Researchers, supported by United States (US) Food and Drug Administration guidelines,4 rely on limited primary endpoints: extent of skin involvement in area, non-progression of the area involved, indirect clinical signs of sepsis such as fever, and biochemical markers (such as C-reactive protein [CRP] and white cell count [WCC]). But there are difficulties with all of these endpoints.5 Many patients who start hospital treatment no longer have fever. Non-progression of area refers to the time from presentation and initiation of treatment until there is no further visible extension in the surface area of skin directly involved in the infection. In clinical practice this results in the widespread use of indelible ink markers. In many cases, progression of area has either already stopped or stops soon after treatment.6 Spellberg argues that non-progression is not improvement.7 Severity can proceed dramatically within a non-extending area of infection, and conversely, extension of erythema can sometimes be caused by activity or limb dependency in a resolving infection.
There have been other attempts to categorise severity of skin infection.8–12 These classifications mostly dismiss clinical signs as not objective enough, and by extension, clinical signs have less impact when deciding appropriate therapy. None have ever been validated in clinical studies, as they appear cumbersome and difficult to apply.
However, skin is an uncommon organ in that it allows direct inspection of its inflammatory response to infection. Severity and complications, including superficial or deep necrosis, tenderness, erythema and heat can be directly observed. Clinicians still rely on these direct findings to determine severity and treatment. Direct inspection has not been used in standardised measures of severity and outcome of SSTI due to poor validity and reliability in the classification of visual and thermal cues.
This study seeks to explore a new, simple method for assessing the severity of SSTI, and a primary end-point response to treatment. Skin surface temperature has not been previously reported. This study sought to explore the difference in skin temperature between skin affected by cellulitis and the non-affected site. The aims were to explore:
- Whether there is a significant change in baseline skin surface temperature difference and temperature difference upon presentation with SSTI
- Whether changes in skin surface temperature difference reflect clinical progress.
Method
A prospective observational clinical study of patients admitted with SSTI for intravenous antibiotic therapy.
Setting and patients
All consecutive patients treated with intravenous antibiotic therapy with a discharge diagnosis of SSTI from two ‘Hospital in the Home’ (HIH) units from May to October 2011 were included.
Inclusion criteria
- All consecutive patients referred from the emergency department, short stay unit, or directly from the community by another doctor, for treatment of acute cellulitis with intravenous antibiotic therapy
- A subsequent discharge diagnosis of cellulitis
- This study was conducted through the HIH services of two large city hospitals. Patients were suitable for home-based acute care, and were admitted to hospital under the respective HIH service. Inclusion criteria for HIH were: the need for intravenous antibiotic therapy, no need for injected narcotic, no uncontrolled sepsis, and the ability to manage the bathroom without assistance.
Exclusion criteria
Patients were excluded from this study if they had cellulitis of the face, head or trunk, if they had bilateral cellulitis, or if they had no contralateral limb. Surgical wound infections were excluded. Cessation of treatment for any reason other than a medical assessment that no further intravenous therapy was required, resulted in exclusion of the patient. Patients who did not have cellulitis as their discharge diagnosis were excluded. Incomplete temperature data recordings resulted in exclusion.
Temperature measurement
This study measured the surface temperature of skin. Temperature was measured using a standard device – the Digitech Compact Infrared Thermometer with Dual Laser Targeting. This is a non-contact thermometer with an objective measurement protocol. The device can be adjusted for thermal emissivity, and in this study the thermal emissivity was set at the recommended score for human skin (0.98). Identical devices were used in this study.
Baseline
Ideally, we would have sought the difference in skin temperature between the affected and non-affected limb without the presence of a SSTI. This was not practical in the timing of the conduct of this trial. Thus, a proxy was necessary for this baseline measure.
We measured skin temperature in both limbs of the alternate limb girdle (ALG) to establish an internal, self-matching baseline for the difference in temperature between non-affected limbs. Thus, in a patient with lower limb SSTI, skin temperature was measured in the two upper limbs, at standard reference points, and the difference in temperature calculated. This is assumed to be the difference in SSTI affected limbs before the onset of disease.
This was recorded once, at the first attendance by nursing staff.
Limb affected with SSTI
Skin temperature was measured daily at the point of maximum heat on the affected limb and at the corresponding point on the non-affected limb. Based on clinical experience, we assumed that the point of maximum heat could alter from day to day.
Temperature measurement was taken within 24 hours of the onset of hospital treatment, and daily thereafter until the day of discharge. Nursing staff attending the patient measured and recorded the temperatures each day. They also recorded type of intravenous therapy, core temperature, pulse, and blood pressure each day.
In the HIH units involved, HIH medical staff attended each patient daily to assess progress and supervise management. HIH medical staff also decided the day of discharge, which coincided with cessation of intravenous antibiotic therapy. HIH medical staff were blinded to the temperature readings recorded by nursing staff.
Baseline
Data including blood tests (C-reactive protein, white cell count) and results of microbiological investigations such as swabs and blood cultures were collected from the hospital databases or records on discharge and included for analysis. However, no extra testing was specified for the purposes of this study. This was a naturalistic design, and data was taken where the variables were found.
Baseline data were recorded for each patient, derived from the medical record or direct observation. These included:
- Patient age and gender
- Diabetes
- Immunosuppression
- Treatment prior to admission
- Presence of trauma, bite or ulcer
- White cell count
- C-reactive protein
- Microbiology sampling (results of)
- Blood culture (results of)
- Outcome data and other severity variables (other than limb temperature) included:
- Length of stay (nights of stay)
- Core temperature (°C) measured daily
- Heart rate (beats per minute) measured daily
- Blood pressure measured daily.
Analysis
- The analysis was performed using Stata 10 (StataCorp, College Station, TX, USA).
- The descriptive information is presented as N (%) for categorical data and mean (SD) for continuous data.
- Temperature differences were assessed using the paired t-test. Multivariate regression analysis was used to determine factors associated with the temperature change.
Results
Study participants
Eighty-four patients were enrolled and after 21 patients were excluded, 63 patients with completed datasets were included in the study (Table 1). The mean age was 49.5 years (19–91 years). Forty-two (66.7%) of the patients were men and twenty-one (33.3%) were women. The SSTI affected the lower limb in 47 patients (74.6%), and the upper limb was affected in 16 patients (25.4%). In 20 patients (31.7%), there was a history of trauma or a bite, and eight patients (12.7%) had a pre-existing ulcer. Thirty-four patients (54.0%) had taken oral antibiotics prior to presentation. There were 12 patients (19.0%) with diabetes among the study population, and three were insulin dependent. Three patients (4.8%) were on immunosuppressive drugs for the treatment of inflammatory disease.
Table 1. Reasons for exclusion in patients enrolled in study
| Reason for exclusion | Number |
|---|
| Incomplete data collection |
7 |
| Discharge diagnosis – abscess/infected haematoma |
4 |
| Discharge diagnosis – inflammatory arthritis/synovitis |
4 |
| Discharge diagnosis – surgical wound |
2 |
| Patient self discharged against advice |
1 |
| Discharge diagnosis – septic arthritis |
1 |
| Diagnosis – urticaria |
1 |
| Bilateral cellulitis |
1 |
| Total exclusions |
21 |
Investigations
A serum white cell count was taken in 58 patients (92.1%), with a mean of 10.7 (range 4.5–25.2). CRP was taken in 47 patients (74.6%). The mean CRP was 78.1 (range 1–311). Blood cultures were taken in four patients (6.3%) and none were positive. Swab of the wound for microscopy and culture was taken in 15 patients (23.8%) and a result was obtained in 12 patients (80% of those who had a swab taken, 19% of all subjects). Methicillin-sensitive Staphylococcus aureus was found in eight cases, methicillin-resistant S. aureus in two cases, proteus mirabilis in one case, mixed Gram-negative organisms in one case, and no growth in three cases.
Treatment
Thirty-five patients (55.6%) were treated with either daily or twice-daily cephazolin. Twenty-seven patients (42.9%) were treated with ceftriaxone. One patient (1.6%) was treated with flucloxacillin.
Ten patients (15.9%) required a treatment change: two patients were commenced on oral ciprofloxacin; two patients had their dose of intravenous cephazolin increased; two patients were started on intravenous gentamycin; one patient was switched to cephazolin; one to intravenous clindamycin; one to teicoplanin; and one patient was commenced on oral metronidazole.
Six patients (9.5%) had a complication recorded. Five developed superficial blistering necrosis of the skin, and one patient experienced temporary hallucinations. All patients completed their treatment.
Length of stay
Four patients had a stay in hospital prior to HIH of 1 night each. Patients stayed in HIH between two and 15 nights. The total stay in HIH was 312 days and the total length of stay was 316 days (a mean of 5.0 days).
Temperature difference
The ALG temperature was checked once (at day one). No difference between left and right ALG temperature (31.5° C (1.7) vs 31.6° C (1.7) respectively, p=0.555) was identified. Thus, rather than use temperature difference, the average temperature for ALG was calculated and used during the data analysis (Table 2).
Table 2. Comparison between first and last day observations
| | Day 1 | Last day | p |
|---|
| | Mean (SD) | Mean (SD) | |
|---|
| Affected limb temperature (°C) |
34.4 (2.0) |
32 (1.9) |
<0.001 |
| Unaffected limb temperature (°C) |
30.9 (1.9) |
29.9 (2.0) |
0.002 |
| Difference between affected and unaffected limb temperature (°C) |
3.5 (1.9) |
2.1 (1.9) |
<0.001 |
| Alternative limb – right |
31.6 (1.7) |
|
|
| Alternative limb – left |
31.5 (1.7) |
|
|
| Alternative limb – mean |
31.5 (1.6) |
|
0.555* |
| Pulse (beats per minute) |
78.7 (13.5) |
72.7 (11.2) |
<0.001 |
| BP (mmHg) |
|
|
|
|
|
125.5 (14.6) |
122 (13.2) |
0.090 |
|
|
72.4 (11) |
72 (9.6) |
0.777 |
| Core temperature (°C) |
36.4 (0.7) |
36.0 (0.4) |
<0.001 |
| *p value for difference between left and right alternate limb girdle |
Table 2 reveals that the difference between affected and unaffected limb was 3.5 (95% CI 3.0–3.9) at day one and 2.1 (95% CI 1.7–2.6) on the last day. That demonstrates a 1.4° C (95% CI 0.7–1.9) change from day one to the last day in the difference between affected and unaffected limb. At day one, the affected limb was 2.9° C warmer compare to ALG (95% CI 2.4–3.4, p<0.001) and there was a small, statistically significant but not clinically important difference between ALG and unaffected limb (mean difference 0.6° C, 95% CI 0.1–1.2).
Between day one and the last day, there was a significant reduction in affected limb temperature (mean reduction of 2.4 °C, 95% CI 1.9–3.0, p<0.001) and small drop in unaffected limb temperature (mean reduction of 0.9 °C, 95% CI 0.3–1.5). The affected limb was warmer compared to both the unaffected and alternative limb at day one (Figure 1) and remained warmer compared to unaffected limb on the last day.
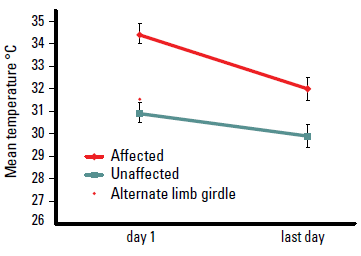
Figure 1. Changes in skin surface temperature between SSTI affected and non-affected limbs
Figure 2 examines the variation in skin surface temperature by day of admission. There is a statistically significant decrease in temperature change between the affected and unaffected limb between day one and day two (–1.1° C (95%CI -1.6 – -0.7), and between day two and day three (-0.6° C (95% CI -1.1 – -0.1). There were no significant decreases in temperature from day four until day seven. The number of study subjects decreased from day three onwards, and this will influence statistical significance.
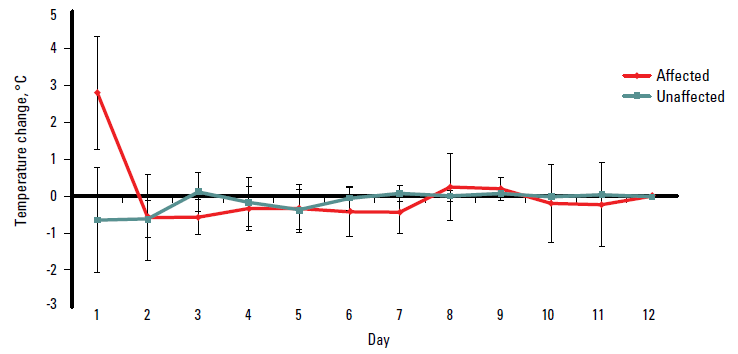
Figure 2. Mean daily temperature changes of SSTI affected and non-affected limbs (with 95% CI)
Other variables
There was no change in patients’ blood pressure (BP) (p=0.090 for systolic and p=0.777 for diastolic) and core temperature (p=0.067), but there was a significant drop in patients’ pulse (mean change six beats per minute [bpm]), 95% CI 3.0–9.0, p<0.001) (Table 2).
Further analysis
There was no correlation between temperature change/temperature differences and patients’ age, gender, type of treatment or length of stay.
Discussion
This study demonstrated that a significant skin surface temperature difference exists in patients on presentation with SSTI. It also showed that the skin surface temperature difference between SSTI affected and non-affected limbs was significantly higher on presentation than at discharge. Although the affected limb in SSTI on day of discharge was significantly cooler than the day of presentation, the temperature difference had still not returned to the baseline.
With additional validation, the measurement of skin surface temperature may be a useful and objective primary end-point measure of the severity of SSTI and of the progress of the illness.
This measure offers some improvement on the current published approaches to classifying SSTI severity. It does not rule out the role for signs of severe or uncontrolled sepsis in determining severity of illness, but those factors may be overstated in current classifications and may not assist clinicians who manage cellulitis of a moderate severity.
While heart rate held some value in assessing the severity of SSTI, the non-illness baseline in an individual patient is not known at the time of presentation, so it can be difficult to interpret. Indirect measures such as core temperature and systolic blood pressure did not significantly change through the course of an admission for SSTI in this study. However, in most patients, core temperature was taken once or twice daily and it is possible that spikes in temperature were missed. While this probably would have ensured that core temperature was a significant indicator of severity in SSTI, it is possible that skin temperature does not demonstrate those fluctuations, and this may be taken as another advantage over core temperature.
The SSTI study population was similar to that found in many hospitals. The fact that the study occurred in a HIH setting implies only that these patients did not have severe uncontrolled sepsis or uncontrolled pain. The proportion of patients with cellulitis treated in medically managed HIH is growing and we believe that treatment of SSTI in HIH is, or soon will be, the standard option in all hospitals.13 It would be possible to duplicate this study among patients with SSTI who have severe uncontrolled sepsis and require admission to an intensive care unit (ICU). It would also be of interest to repeat this study in patients with SSTI managed in primary care with oral antibiotics.
This finding opens other possibilities. As found in this population, most patients with SSTI do not have positive microbiology. Future correlative work between temperature measurement and patient subgroups with positive microbiology might help us understand whether micro-organisms demonstrate a different temperature difference profile to others – for example, whether the temperature differential in streptococcal disease may be different to that in pseudomonal disease or methicillin-resistant S. aureus.
The device used in this study was selected because of its size, portability, ease of use and relatively low cost, and is readily available to most health services.
There are many limitations to our study. Not everything that causes heat in a limb is cellulitis and we do not propose this is the case. Temperature difference is not pathognomonic of SSTI, and nor is temperature change over the course of treatment is not restricted to SSTI. Cellulitis is still a clinical diagnosis.
Not all cellulitis occurs in limbs, and some patients have no comparator limb. Further, some of the observed non-conforming results could have been caused by a variety of confounders including: the ambient temperature; the time of day the reading was taken; whether the patient was in bed or sitting out of bed; clothed or not; whether a stocking was used; the season; and other environmental causes for temperature differences. However, the use of the opposite limb’s temperature measurement should have controlled for many of these confounders.
SSTIs have been neglected over a long period, despite its prevalence in human disease. Ironically, we believe that the almost unique capacity to directly observe an organ in distress, and the self-evident changes that occur, has been the basis for this neglect. Research study into this field has been limited by this self-evidentiary nature of outcome measurement. We hope we have assisted in the development of further evidence in the study of SSTI.
Competing interests: None.
Ethics approval: This study was assessed as low risk by the institutional ethics committees of both hospitals involved in the study (Epworth Hospital and Royal Melbourne Hospital). Verbal consent was obtained from patients included in the study.
Funding: This study was supported by a 2011 Epworth Research Institute Small Grant award. Epworth Research Institute has had no role in the preparation of this report.
Provenance and peer review: Not commissioned; externally peer reviewed.
Acknowledgements
The authors wish to acknowledge Mr John Rimmer and Ms Alexandra Gorelik for their contribution to this study.