Question 1
What differential diagnosis should be considered for oedema of the soft palate and uvula in a patient of this age group?
Question 2
What urgent consideration is required? What immediate management is appropriate?
Further information
Jack was treated with an intramuscular antihistamine (promethazine 25 mg) and transferred by ambulance to the local emergency department for airway assessment. There was no clinical response to promethazine. Examination of the oropharynx showed oedema of the soft palate and uvula as previously. Flexible laryngoscopy by an otolargyngologist showed there was no laryngeal oedema and the airway was patent.
Question 3
What is the pathophysiology of angioedema and is it clinically relevant?
Question 4
Which angioedema syndrome is most likely in this case?
Question 5
Why was promethazine unhelpful in Jack’s case?
Further information
Jack’s medication list was reviewed. It was confirmed that he was taking candesartan, an angiotensin receptor blocker (ARB), but not an angiotensin-converting enzyme (ACE) inhibitor. He had been previously treated with an ACE inhibitor, ramipril, but this had been stopped 5 years before the current presentation due to coughing.
Question 6
What is the relationship between drugs acting on the renin–angiotensin–aldosterone system (RAAS) and angioedema? Is the relationship the same for ACE inhibitors and ARBs?
Question 7
Was the introduction of saxagliptin 6 months previously relevant?
Answer 1
The differential diagnosis for oedema of the soft palate and uvula is:
- angioedema
- viral infection (herpes simplex virus, Epstein Barr virus or Coxsackie virus)
- bacterial infection (peritonsillar or retropharyngeal abscess)
- neoplasia
- foreign body
- erythema multiforme (Stevens-Johnsons syndrome)
- pemphigus vulgaris.
Given the relatively sudden onset, the absence of other symptoms, such as fever, rash or preceding weight loss, and no history suggestive of a foreign body, angioedema is the most likely diagnosis.
Answer 2
Angioedema, a localised swelling of the subcutaneous and submucosal tissues, may occur as part of an anaphylactic reaction or as a separate condition. Anaphylaxis is characterized by the presence of associated systemic manifestations, such as respiratory difficulties or hypotension, in addition to the localised skin or mucosal changes of angioedema. Jack did not have other features suggestive of anaphylaxis at the initial presentation.
In localised angioedema, the first priority is assessment and protection of the airway. Angioedema occurring in the oropharynx (i.e. tongue, palate, uvula) is of more threat to the airway and of more urgent concern than angioedema isolated to the lip.
Answer 3
There are several different types of angioedema1 (Figure 1). All occur as a result of increased permeability of post-capillary venules, mediated by a range of vasoactive substances (eg. histamine, bradykinin). The different types of angioedema are distinguished by differences in their underlying pathophysiology and clinical presentation.2
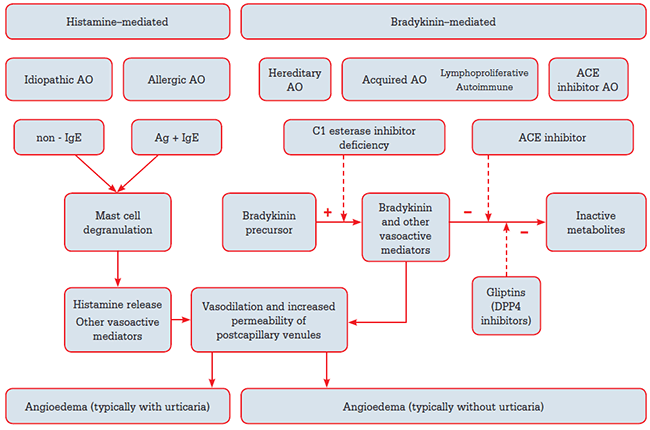
Figure 1. Schematic representation of mechanism and clinical features of major angioedema syndromes
AO = angioedema; IgE = immunoglobulin E; Ag = antigen; ACE = angiotensin converting enzyme; DPP4 = dipeptidyl peptidase-4
For example, allergic angioedema occurs when an allergen triggers histamine release from activated mast cells.1 Swelling begins shortly after allergen exposure, and itch and urticaria (due to histamine release) are common features, although atypical presentations without skin manifestations are possible.
Hereditary angioedema, on the other hand, results from C1 esterase inhibitor deficiency, which leads to increased production of bradykinin. Angioedema caused by bradykinin typically occurs without itch or urticaria.2 C1 esterase inhibitor deficiency can also be acquired in association with lymphoproliferative disorders or autoimmunity. ACE inhibitors impair the breakdown of bradykinin and thereby increase the risk of bradykinin-mediated angioedema.3
Answer 4
As Jack had no itch or urticaria, his presentation is suggestive of angioedema caused by bradykinin. He had no family history of angioedema, and this was his first episode, suggesting a hereditary angioedema was less likely. Follow-up tests showed normal levels of complement. He was systemically well, suggesting an underlying lymphoproliferative disorder or autoimmune condition was less likely.
Answer 5
As bradykinin rather than histamine seems more likely to have caused this case of angioedema, it is unlikely that promethazine would be helpful. Antihistamines, steroids and adrenaline are traditional agents for treatment of angioedema; however they are ineffective for bradykinin-mediated angioedema, which includes hereditary angioedema and ACE inhibitor angioedema. They are also potentially dangerous in patients with ACE inhibitor angioedema, who are usually older and have an elevated cardiovascular risk. Use of a sedating antihistamine may be hazardous in patients with potential airway compromise and should be avoided. Bradykinin antagonists are an emerging therapy for this group of patients,4 although their role in patients with angioedema related to use of ARBs has not yet been investigated.
Answer 6
The RAAS is central to the regulation of the cardiovascular system and blood pressure. Therapeutic agents that target this system, initially ACE inhibitors and then ARBs, have been used increasingly in clinical practice over the last 40 years. Angioedema was recognized as an adverse effect of ACE inhibitors during the 1980s and 1990s5–8 and was found to be due to increased levels of bradykinin.9,10 Fatalities resulting from airway obstruction have been reported.11
Compared with what is known for ACE inhibitors, the relationship between ARB use and occurrence of angioedema is not as clear. Initially it was thought that losartan lacked the bradykinin-potentiating effect of ACE inhibitors12 and therefore ARBs ‘…do not have two side effects that can interrupt therapy with ACE inhibitors – cough and angioedema’.12 However, cases of angioedema associated with ARBs began to be reported13–15 and a study showed that ARBs increase bradykinin levels in hypertensive patients.16 Although it seemed that the risk of angioedema was lower with ARBs than with ACE inhibitors, there remained some uncertainty.17
A recent study reviewed the risk of angioedema with various RAAS inhibitors in clinical trials.18 The overall incidence of angioedema with ARBs was 0.11%, which was not significantly different from placebo (0.07%). However, the risk was increased in heart failure patients (relative risk 3.16; 95% confidence interval (CI) 2.20–4.53). Overall the incidence may have been minimised in these trials by excluding patients at risk of angioedema.19
In recognition of the limited availability of safety information for medicinal products from clinical trials and at the time of marketing, the 2007 US Food and Drug Administration Amendments Act20,21 has required an increased focus on post-marketing surveillance. As part of this program, the relationship between drugs acting on the RAAS and angioedema was evaluated among a large, diverse, population-based cohort in real-world (i.e. non-trial) clinical settings.22 Risk for the occurrence of angioedema was compared in large cohorts of patients taking ACE inhibitors or ARBs with beta-blockers. Beta-blockers were the reference group as they are not known to be linked to angioedema. Risk of angioedema was increased threefold with ACE inhibitors (hazard ratio 3.04; 95% CI 2.81–3.27) and was slightly higher with ARBs (hazard ratio 1.16 95% CI 1.00–1.34). Jack’s candesartan may have contributed to the development of angioedema and it was discontinued.
Answer 7
Saxagliptin is a dipeptidyl peptidase-4 (DPP4) inhibitor used in the treatment of diabetes.23 DPP4 inhibitors potentiate incretins, which are gastrointestinal hormones that increase insulin release and also inhibit the breakdown of bradykinin, although this is a secondary role when compared with ACE inhibitors.24 In individuals taking an ACE inhibitor, concurrent vildagliptin use was associated with an increased risk of angioedema (odds ratio 4.57; CI 1.57–13.28), although there was no increased risk with vildagliptin alone.24 The effect of combined gliptins and ARBs on angioedema has not specifically been investigated, so the relevance of the introduction of saxagliptin 6 months previously in this case is somewhat uncertain.
Key points
- Consider whether angioedema is histamine- or bradykinin-mediated.
- Antihistamines, steroids and adrenaline have not been shown to be effective for bradykinin-mediated angioedema and may have adverse effects, particularly in older patients who are at increased cardiovascular risk.
- ARBs may be associated with a small increase in the risk of angioedema, although the risk is less than that with ACE inhibitors.
Competing interests: Genevieve Gabb has received a speaker fee from AstraZeneca.
Provenance and peer review: Not commissioned; externally peer reviewed.