise are unsuccessful in achieving weight reduction, bariatric surgery may be considered.
Novel treatments
Various therapeutic agents have shown some promise in the management of NAFLD.18–22 However, weight loss remains the only therapy with proven benefit and safety.23–27
What follow up should be performed?
A pragmatic approach to the investigation and management of NAFLD is presented in Figure 2.28 Normalisation of LFTs associated with mild weight loss may be an encouraging sign that the disease activity is driven by NAFLD. However, it is important to stage the disease initially (with ultrasound and histology if appropriate). The presence of normal LFTs does not exclude underlying cirrhosis, nor does a ‘non-cirrhotic’ ultrasound. Monitoring full blood examination (FBE), LFTs, International Normalised Ratio (INR), blood pressure and lipid profile every 6 months is a reasonable approach.
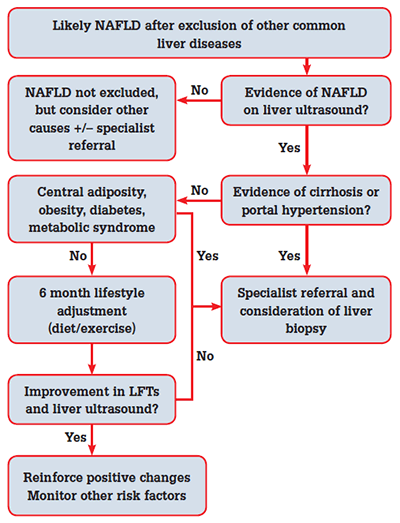
Figure 2. Algorithm for management of suspected NAFLD
When to refer for specialist management
Referral for specialist management should be undertaken whenever there is a suspicion of severe fibrosis or cirrhosis, whether at initial assessment or at any time during monitoring.
Key points
- Non-alcoholic fatty liver disease is increasingly common, and will have a significant impact on morbidity and mortality on a growing number of Australians.
- NAFLD should be considered when abnormal LFTs are found, particularly in the presence of features of the MetSy.
- Weight reduction via caloric restriction and regular exercise are important, and no medications can currently be recommended as specific therapy.
- General practitioners are vital in identifying patients at risk of NAFLD, and encouraging initiation and maintenance of appropriate lifestyle changes.
Further reading
Competing interests: None.
Provenance and peer review: Commissioned; externally peer reviewed.