Case study
Mr B, 65 years of age, presented with a painful black left toe. He complained of pain and bluish discoloration of his left foot for the past 8 months, which had worsened in the past 3 weeks. He experienced no numbness or paraesthesia, and did not recall any trauma to his left foot or toe. Relevant past medical history included hypertension and type 2 diabetes mellitus, neither of which have been optimally controlled.
Mr B is a retired restaurant owner and a nonsmoker. He has regularly drunk 1–2 glasses of white wine a day for the past 50 years. His father and two of his brothers died of heart attacks. Mr B has led a sedentary life for the past 4 decades, with minimal sports activity.
He attributed the cause of the presentation to his ‘cutting the toenail in the wrong way’, which he believed led to the infection and blackening of his toe. He initially applied Vaseline and olive oil to the toe until the increasing pain disrupted his sleep and alerted his wife to the problem. His wife stated that Mr B was known to be stoic, and he did not complain even when he accidentally severed the tip of his little finger while working as a trainee chef when he was young.
On examination, there was a 2 x 2 cm black eschar on the plantar aspect of the left big toe with a well demarcated margin of granulation tissue (Figure 1). The eschar was not tender to palpation. There was cyanosis over the second and third toe extending to the dorsum of the left foot. The foot felt cold to palpation with absent dorsalis pedis and posterior tibial pulses. The right foot was normal in comparison. The patient could not weight bear on his left foot and limped due to poor balance.
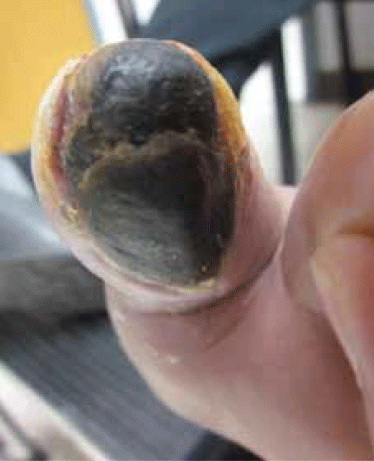
Figure 1. The patient’s toe at presentation
Question 1
What is the diagnosis?
Question 2
What are the differential diagnoses?
Question 3
How would you assess the severity of the condition?
Question 4
What are the goals of treatment and possible treatments?
Question 5
What is the prognosis?
Answer 1
Gangrene of the great toe due to critical limb ischaemia (CLI).
Answer 2
Differential diagnoses included cutaneous anthrax and necrotising fasciitis secondary to group A haemolytic streptococci. Anthrax infection, characterised by an eschar at the site of inoculation, would resemble Mr B’s lesion; however, the eschar is usually painless. Necrotising fasciitis is a severe form of spreading tissue infection leading to necrosis and sloughing secondary to haemolytic group A streptococci infection; however, the condition often develops within hours, with intense pain, swelling and rapid necrosis of tissue in the local area, which may appear black in colour.
Answer 3
Critical limb ischaemia manifests as chronic rest pain, nonhealing ulcers and gangrene of the affected limb as a result of nonacute arterial occlusion from atherosclerosis. It is per se a marker for generalised and severe stage peripheral arterial disease (PAD) with poor prognosis and survival.1 The prevalence of PAD ranges from 3–5% in the general population and prevalence of CLI is around 1% of all patients with PAD.2 Annual incidence for CLI is around 500 to 1000 per million, with the elderly, smokers and diabetics more at risk.3 Critical limb ischaemia often presents with nonhealing ulcers and ischaemic pain, and less commonly with acute gangrene. Compared to patients with intermittent claudication, patients with CLI have a threefold higher risk for all cardiovascular events and deaths.3
Given the multiple comorbidities of CLI and the interdisciplinary nature of care (physicians, general surgeons, vascular interventionists), the best algorithm for treatment is not straightforward but often a case-by-case consideration. In view of this, the Lower Extremity Grading System was proposed in 20044 to standardise decision making but further validation is needed. The Ankle Brachial Index (ABI) is considered the first line investigation for diagnosing peripheral arterial disease of the lower limb. Critical limb ischaemia is defined as having an ABI of 0.4 or less. To further locate the levels of stenosis and assess their severity, noninvasive imaging techniques like computed tomography angiography and magnetic resonance angiography can be considered before the more invasive gold standard investigation of digital subtraction angiography.
Peripheral arterial diseases can be classified into Fontaine stage I to IV,5 or Rutherford category 0 to 66 according to clinical symptoms and evidence of tissue damage (Table 1).
Table 1. Classification of peripheral arterial diseases according to clinical symptoms and evidence of tissue damage
| Fontaine stages5 | Clinical | Rutherford category6 | Clinical |
|---|
| I |
No symptoms |
0 |
No symptoms |
| IIa |
Mild claudication |
1 |
Mild claudication |
| IIb |
Moderate to severe claudication |
2 |
Moderate claudication |
| 3 |
Severe claudication |
| III |
Ischaemic pain at rest |
4 |
Ischaemic pain at rest |
| IV |
Ulceration/gangrene |
5 |
Tissue loss (mild) |
| 6 |
Tissue loss (major) |
Answer 4
Goals of treatment for CLI include pain relief, management of ulcers and infections, improvement of quality of life, revascularisation without amputation and longer overall survival. Current consensus is to avoid limb amputation if possible, and when necessary, a below knee approach is preferred for better ambulation and lesser mortality (5% mortality for below knee amputation vs 16% for above knee amputation).7
Depending on the level and degree of arterial stenosis, either endovascular angioplasty or definitive vascular reconstructions are indicated to revascularise the ischaemic limb. In the last decade, advances in percutaneous angioplasty and stenting have made endovascular revascularisation the preferred primary intervention as revealed by a 12 year retrospective study by Kudo et al.8 That said, the chance of success and patency rate decrease tremendously from aorto-iliac to infra-inguinal levels. Surgical reconstruction, which includes aorto-femoral, ileo-femoral or femoro-femoral bypass and aorto-iliac endartectomy, all produce better patency rates but carry a higher rate of mortality.9 Hybrid procedures of endovascular and surgical revascularisation is gaining favour; this enhances rates of vessel patency and limb salvage.10,11
Pharmacological therapies like statins, beta-blockers and antiplatelet medications help to reduce perioperative risks for surgical revascularisation and improve postoperative patency of arteries and grafts.
In advanced CLI that is nonamenable to vascular reconstruction or amputation, spinal cord stimulation,12 intermittent pneumatic compression13 and hyperbaric oxygen14 have been used to relieve ischaemic symptoms and salvage the limb. Finally, improvements in perfusion have been reported in patients with CLI after treatment with bone marrow stem cells15 or activated peripheral blood mononuclear cells.16
Answer 5
Untreated, 50% of patients with CLI will proceed to limb amputation and 25% will die within 1 year.17 In view of that, the amputation free survival (AFS) has been proposed to address these two outcomes (amputation and mortality) as the primary measure of success for treating CLI in major clinical trials like the BASIL18 and PREVENT.19 It has been reported that in end stage no option CLI (NO-CLI), AFS has improved over the last 2 decades, which is most probably a result of improved medical care.20
Case study continued
Mr B decided not have an operation; a decision the vascular surgeon agreed with. The family requested palliative care. Unfortunately, due to the lack of a terminal diagnosis, the local palliative care team declined a role in the management of Mr B’s symptoms. In order to alleviate the intense ischaemic pain, Mr B was put on potent opiates and a mild hypnotic for his sleep. He was reviewed weekly to monitor and treat possible bacterial infections due to the gangrenous wound. Within 6 weeks of initial presentation, Mr B’s gangrenous toe dried up remarkably to form a mummified digit connected to the first metatarsal bone (Figure 2–4). Two courses of oral cephalosporins were prescribed to control cellulitis at the base of the toe. Pain control was deemed adequate as Mr B managed to have 5 hours sleep every day. Paradoxically, the ischaemic pain became less when the toe was totally mummified (Figure 5). In view of his inoperable stage of CLI and inability to involve the palliative care services locally, Mr B was referred to another service for a second opinion regarding eligibility of spinal cord stimulation and intermittent pneumatic compression therapy. Unfortunately, Mr B died of a massive cerebrovascular accident a week after the referral was made.
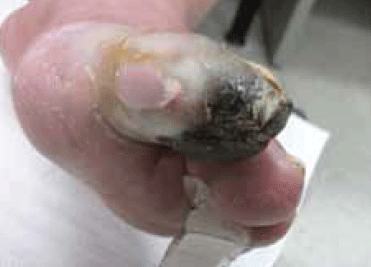
Figure 2. The patient’s toe at 3 weeks
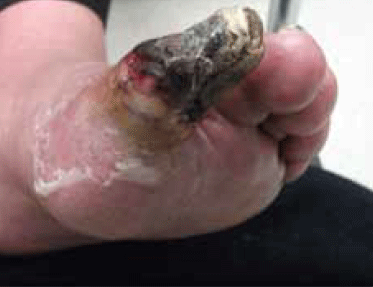
Figure 3. The patient’s toe at 4 weeks
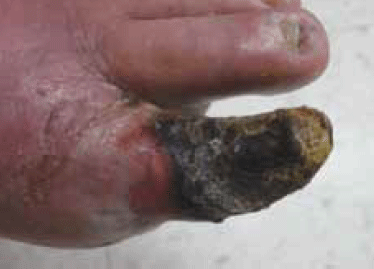
Figure 4. The patient’s toe at 5 weeks
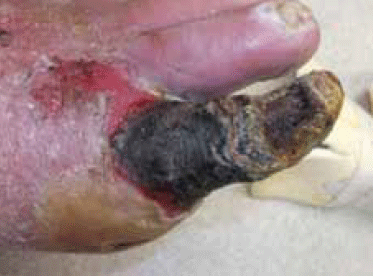
Figure 5. The patient’s toe at 6 weeks
Key points
- Critical limb ischaemia is the most severe form of peripheral arterial disease and is often a result of arteriosclerosis or diabetes.
- It is characterised by resting ischaemic pain, nonhealing ulcer and gangrene.
- Untreated, 50% of patients with CLI will proceed to limb amputation and 25% will die within 1 year.
- Definitive treatment aims at revascularisation to salvage the ischaemic limb if possible, which includes endovascular procedures, surgical bypass or a hybrid combination of both.
- Medical treatment aims at reducing the operative/perioperative risks and improving overall vessel patency, limb, and patient survival.
- When revascularisation fails, other options such as spinal cord stimulation, intermittent pneumatic compression and hyperbaric oxygen may help improve symptoms and wound care.
- Without intervention, the ischaemic anatomy may dry up and mummify: a reminder of how nature will take its course.
Conflict of interest: none declared.