On presentation the treating doctors thought he had an allergic reaction and he was taken to the resuscitation room of the hospital. The swelling of his tongue rapidly progressed and was compromising his airway. There was no improvement in his condition after administration of adrenaline, promethazine and hydrocortisone. The decision was made that he would have to be intubated or, if that failed, would need a surgical airway. During pre-oxygenation the patient’s airway patency deteriorated rapidly and he was intubated with a traditional MacIntosh laryngoscope using a bougie technique. Suxamethonium was used for muscle relaxation and propofol for sedation.
At intubation the tongue was massively swollen along with some swelling of the submandibular tissues and the pharynx. A photograph of the patient was taken a few minutes after intubation and shows the size of the tongue (Figure 1). The patient was flown to the nearest intensive care unit, and was kept sedated and ventilated during the 700 km flight. Three days later the swelling had subsided.
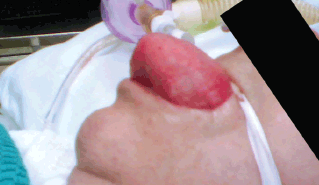
Figure 1. Tongue swelling may be the most prominent feature of ACE inhibitor angioedema and can make intubation challenging
The diagnosis and differential diagnosis
This is a case of angioedema, a significant swelling of tissues deep to the skin and mucous membranes. Angioedema differs from conditions such as urticaria where there is swelling of the skin. Angioedema most commonly affects the tongue and face. It can also affect abdominal organs.
Angioedema without urticaria can be induced by NSAIDs, or it can be part of a histamine-driven allergic reaction, or be driven by accumulation of bradykinin.1 The patient had not taken any NSAIDs and there was no response to treatments directed towards a histamine-driven allergic process. Reasonable explanations of this presentation of angioedema without urticaria include:1–3
- ACE inhibitor angioedema, which occurs in ACE inhibitor users at a prevalence greater than 1:1000. Most cases are much milder than this one.
- Hereditary angioedema is a condition with a prevalence of about 1:10 000 or less.2 There is a deficiency in C1 esterase inhibitor, or C1 esterase inhibitor is present but not functional, allowing accumulation of active C1 esterase. This in turn leads to, among other things, a periodic accumulation of bradykinin in tissues that results in angioedema.
- Acquired C1 esterase inhibitor deficiency is an autoimmune condition in which there is autoimmune inactivation of C1 esterase inhibitor.4 This renders the C1 esterase inhibitor ineffective and excess C1 esterase activity can develop. This can result in accumulation of bradykinin in tissues. This condition is very rare with less than 150 described cases.
- Hereditary angioedema without C1 esterase inhibitor deficiency appears to be due to abnormalities in clotting factor XII and occurs with a prevalence of less than 1:100 000 people.5 The majority of cases are female.
The absence of a family history made either type of hereditary angioedema very unlikely. Further this would be an extraordinarily delayed onset for either type of hereditary angioedema.
The patient was investigated for C1 esterase inhibitor deficiency. Functional C1 esterase inhibitor level was 105% (reference range 70–130%). This ruled out hereditary angioedema and acquired C1 inhibitor deficiency. With respect to hereditary angioedema without C1 esterase inhibitor, over 90% of patients with this very rare condition have a positive family history and most cases are female.
From the above we can conclude the diagnosis is ACE inhibitor angioedema. Neither of the treating doctors recognised the condition as ACE inhibitor angioedema at time of presentation and they treated it as an allergic reaction.
Discussion
ACE inhibitor angioedema has long been a known adverse drug reaction occurring with the whole group of ACE inhibitors. It has been estimated by various means to occur in 0.1–0.42% of patients on an ACE inhibitor.6–8 It occurs most commonly in the first year of treatment with an ACE inhibitor but can occur after years of use.9,10 Patients may have multiple episodes of ACE inhibitor angioedema before the condition is recognised, and patients with initially mild episodes may progress to having severe life-threatening episodes.9,10
The condition is usually not correctly diagnosed on initial presentation and even in fatal cases the condition often appears to have been unrecognised.11,12 The incidence is likely to be underestimated, which is concerning because of the potential for the condition to be lethal.
The typical case involves swelling of the tongue, but the lips, pharynx, larynx and submandibular tissues can also be involved.9,12 In severe cases failure to secure an airway has led to death by asphyxia.11,12 There have been a number of reported cases where the condition has manifested only as angioedema of the gut. Such cases are usually initially diagnosed with primary abdominal complaints, such as irritable bowel or ischaemic colitis.13,14 Such patients are at risk of undergoing laparotomy.15
There are a number of risk factors for development of ACE inhibitor angioedema. African American patients have repeatedly been shown to be at approximately four times the risk of other patients.6,8,16 Smoking also increases the risk of developing this condition.16 A history of an ACE inhibitor-induced cough is associated with a 9-fold increase in the risk of angioedema.16 Thus it is important to take patients with an ACE inhibitor-induced cough off this group of drugs.
The lack of response to adrenaline, antihistamines and steroids in this case is typical and is due to the condition being related to accumulation of bradykinin and not histamine. With ACE inhibition the breakdown of bradykinin is partly dependent on dipeptidyl peptidase 4 (DPP4). Levels of DPP4 may be reduced in patients who have ACE inhibitor angioedema.17–19 The incidence of angioedema with the combination of an ACE inhibitor and a DPP4 inhibitor seems to be 4–5 times higher than the risk with an ACE inhibitor alone.19
A concern for the future is that DPP4 inhibitors are rapidly entering the market for the treatment of type 2 diabetes. These drugs help control type 2 diabetes by inhibiting the breakdown of incretins. As ACE inhibition is usually seen as first-line therapy for hypertension in patients with type 2 diabetes, the incidence of angioedema, a potentially lethal condition, may become more common now that DPP4 inhibitors are regularly being used. If, as in this case, patients can develop angioedema after many years of stable ACE inhibitor use then adding a DPP4 inhibitor may become the destabilising trigger for an episode of angioedema.
Treatment options
In the long term, after an episode of ACE inhibitor angioedema, the patient must not take this class of drug again. Frequently, the patient will be switched to an angiotensin II receptor blocker (ARB). These drugs can also induce angioedema though at a much lower rate than ACE inhibitors. Further, ARBs do not seem to induce severe episodes.20
The acute management of this condition is not clear-cut. The use of antihistamines, steroids and adrenaline is often quoted although there does not seem to be any evidence for efficacy with this line of treatment and certainly in this case there was no apparent benefit. There is often an automatic response among clinicians to give an antihistamine for any condition that could be an immediate hypersensitivity reaction. In a case such as this there is a risk with the use of a sedating antihistamine as the airway may be about to become compromised and a sedating drug may lead to respiratory decompensation.
There are case reports of ACE inhibitor angioedema being successfully treated with fresh frozen plasma.21–23 These reports have appeared for over a decade now. There is a recent study of seven patients given fresh frozen plasma for this condition, all of whom had failed to respond to antihistamines, steroid and adrenaline.24 All seven rapidly improved after fresh frozen plasma. Thus, faced with a patient exhibiting severe ACE inhibitor angioedema, a trial of a couple of units of fresh frozen plasma would seem very reasonable to hopefully shorten the attack or to reduce the need for what could be a very difficult intubation.
There are also reports of the successful use of the bradykinin receptor antagonist icatibant,25 a short peptide that has a structure related to bradykinin. It can be administered by subcutaneous injection and is used by patients with hereditary angioedema, often by self-administration. It is not available in rural hospitals and it costs about $3000 per dose.26 By comparison, fresh frozen plasma is available in most rural hospitals and is relatively cheap at $300 a bag.27 Fresh frozen plasma, however, has the disadvantage of having to be thawed, and the minor risk of being a blood product.
Clearly, cases of the severity of this one require urgent airway management and may require emergency cricothyroidotomy.
Competing interests: None.
Provenance and peer review: Not commissioned; externally peer reviewed.
Acknowledgements
The author thanks Dr Walter Byrne and the staff at Esperance District Hospital who made effective treatment of this patient possible in the somewhat medically resource-poor setting of Esperance, Western Australia. They were most helpful in providing access to clinical data. The author also thanks the patient for generously consenting to publication of this case.