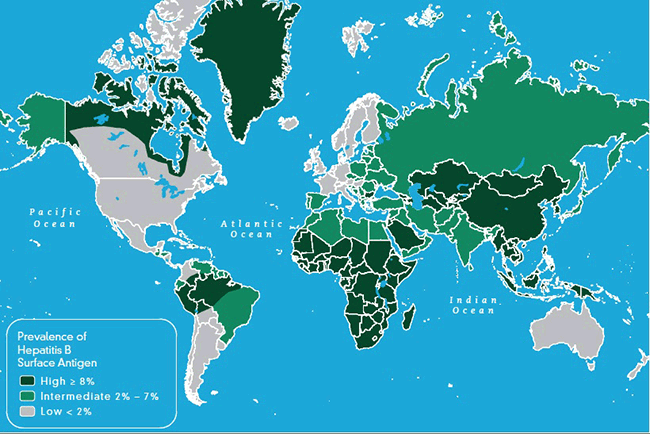
Figure 1. Global prevalence of chronic hepatitis B28
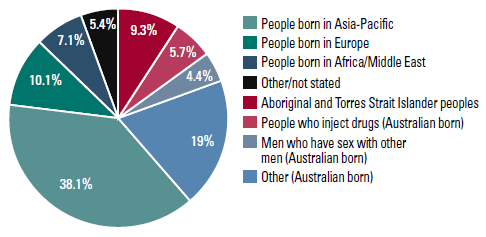
Figure 2. Distribution of Australia’s burden of chronic hepatitis B by priority population, 20111
In most cases, CHB infection has no specific symptoms, but leads to the development of liver disease such as cirrhosis and liver cancer in around 15–25% of cases if not adequately managed.4 The generally long lead-time before these outcomes reinforces the need for timely diagnosis and intervention.
What’s new?
The increased prevalence of CHB in Australia is primarily due to increased migration from endemic countries. This, along with the increase in people living with hepatitis C virus (HCV), has resulted in liver cancer being the fastest increasing cause of cancer death in Australia.5
Who should be tested for hepatitis B?
All of the at-risk groups outlined above should be opportunistically screened for hepatitis B virus (HBV) if their status is unknown, by ordering all three of the following serological tests:6
It is estimated that nearly half of all Australians living with CHB remain undiagnosed;1 this facilitates poor outcomes in those infected and ongoing transmission to susceptible contacts. Routine testing of people from high risk groups does not contravene the screening provisions in the Medicare Benefits Schedule (MBS), and is recommended in Australia’s National Hepatitis B Testing Policy.7
All patients about to undergo chemotherapy or other immunosuppressive treatment should also be screened for HBV, due to the risk of life threatening flares. People living with HIV or HCV are also at higher risk of HBV infection, and of more rapid progression to advanced liver disease if they have CHB.7
All pregnant women should be routinely tested for HBV as part of antenatal screening. Routine testing of all household and sexual contacts of anyone diagnosed with CHB is also necessary.
For further details regarding HBV testing see the article ‘Hepatitis B serology’ in the April 2012 issue of Australian Family Physician.6 Other useful resources are available from HepBHelp, the Australasian Society for HIV Medicine (ASHM) and the National Hepatitis B Testing Policy (see Resources).
What’s new?
Australia’s first National Hepatitis B Testing Policy outlines appropriate testing pathways and interpretation guidelines for serology, as well as information regarding pre- and post-test counselling and how to conduct HBV testing in a culturally sensitive and supportive way (see Resources).
How should patients with chronic hepatitis B be managed?
After diagnosing a patient with CHB, it is important to remember that many jurisdictions require the condition to be notified by both the testing laboratory and the treating doctor. It is also important to convey to the patient the test result, what it means and what needs to happen next. Emphasising the life-long nature of CHB, and the availability of treatment (if required), is also recommended.
Resources about living with CHB are available in a range of languages; some useful websites are listed in the Resources section at the end of this article.
Check for other infections
Infection with hepatitis A can cause a life threatening flare of liver disease in people with CHB, so it is important to test for immunity and offer vaccination if susceptible.8
Hepatitis C,9 hepatitis D (HDV),10 and HIV11 can all complicate the assessment, monitoring, management and treatment of people living with HBV. All patients diagnosed with CHB should be screened for these infections, and referral to a specialist should be considered in the setting of co-infection.
Identify, screen and vaccinate contacts
Hepatitis B is a vaccine preventable disease. Family members and household contacts, as well as sexual partners, should be screened (using the three serological tests previously outlined) and vaccinated if they are susceptible. Hepatitis B vaccine for contacts of a person with CHB is provided free of charge in many jurisdictions.
Establish the phase of chronic hepatitis B
An essential part of the post-diagnosis work-up of a patient with CHB is determining the phase of infection. The phase of infection is largely determined using HBV DNA levels, alanine aminotransferase (ALT) and HBeAg/anti-HBe status (Figure 3). The phase of infection indicates the likelihood of progressive liver damage, and is used to help decide which patients need further investigation (eg. liver biopsy) and for consideration of antiviral treatment. A simple schematic of the phases of chronic HBV infection, and when specific treatment is generally considered, is illustrated in Figure 3.12
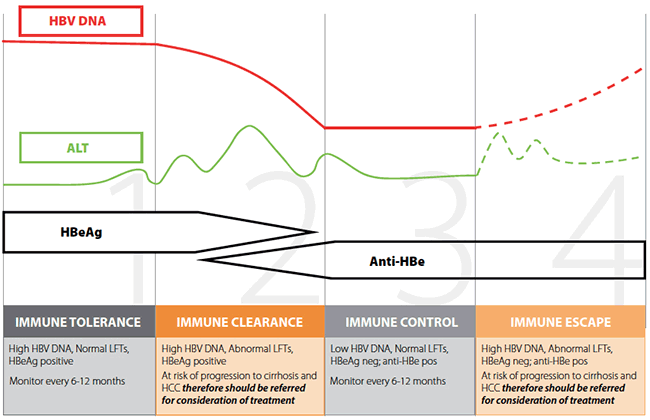
Figure 3. Phases of chronic hepatitis B infection12
Adapted from Decision-making in HBV. ASHM. Available atwww.ashm.org.au/resources
Arrange regular monitoring
All people diagnosed with CHB require regular monitoring. The frequency of this monitoring varies according to the phase of infection, the extent of liver damage present, whether the person is receiving treatment, and the presence of other complicating factors such as co-infections, immunosuppression and other causes of liver disease.
However, the minimum recommended interval for this monitoring is yearly, including clinical review, liver function tests (LFTs) and HBV DNA viral load (annual HBV DNA testing is funded by Medicare for all HBsAg positive patients).13 Other tests can be indicated, including HBeAg/anti-HBe status in phases 1 and 2, and hepatocellular carcinoma (HCC) surveillance in eligible patients.
These tests can be included in a patient’s chronic disease management plan, if applicable. A sample template for a chronic disease management plan for CHB has been developed by ASHM and is available at www.ashm.org.au/images/ hbv/hbv_gpmp.rtf. New evidence regarding the natural history and clinical management of CHB is emerging, and we now know there is no such thing as a ‘healthy carrier’.
Consideration should be given to auditing clinic records for patients previously diagnosed with CHB who have not been engaged in regular monitoring and ongoing management.
Hepatocellular carcinoma surveillance
Chronic hepatitis B is a leading cause of hepatocellular carcinoma in Australia.14 People who develop primary liver cancer are generally diagnosed late and have poor survival, with recent Victorian data indicating that most people diagnosed with HCC survive less than a year after diagnosis.15
Research from Australia16 and overseas17,18 indicate that those whose liver cancer is detected through screening have significantly improved survival, and that in combination with appropriate treatment for CHB, HCC surveillance is cost effective as a cancer prevention intervention.19
For these reasons, HCC surveillance with liver ultrasound (with or without serum alpha-fetoprotein [AFP] testing) is recommended for patients at increased risk20 (Table 2). This surveillance should be performed every 6 months.
Table 2. People living with CHB who are at high risk of HCC and recommended for 6 monthly surveillance20
- Those with cirrhosis
- Those with a family history of liver cancer
- Asian men aged over 40 years; Asian women aged over 50 years
- African men and women aged over 20 years
|
Who should be referred for treatment?
Determining a patient’s phase of CHB infection is crucial to determining the need for treatment. In general, those with active viral replication (indicated by elevated HBV viral load) and active liver damage (indicated by elevated ALT or inflammation on biopsy, or evidence of fibrosis on biopsy or non-invasive testing, eg. FibroScan®) should be considered for treatment. Appropriate further testing to perform before referral is outlined in Table 1.
Table 1. Post-diagnosis (or pre-referral) work-up for a person diagnosed with CHB22
| Test | Why the result is important |
|---|
HBeAg/Anti-HBe serology
HBV DNA viral load |
Quantify replication
Identify phase of infection
Suitability for treatment |
| HAV, HCV, HDV, + HIV serology |
Identify any co-infections
Identify need for hepatitis A vaccination |
LFTs, full blood examination (FBE), clotting studies
Abdominal ultrasound including portal venous Doppler, +/- alpha-fetoprotein |
Quantify inflammatory activity, synthetic function (low platelets could suggest cirrhosis)
To investigate for the presence of cirrhosis, HCC |
| All these investigations are rebatable by Medicare for patients diagnosed with hepatitis B |
Any patient with active liver disease or suspected cirrhosis should be referred for specialist assessment and consideration of therapy. Seeking expert advice is also recommended when CHB is complicated by other factors. For example, pregnant women may require therapy to prevent vertical transmission,21 and those co-infected with HIV, HCV and/or HDV require special treatment approaches.22 Patients undergoing immunosuppressive therapy should be referred for consideration of pre-emptive treatment to avert a flare of CHB during therapy.
What’s new?
Since November 2011, liver biopsy is no longer mandatory in order to be eligible for Pharmaceutical Benefits Scheme funded HBV treatment. New, non-invasive methods of establishing the degree of hepatic fibrosis (eg. FibroScan®) are becoming increasingly available in hospital hepatitis clinics and some community settings.
How successful is treatment?
The short term goals of therapy are suppressing HBV viral load and reducing liver inflammation (reflected by the ALT level), and these are the key indicators used to monitor response.23
For most patients, antiviral therapy for HBV is effective at reducing the risk of progressive liver damage, cirrhosis and liver cancer. Recent evidence suggests that just 4 years of treatment reduces the risk of cancer by more than half, and reversal of cirrhosis is increasingly recognised.24–26 The first-line oral antiviral therapies (entecavir and tenofovir) are potent, are well tolerated, and demonstrate very low incidence of resistance.27
What’s new?
Support for increased management of HBV in primary care is recommended in the National Hepatitis B Strategy, including prescribing of antivirals for HBV by accredited general practitioners in order to expand access to treatment.
Summary
Chronic hepatitis B is an increasingly common condition in Australia. With an estimated 100 000 people living with undiagnosed HBV, and less than one-fifth of those needing treatment currently receiving it, there is clear evidence that our response needs improvement.
Any person living with CHB requires regular monitoring, at least yearly; there is no such thing as a healthy carrier. Increasing access to antiviral treatment will prevent deaths due to advanced liver disease, and help address the rising burden of HBV related liver cancer. General practitioners must be supported to help improve access to diagnosis and care for Australians living with CHB.
Resources
Fact sheets for patients in languages other than English
Competing interests: None.
Provenance and peer review: Commissioned; externally peer reviewed.