Australia had 3.8 million children aged 0–12 years in 2013.1 Australia has a good record in child health, with many initiatives to lower mortality rates, enhance immunisation coverage and manage chronic conditions.2
Understanding medicine use in children is an important component in the assessment of therapies used in the paediatric population.3 A recent systematic review of Australian studies using national health data to explore prescribed medicines identified that little is known about the overall uses of medicines in children.4 The majority of published studies focus only on a specific group of medicines such as anticonvulsants,5 antipsychotics6 and hypoallergenic formula.7 Information on antibiotic prescription in children comes from one randomised controlled trial conducted among 54 general practitioners (GPs) in 1997–99;8 two studies on prescribing in general practice that compared cross-sectional surveys from 1990–91 to 2000–03;9,10 and one point-prevalence survey on antimicrobial use on a single day in 2012 in eight children’s hospitals across Australia.11 None of these studies used a nationally representative dataset.
The aim of this study was to examine the current use of prescribed medicines in Australian children by using a large, national prescription claims dataset, with a special focus on antibiotic prescribing.
Methods
De-identified national pharmacy claims data from the Australian Government’s Department of Human Services (DHS) were used, providing a 10% random sample of individuals with medicines subsidised and dispensed under the Pharmaceutical Benefits Scheme (PBS).12 For the randomly selected individuals, all their associated dispensings were included in the sample. PBS data are collected from pharmacies and private hospitals, and discharge or outpatient dispensings from many public hospitals. Data include information on the patient’s age, gender and beneficiary status (general or concessional beneficiary status), as well as dispensing information, which includes date of supply, drug code, therapeutic class, generic name, form, quantity dispensed and number of repeats. Since 2012, PBS data represent full capture of dispensing records for both general and concessional beneficiaries, including under co-payment prescriptions. Medicines were coded in the dataset according to the World Health Organization’s (WHO’s) Anatomical Therapeutic Chemical (ATC) classification system.13
A longitudinal study was undertaken that included children aged 0–12 years who were dispensed medicines (concessional and general) in 2013. All prescribed medicines were identified, and population rates and prescribing patterns were reported by therapeutic class.13 Proportions of children with one, two or more antibiotic dispensings in the year were reported.
Patient-level data were analysed to determine the time between antibiotic prescriptions, as well as the type of antibiotics and their sequence in time. Monthly rates of children who received at least one antibiotic prescription (but no prescriptions for any other medicine) and those who received antibiotic dispensing together with other medicine(s) in the same month were reported as a proportion of the total number of children with at least one antibiotic dispensing in a given month.
Comorbid conditions of children who had antibiotics dispensed were determined using the Rx-Risk-V Comorbidity Index.14 The individual comorbidity index was based on medicines for distinct chronic conditions dispensed to a patient across 2013 (ie the index would be zero if there were no medicines for chronic conditions).
To extrapolate to national rates, we multiplied the results by a factor of 4.5% to reflect the representativeness of our sample of children using the Australian Bureau of Statistics (ABS) population estimates of children aged 0–12 years at 30 June 2013.1
Statistical analyses
Pearson’s chi square tests were used to compare proportions. Poisson regression models were used to assess the effects of age and gender on population rates. All data analyses were performed using SAS for Windows, Version 9.3 (SAS Institute, Cary, North Carolina, US).
Ethics approval
The study was approved by DHS’s External Request Evaluation Committee for analysis of PBS data (reference number MI4524). All analyses were conducted using de-identified data and in accordance with DHS Privacy Policy.
Results
Overall prescribing
There were 107,707 unique children aged 0–12 years who received 383,515 dispensings for medicines in 2013. Age distribution showed that 40% of these children were aged 0–4 years, another 40% were aged 5–9 years, and 20% were aged 10–12 years. More boys than girls received medicines in any age group (chi-square test, P <0.0001).
Antibiotics for systemic use were the most commonly dispensed medicines in children (41% of all dispensings), followed by medicines for obstructive airway diseases (18%) and psychoanaleptics (9%). The rate of antibiotic prescriptions extrapolates to 414 prescriptions per 1000 of the estimated children’s population aged 0–12 years in Australia.
Antibiotic prescribing
There were 75,518 unique children in the 0–12 years age range who received at least one antibiotic for systemic use (ATC code J01) in 2013. Of these 75,518 children, 44% were aged 0–4 years, 39% were aged 5–9 years, and 17% were aged 10–12 years; 48% were girls and 52% were boys. Two-thirds of antibiotic dispensings were prescribed by GPs.
The population rates for children who were prescribed at least one systemic antibiotic in 2013 were: 49% for those aged 0–4 years, 44% for those aged 5–9 years, and 33% for those aged 10–12 years. Poisson regression models showed that age and gender were significantly associated with antibiotic use. Younger children were more likely to receive antibiotics than older children (0–4 versus 5–9 years of age: relative rate (RR) = 1.066, 95% confidence interval (CI) = 1.049–1.082, P <0.0001; 0–4 versus 10–12 years of age: RR = 1.404, 95% CI = 1.378–1.432, P <0.0001; 5–9 versus 10–12 years of age: RR = 1.317, 95% CI = 1.290–1.344, P <0.0001), and boys were more likely than girls to be dispensed antibiotics (RR = 1.015, 95% CI = 1.002–1.029, P = 0.040).
Penicillin was the most commonly used class, accounting for 62.3% of all antibiotics dispensed in 2013. The moderate-spectrum amoxicillin accounted for 43.7% and broad-spectrum amoxicillin with clavulanic acid accounted for a further 11.9% of antibiotic use. Cephalosporin was the second most widely used class (23.7%), with cefalexin contributing for 17.2% of all antibiotic use. Macrolides contributed to 10.9% of all antibiotic use.
The majority of the children (53%) received just one antibiotic dispensing for the whole year (mainly amoxicillin, followed by cefalexin); however, 47% received multiple dispensings – 22% received two dispensings, 11% had three dispensings and the remainder received four or more dispensings in one year (Figure 1). The pattern was similar when stratified by year of age. Of the 47% of children who received multiple dispensings, 30% received various antibiotics, with the average time between prescriptions being two months, and 17% had the same antibiotic on multiple occasions. One-third of children had a repeat prescription filled within 10–14 days of the original prescription, and the other two-thirds received the same antibiotic later (on average, two months after the previous prescription). Figure 2 presents some of the common treatment choices for children who needed two or more antibiotic dispensings in the year, reflecting preference to use mostly penicillins.
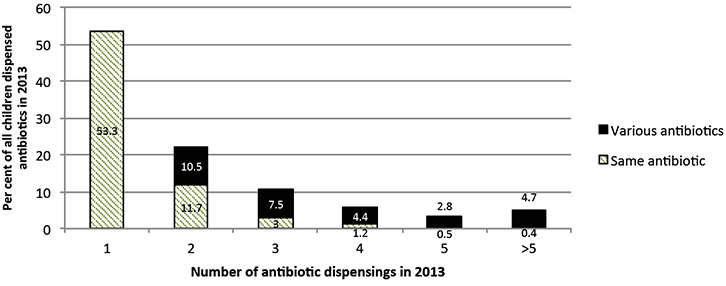 |
Figure 1. Antibiotic dispenses in all children with antibiotics in 2013 (n = 75,518)
Note: for children with multiple dispenses, ‘same antibiotic’ was attributed only when all the dispensings were for the same antibiotic |
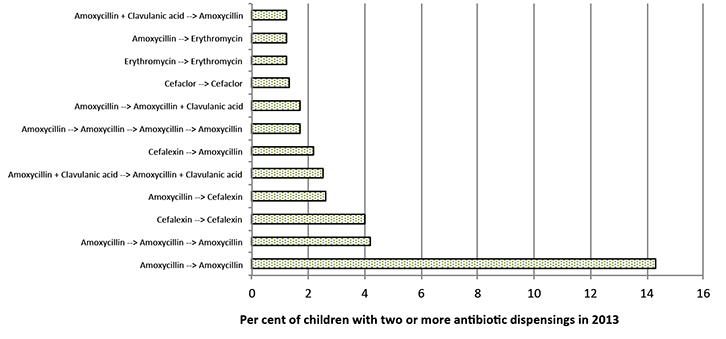 |
Figure 2. Common treatment choices (in time order) for children with two or more antibiotic dispenses in 2013 (n = 35,248)
Note: this graph only presents proportions above 1.0% |
Antibiotic mono-use and co-use with other medicines
Antibiotics were dispensed without other medicines (mono-use) for three times as many children as those who received antibiotics co-dispensed with other medicines (Figure 3). Fifty-three per cent received just one dispensing of systemic antibiotic in 2013. The majority of children who received antibiotics (71%) had no comorbid condition. Eleven per cent had comorbidities and received one dispensing, and a further 18% had comorbidities and received multiple antibiotic dispensings in the year. The most common comorbid conditions were reactive airway disease, steroid responsive disease, gastric acid disorders and allergies.
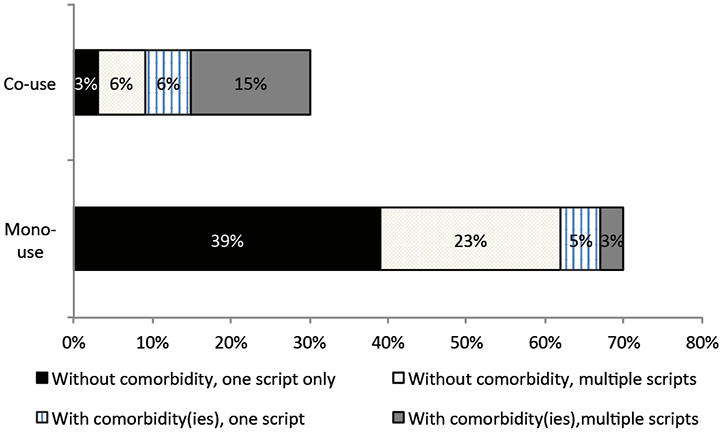 |
Figure 3. Antibiotic use as a monotherapy or with other medicines, by comorbidities and number of dispensings in 2013 as a proportion of the antibiotic cohort (n = 75,518)
Note: ‘mono-use’ denotes that every time an antibiotic(s) was dispensed, it was dispensed without any other medicines in the same month |
Month-by-month analysis confirmed that the majority of the children (75–80%) received antibiotic(s) only and no other medicine(s) in the same month (Figure 4; mono-use). Between 5% and 7% of children who received antibiotic(s) also received asthma medicine(s), with rates peaking in the winter months. Three to six per cent had corticosteroids for systemic use, and another 1–3% received the antibiotic(s) together with asthma medicines and systemic corticosteroid. Only 3% or less of antibiotic users had antibiotics and corticosteroids for dermatological use, or ophthalmologicals, otologicals or other medicines, dispensed in the same month.
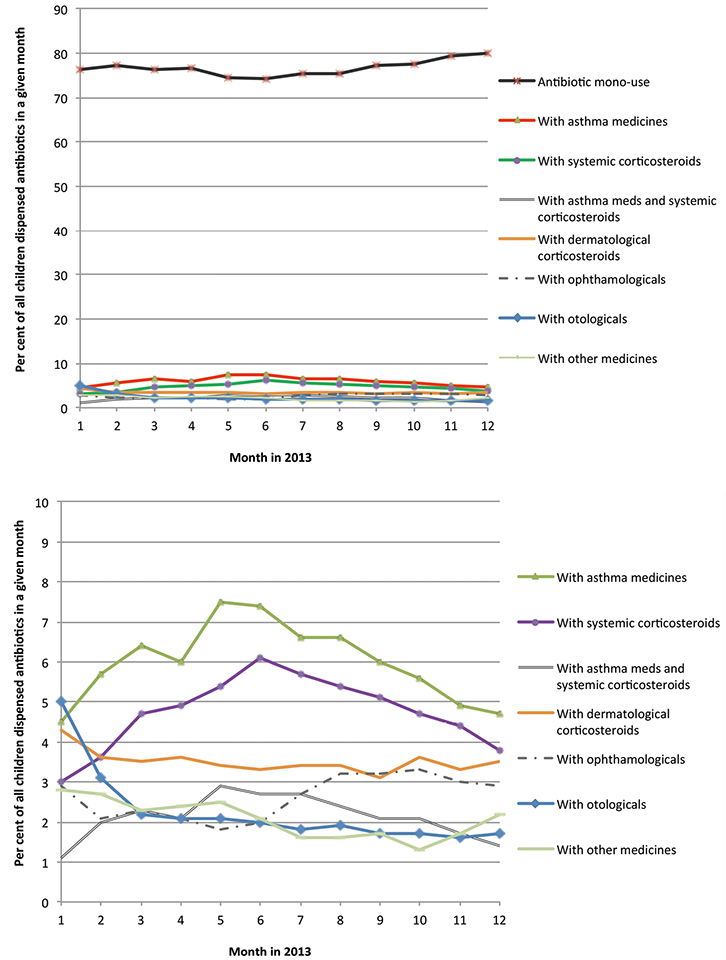 |
Figure 4. Monthly rates of antibiotic use as a monotherapy or with other medicines
Note: ‘mono-use’ denotes that the antibiotic(s) were dispensed without any other medicines in the given month |
Discussion
In 2013, we found that at least one systemic antibiotic was dispensed to almost half of children aged 0–4 years, 44% in those aged 5–9 years, and one in three of those aged 10–12 years, with more boys than girls receiving it in every age group.
Our analysis confirmed that antibiotics continue to be the major medication group prescribed for children since 2000–01.9 Our study found that more than half of the children (53%) who received antibiotics in 2013 received a one-off dispensing. The majority (71%) had no comorbid condition. Most of the time, antibiotics were not co-dispensed with any other medicines indicative of a serious illness. Recommended first-line antibiotics such as amoxicillin (moderate-spectrum penicillin) and cefalexin (first-generation cephalosporin) contributed to the majority of antibiotic use (61% of all systemic antibiotic dispensings).
An earlier study in Australian general practice in 2002–03 found that respiratory problems, such as upper respiratory tract infection (URTI), bronchitis/bronchiolitis, tonsillitis and acute otitis media, were the most common problems for which antibiotics were dispensed by GPs.10 While antibiotics were prescribed less often for the management of URTI and bronchitis/bronchiolitis, their use increased for acute otitis media.10 Some of these conditions usually resolve without the need for antibiotics. Coordinated interventions to improve appropriate and rational antibiotic prescribing are vital for preventing the development of resistance.15,16 Appropriate use involves selection of a ‘targeted’ spectrum antibiotic, as well as an appropriate dose and duration.15
We found that antibiotics accounted for 41% of all prescriptions, which is higher than that found in a US study using a large prescription dataset that reported that systematic antibiotics accounted for 27% of all prescriptions dispensed to children aged 0–17 years in 2010.3 However, in our study, the prescription rates per 1000 children are very similar to another US study that reported a rate of 410 prescriptions per 1000 children aged 0–17 years in 2010,17 and a UK study that reported an average rate of 511 prescriptions per 1000 persons aged 0–19 years.18
We analysed data from a national dataset that captured prescription data of all Australians. We included general and concessional beneficiaries as since 2012 all co-payment prescriptions have been recorded in the PBS data, allowing for generalisation of the results to the entire Australian population. However, as the child population in our sample appeared to be under-represented compared with a recent report from the Drug Utilisation Sub Committee (DUSC),19 we had to factor that in when calculating population rates by age.
Our estimated population rates are slightly lower than those reported by DUSC for any antibiotic(s) used in 201319 (58% for those aged 0–4 years, 45% for those aged 5–9 years, and 36% for those aged 10–14 years) as the DUSC estimate includes topical antibiotics. This limitation is not expected to affect the remainder of the analysis (eg co-prescribing rates) as it was based on patient-level data and the data were complete for each individual person.
Another limitation of the study is that we were unable to determine the indication for medicine use, as diagnostic information was not available in the data. We also could not ascertain whether the dispensed medicines were actually taken. We could not capture medicines that are not listed under the PBS, including those available over the counter; however, all systemic antibiotics are prescription-only.
Implications for general practice
Australia’s antibiotic prescription rates are high by international standards. Australia ranks 11th when compared with 27 European countries, and rates of use are more than two times higher than some comparable high-income countries, such as the Netherlands.20 At the same time, antimicrobial resistance is increasing. The WHO has described antimicrobial resistance as having ‘reached alarming levels in many parts of the world’.21
Improving antibiotic use in children, particularly minimising their use for URTIs, should be a priority for action as intervening in children has the potential to make a sustained improvement in practice over time. Our analysis found that more than half of all children who received an antibiotic in 2013 received only a single dispensing, and the majority had no comorbidities nor required any other prescription medicines to manage their conditions. Furthermore, the majority of antibiotics used were those commonly used for URTIs. Given the increasing global antimicrobial resistance, interventions to reduce inappropriate antibiotic use in children are required. GPs can play a pivotal role in educating children and their parents about the appropriate place of antibiotics in healthcare, with the overall aim of reducing inappropriate use.
Authors
Svetla Gadzhanova PhD, Research Fellow, Quality Use of Medicines and Pharmacy Research Centre, School of Pharmacy and Medical Sciences, University of South Australia, Adelaide, SA. svetla.gadzhanova@unisa.edu.au
Elizabeth Roughead, PhD, Professor, ARC Future Fellow, Quality Use of Medicines and Pharmacy Research Centre, School of Pharmacy and Medical Sciences, University of South Australia, Adelaide, SA
Competing interests: None.
Provenance and peer review: Not commissioned, externally peer reviewed.