Barriers to the use of warfarin in AF include advancing age, patient reluctance or refusal, inconvenience of regular blood tests for the International Normalised Ratio (INR) of prothrombin time, dose adjustments and regimen complexity, risk of falls and risk of bleeding.6,11,12
In a Canadian examination of 125 000 patients initiated to warfarin for AF, 9% did not fill the first repeat prescription, while 32% and 43% had discontinued within 12 and 24 months respectively.7 Of 16 000 patients in the United States hospitalised with non-valvular AF, 43% of warfarin users had discontinued within 12 months.8 In a separate United States examination of 2 457 patients hospitalised with ischaemic stroke, 34% had discontinued warfarin within 12 months,9 and of 278 patients with AF, 38% had discontinued within 12 months.9 Based on Australian Pharmaceutical Benefits Scheme (PBS) data for the period 2003–2006, of 93 000 patients newly started on warfarin, 16% had discontinued within 6 months and 43% had discontinued within 24 months.10 But an unspecified proportion of these patients would have been using warfarin for indications other than AF.10 In the present report we have analysed medication persistence in Australian patients with presumed AF who had been newly started on warfarin therapy between October 2006 and September 2007.
Methods
Data source
A longitudinal analysis was performed on PBS pharmacy payment claims in a 10% random sample of the Australian population, the data being drawn from de-identified records held by Medicare Australia. Warfarin is priced below the general patient co-payment threshold and is only recorded for patients classified as ‘concessional’. Hence, this study is restricted to patients described as long-term concessional (no general prescription during the previous 5 years). We selected a group of patients who initiated warfarin during the time window October 2006 to September 2007 and followed patients through September 2009. They could not have been dispensed warfarin in the preceding 12 months. They also needed to have received an anti-arrhythmic drug (disopyramide, flecainide, amiodarone, sotalol) in the prior 12 months and hence they were ‘likely’ patients with AF.
Persistence analyses
Treatment persistence was assessed from October 2006 to September 2009. Treatment cessation was defined as 3 months without a warfarin prescription, suggesting at least 40 days without treatment. Using the Kaplan–Meier method, we generated persistence curves for warfarin. Patients who had not discontinued warfarin were censored at 30 September 2007, and no death information was available. Key outcomes were:
- proportion failing to fill first repeat prescription;
- median persistence time on medication; and
- long-term persistence at 33 months.
The PBS administrative database does not provide clinical information on reasons for discontinuation.
Results
The claims database yielded information on 1108 patients newly prescribed warfarin (representative of 11 080 patients nationally). Mean age was 74 years, 50% were females and 65% of prescriptions were initiated by general practitioners (who in some instances would have been continuing an initial hospital discharge prescription). More than 70% of patients were taking two or more strengths of warfarin.
The persistence curve for likely AF patients prescribed warfarin is presented in Figure 1. The curve indicates that 15% (95% CI: 13–17) failed to collect the first repeat prescription, median persistence time was 12 months (10–13), and long-term persistence to 33 months was 26% (23–27).
Discussion
There is ample clinical evidence demonstrating the benefit of warfarin in prevention of thromboembolic complications in patients with non-valvular AF.3–5 Yet patient persistence with warfarin, as shown in Figure 1, appears to be poor, a truly unfortunate situation given the significant morbidity and mortality associated with embolic stroke.2 The problem of poor persistence with chronic medication is not unique to warfarin: similar persistence issues have been reported in relation to prescriptions for statins and anti-hypertensive drugs.7–9,13,14
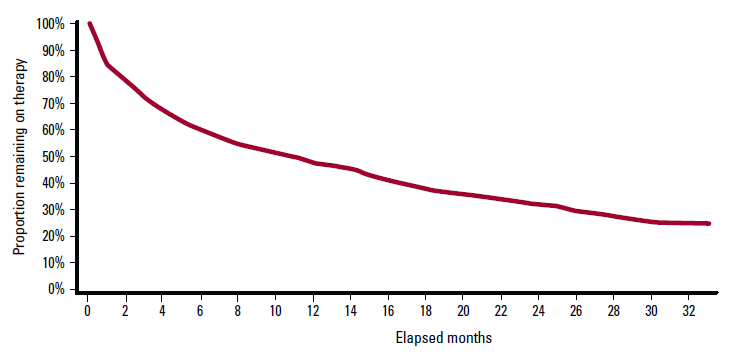
Figure 1. Persistence curve for patients initiated to warfarin
After allowance for potential differences in methodology, the discontinuation rates reported in this paper are broadly in line with the North American experience,7–9 yet they seem to be at variance with local data published by the Australian Institute of Health and Welfare (AIHW) for the period 2003–2006.10 While also using a 90-day cessation period, the AIHW report indicates that 14% of patients filled only one prescription for warfarin, the present analysis was similar at 15%. However, a careful review of Tables 8 and 9 in the AIHW report strongly suggests that patients filling only one prescription were excluded from the long-term analysis. If we apply the same approach to the present analysis, median persistence time now becomes 21 months, a finding highly compatible with the previous Australian data (which in itself included patients with any indication for anticoagulation, not just AF).
Patients and doctors have difficulty with warfarin therapy for a variety of reasons, including the need for frequent blood tests, variations in INR, dose adjustments, confusion from the use of multiple and changing doses, the potential for drug–drug and drug–food interactions, risk of falls and the risk of bleeding.6,11,12 Some discontinuation of warfarin may be medically indicated rather than just patient non-compliance. This is often in the presence of bleeding in the elderly.15 The PBS administrative records do not disclose the reasons for discontinuation.
In the face of non-compliance, a number of approaches might potentially improve this situation, including:
- taking warfarin exactly as prescribed
- having blood tests exactly as ordered
- making dose changes exactly as instructed
- increased use of anticoagulant clinics, where available
- not stopping or starting new medicines (including over-the-counter [OTC] products) without discussion with doctor
- making no major changes to diet
- limiting daily alcohol consumption to 1–2 standard drinks/day.
There are limitations in the present analysis. It is restricted to long-term concessional patients, and we have made the assumption that patients using an anti-arrhythmic drug who are then initiated to warfarin most likely are manifesting AF. Hence, the findings are generalisable to patients with more chronic non-valvular AF who could not be successfully reverted to sinus rhythm, where applicable. We have no information on any patients who may have died during the follow up period. It could be argued that a 180-day cessation period might need to be considered, rather than 90 days, because patients receive a 50-tablet pack and are generally using two or more strengths of warfarin. While all the quoted studies have used 90 days cessation, recalculation of our persistence data employing the longer cessation period produced similar broad conclusions of poor persistence.
If and when poor persistence with warfarin relates only to non-compliance rather than to serious side effects, we will be wasting valuable resources and a golden opportunity for stroke prevention will be missed.
Implications for general practice
- Long-term persistence with warfarin is unsatisfactory.
- Early intervention is desirable and will require relevant educational materials.
- Behavioural changes have been suggested which may assist with the problem.
Competing interests: The study was funded by Boehringer Ingelheim Pty Limited, Sydney, NSW. Raw data for the study were supplied by Medicare Australia. All authors have completed the ICMJE form. Michael Ortiz has received payment from Boehringer Ingelheim for preparing an assessment of warfarin compliance for a PBAC review. He has, until recently, received payment for consultancy from several pharmaceutical companies. Leon Simons has received payment from several pharmaceutical companies for board membership and lectures, all unrelated to this study.
Leon Simons contributed to this study in an honorary capacity, Michael Ortiz and Gordon Calcino were paid consultants to Boehringer Ingelheim Pty Ltd; Peter Germanos is an employee of that company.
Gordon Calcino has received payment for consultancy from Boehringer Ingelheim as well as various other pharmaceutical companies.
Ethics approval: Patient identities remained totally anonymous during this investigation and ethics approval was obtained from the Medicare Australia Ethics Committee.