Case
DT, a man aged 76 years, presented to his general practitioner with a red lump in the right upper abdominal wall that had increased in size in the previous 2–3 months.
DT’s background history included a duodenal ulcer in 1979, treated Helicobactor pylori infection, hypertension and gastroesophageal reflux disease. DT never smoked, and drank alcohol in moderation. He had no family history of colorectal or other cancers. He denied any change in weight or appetite.
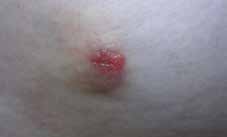
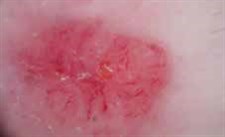
DT looked well and had normal vital signs. Local examination revealed a 20-mm red, fleshy, nodular skin lump on the right upper quadrant of the abdomen (Figure 1). The site corresponded to the inferio-lateral port of laparoscopic cholecystectomy surgery that had been done 2 years earlier. The lump was firm, non-tender and freely mobile. Regional and supraclavicular lymph nodes were not enlarged and the liver was not palpable.
  |
|
Figure 1. Cutaneous nodule on the abdominal wall
|
|---|
Question 1
What differential diagnoses should be considered?
Question 2
What immediate management is appropriate?
Answer 1
Given the nature of the lump, the differential diagnoses include:
- nodular basal cell carcinoma or keloid, which is the likely diagnosis
- skin manifestation of visceral malignancy
- adenocarcinoma of unknown primary (ACUP): poses a significant challenge to the physician1
- primary cutaneous mucinous carcinoma: may be misdiagnosed as metastatic adenocarcinoma2
- dermatofibrosarcoma protuberans: an uncommon (<2% of all soft tissue sarcoma3) skin tumour that usually arises in the trunk as a painless thickened area of skin or nodule
- non-tubercular mycobacterium skin lesions: rare in Australia4
- fungal infection of skin such as sporothrichosis: may present as subcutaneous nodule
- granular cell tumours: uncommon tumours of uncertain histogenesis and involvement of skin is rare5
- keratoacanthoma: as of 2013, there has been no decision as to whether this is a benign or malignant entity.6
Answer 2
Traditional surgical treatment of non-melanoma skin cancer includes excision with a clear margin.7 Radiology investigations are rarely required; however, to confirm the exact size, consistency, extent and degree of fixation, aiming for an excisional biopsy, an ultrasound may be considered.
An incisional biopsy could be considered if the lesion were thought to be deep-seated, vascular or fixed.
Further information
Excision biopsy was performed under local anaesthesia. Histopathology examination reported it as a mucin-secreting adenocarcinoma consistent with a metastatic deposit. Basic investigations showed anaemia and deranged liver functions, indicating mixed hepatocellular and cholestasis. A computed tomography (CT) scan revealed multiple pulmonary and hepatic metastatic lesions with a suspicion of carcinoid tumour, and calcified Meckel’s diverticulum and a thickened mid-rectal wall.
On review of the gall bladder histology from 2 years ago, a focus of invasive squamous cell carcinoma was identified, which had been missed on the initial examination.
Review of the abdominal CT scan showed a subtle soft tissue focus in the gall bladder fossa, which was likely to be a recurrent gall bladder tumour. CT-guided fine needle aspiration cytology (FNAC) from the growth in the gallbladder fossa could be an option for further investigation.8
Gastroscopy and colonoscopy showed benign polyps with no evidence of malignancy. The tumour markers CA-19.9, CA125 and carcinoembryonic antigen (CEA) were significantly elevated.
The gastrointestinal multidisciplinary team agreed that the cutaneous nodule represented metastatic ACUP, likely to be of pancreatic-biliary or gallbladder origin, although the cellular abnormalities of the subcutaneous lesion did not match up with any of the abnormalities found in the gall bladder specimen taken 2 years earlier when the laparoscopic cholecystectomy surgery was performed. It was agreed, therefore, to treat the lesion as a biliary primary.
Discussion
The incidence of gall bladder malignancy in US is about 1.2 per 100 000 population, widely varies with ethnicity, is more common in the seventh decade of life and 1.3 times more likely to occur in women, compared with men.9 The presence of gallstones is one of the major risk factors. Porcelain gall bladder is associated with an increased risk of gall bladder cancer.10 Obesity is independently linked with increased risk.11 About 50% of all gall bladder cancers are discovered incidentally and 35% have distant metastases at the time of diagnosis.
The overall incidence of cutaneous metastasis is 5.3%.12 Of these, 24% are from breast cancers, and 3.4–4 % are from colorectal, renal, ovarian or bladder carcinoma. In the English literature, only nine cases of adenocarcinoma of the gallbladder with cutaneous metastases have been reported.13
Patients with gall bladder carcinoma may present with constitutional symptoms but sometimes may be asymptomatic, or with cutaneous or subcutaneous nodules. A high index of suspicion is required because the clinical finding may be subtle.14 An acute-onset, persistent, firm papulonodule, especially developing on the chest should alert every practitioner.12
Competing interests: None.
Provenance and peer review: Not commissioned, externally peer reviewed.