Figure 1. Lymph node coloured blue in nodal basin
In SLNB a blue dye and/or radioactive tracer is injected around the primary melanoma on the skin (Figure 1). The tracer finds its way to regional drainage lymph nodes. The closest node(s) showing radioactivity and/or blue colour change are then resected through a minimal incision. The node(s) is examined for evidence of early melanoma metastasis (Figure 2). If this node is positive the remaining adjacent nodes are removed. This idea was subjected to a large prospective randomised controlled trial (RCT) to determine whether the theoretical advantages would be realised. The multicentre selective lymphadenectomy trial (MSLT) involved melanoma units throughout the world, including Australia.
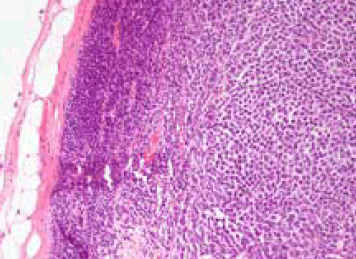
Figure 2. Lymph node with metastatic melanoma. Capsule and lymphoid tissue on the left and melanoma on the right
10-year MSLT study now published1
Of 2001 melanoma patients, intervention patients had a SLNB and, if positive, a regional completion lymphadenectomy (CL). Observation patients underwent delayed lymphadenectomy only if nodal disease developed during observation. The primary study hypothesis was that intervention would improve 5-year2 and 10-year1 survival.
MSLT trial findings
On an intention-to-treat basis, there was no 10-year melanoma-specific survival benefit for intervention patients (77%) versus observation patients (76%). For patients with intermediate-thickness melanoma (Breslow 1.2–3.5 mm), no significant 10-year survival benefit was found in the intervention group (81.4 ± 1.5%), compared with the observation group (78.3 ± 2.0%; P = 0.1).
For patients with thick melanomas (Breslow >3.5 mm), there was also no significant 10-year survival difference: survival rates were 58.9 ± 4.1% in the intervention group and 64.4 ± 4.6% in observation patients (P = 0.56). Overall, the survival of all melanoma patients with melanomas >1.2 mm in thickness followed the disease-specific survival Kaplan-Meier graph, which identifies survival over 10 years for patients in intervention versus control arms of the SLNB trial (Figure 3). There was not even a signal for a melanoma-specific survival advantage at 10 years.
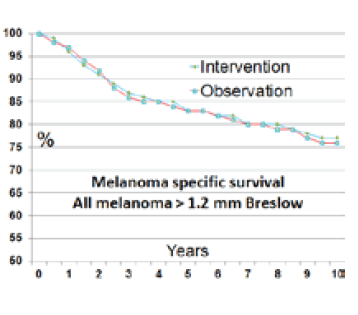
Figure 3. Kaplan-Meier graph
General practitioners may choose to reflect on their referral practices. SLNB might be available but it should no longer be considered part of routine management.
Complication rate3
The complication rate for SLNB was 10%. Most complications were minor and transient, including haematoma, wound infections and transient seroma. However, serious and prolonged complications, including nerve damage and lymphoedema, can occur. The complication rate was 37% for CL. These larger block nodal dissections have a greater risk of more serious complications.
Disease-free survival
There was a greater likelihood of remaining disease-free for a longer period of time in the SLNB group. Of patients with intermediate thickness melanoma, the difference was 71% versus 65% at 10 years. This difference was apparent at 2 years into follow up but never translated into a survival benefit. This is probably not a true benefit. It is an inevitable consequence of the trial design, given the CL group no longer had the immediate regional lymph nodes for melanoma to re-present. As such, the continuing value of this secondary endpoint finding is unclear and difficult to interpret.
SLNB and subsequent CL may seem logical, but melanoma is different from most cancers. When squamous cell carcinoma is present in nodes it could be negligent not to offer further dissection. An evidence-based shift away from this traditional paradigm is now required for melanoma.
SLNB as a predictor of survival
SLNB was confirmed as a major predictor of survival for those patients with a melanoma of 1.2–3.5 mm thickness. The hazard ratio (HR) for being SLNB positive was 2.64 (P <0.001).1 Many patients with intermediate-thickness melanomas may wish to know this added prognostic information.
The three other survival predictors highlighted in this study were Breslow thickness, (HR 1.62 per mm), ulceration (HR 1.4) and site on the trunk (HR 1.42). Of course you don’t need an added operation with a complication rate of 10% to obtain these three survival predictors.
Melanoma patients should no longer be routinely encouraged to have further lymph node resections if their SLNB test is positive. SLNB provides additional prognostic advice for patients with melanoma who choose to be tested.
Comment
As with all RCTs, the key data are the analyses on an intention-to-treat basis. Subgroup or exploratory analyses of the original data may provide novel insights or generate new questions but are potentially problematic. They cannot alter the findings regarding the studies’ primary hypotheses and aims, or the result derived from the primary outcome measure.
Summary
SLNB is confirmed as a prognostic test but does not influence melanoma-specific survival at 10 years. SLNB-negative patients still need regular review and can still die from their melanoma. SLNB-positive patients should be carefully counselled regarding the risks and very limited benefit (if any) of proceeding to CL.
SLNB should not be regarded as essential or standard of care for patients with melanomas. SLNB should be discussed with patients with intermediate-thickness melanoma. Note that ‘discuss’ is not the same as ‘offer’ or ‘recommend’.
The discussion should include:
- SLNB does not alter survival prospects
- SLNB provides added accuracy to survival prognostic figures
- 70% vs 90% for melanoma Breslow thickness of 1.2–3.5 mm
- SLNB has a 10% complication rate
- If positive, no added treatment can be offered that has demonstrated survival benefits
- this includes no apparent survival benefit in proceeding to CL
- SLNB-positive patients can be offered enrolment into further melanoma studies.
Competing interests: None.
Provenance and peer review: Commissioned; externally peer reviewed.