Despite these statistics, dermatology training in medical schools in Australia is poor by international standards and extremely variable.3 Some medical graduates will have received no training in skin examination and skin cancer diagnosis. International medical graduates typically receive more dermatology training but may never have seen a skin cancer before coming to work in Australia. The ethnic diversity in Australia also means that GPs working in metropolitan inner city clinics may see few patients with skin cancer.
Many doctors learn dermatology and skin cancer medicine at a postgraduate level and this training is generally ad hoc rather than a coordinated program. Many have become expert in performing skin checks, others feel anxious when asked to conduct a skin check. A number of factors contribute to this anxiety, including confusingly named benign skin lesions, the variability in clinical presentation of skin cancer, concern about missing a skin cancer, desire to reduce the number of benign skin lesions excised, and limited access to specialist back-up, especially for those working in rural and remote settings.
Despite years of highly successful public health education from state based cancer councils, thousands of Australians die each year from melanoma due to a delay in diagnosis.4 These deaths could potentially be prevented by GPs performing appropriate skin checks.
Current guidelines
A 'skin check' can be defined as a comprehensive assessment and examination of an asymptomatic patient for any evidence of skin cancer. Current Australian guidelines advise against general population screening for skin cancer, citing the lack of evidence for the feasibility of organised screening and the effectiveness of screening in reducing mortality. Patient self examination with opportunistic screening is the current standard.5–7
While early diagnosis and treatment reduce melanoma mortality and nonmelanoma skin cancer morbidity, Cancer Council Australia argues against patients having regular or annual skin checks on three grounds: nonmelanoma skin cancer has a low mortality, melanoma frequency does not justify a mass population screening program on economic grounds, and the instrument that would be used for mass screening (GP assessment) is not sufficiently accurate.
However, many patients self select for skin cancer screening with their GP, a skin cancer clinic or a dermatologist. Patients with higher levels of education, from higher socioecomonic groups and people living in major urban centres tend to be over-represented among those presenting for skin checks. There is inequity in access to care for people from low socioeconomic groups and those living in rural areas.
Evidence for systematic skin checks
Recent evidence demonstrates the feasibility and effectiveness of systematic skin cancer screening. In 2003, the SCREEN project (Skin Cancer Research to Provide Evidence for the Effectiveness of Screening in Northern Germany) carried out 360 288 skin checks over a 12 month period in the German state of Schleswig-Holstein, examining 19% of the eligible population of that state. Participating doctors received an 8 hour training program. A total of 3103 malignant skin tumours were identified. Melanoma incidence increased by 34% during the year of the study – many of these smaller and thinner than previously identified lesions. Five years after SCREEN, melanoma mortality was reduced by over 50%.8 These findings were only reported this year.
Northern Germany has a low skin cancer prevalence compared to Australia. Between 1999 and 2006 there were just over 50 000 skin cancers registered in Schleswig-Holstein among a population of 2.8 million (one skin cancer per 56 people). In the corresponding period there were 4 469 361 NMSCs and approximately 70 000 melanomas in Australia among a population of 21 million (one skin cancer per 4.6 people). Based on the tenfold higher prevalence of skin cancer in Australia, the anticipated benefits of systematic skin cancer screening are likely to be even greater in Australia.
Guidelines on performing skin checks require review as new information becomes available. In view of the magnitude of the problem of skin cancer in Australia, implementation of any changes to the current guidelines will require careful analysis of cost, planning and implementation.
Current realities of practice
Regardless of the current recommendations, patients do present to GPs for regular skin checks and unscheduled, opportunistic skin checks are difficult within the time constraints of general practice. Patients who self select for skin checks are generally at high risk and frequently have skin cancers.8
General practitioners are free to determine their own practices with respect to skin checks. If GPs have capacity in their practice to undertake skin checks, feel confident and are interested then they should consider promoting annual skin checks to patients at high risk, and less frequent skin checks to patients at medium risk. Table 1 provides a ranking of risk for GPs wanting to be more systematic about when to offer skin checks.
Apart from facilitating early skin cancer diagnosis, the skin check is an opportunity to assess the patient's individual risk, educate them on skin self examination and early detection of skin cancer, and to modify their behaviour in the sun and reinforce the primary prevention message of 'slip, slop, slap'.
Table 1. Assessment of skin cancer risk
| High risk (3 monthly self examination and 12 monthly skin check with doctor) |
|---|
- Red hair
- Type 1 skin and age more than 45 years
- Type 2 skin and age more than 65 years
- Family history of melanoma in a first degree relative in patients aged more than 15 years
- More than 100 naevi (more than 10 atypical naevi)
- Past history of melanoma
- Past history of nonmelanoma skin cancer or more than 20 solar keratosis
|
| Medium risk (3–6 monthly self check and 2–5 yearly skin check with doctor) |
|---|
- Blue eyes
- Type 1 skin and age 25–45 years
- Type 2 skin and age 45–65 years
- Type 3 skin and age more than 65 years
- Family history of NMSC
- Past history of solar keratosis
- Multiple previous episodes of sunburn
|
| Low risk (annual self check and one-off skin check with doctor for assessment of risk and advice regarding skin care) |
|---|
- Type 1 skin and age less than 25 years
- Type 2 skin and age less than 45 years
- Type 3 skin and age less than 65 years
- Type 4 and 5 skin
|
Suggested skin check procedure
There are many ways to conduct a skin check. Table 2 provides a framework for GPs who have not yet developed their own approach.
Table 2. A framework for conducting skin checks
- Take a targeted history that includes previous skin cancers, previous excisions, family history, occupation, level of sun exposure and frequency of sun protection
- Ask the patient to nominate all the spots that they (or their partner) are concerned about
- Ensure good lighting and privacy for the examination
- Allow privacy for undressing. Give the patient clear instructions to leave their underwear on, and provide a gown or sheet for the patient to wear
- Have the patient sit in the centre of the couch for the beginning of the examination
- Use a systematic approach that covers all body parts with particular attention to sun exposed areas
- Lightly palpate each lesion for any additional clues
- Examine every lesion in turn with particular attention to any 'different' lesions
- Have the patient seated while examining the hands, arms and face
- Ask the patient to lie face down to examine their back and legs
- Ask the patient to roll over so that you can examine the patient's chest, abdomen and legs
- Palpate the liver and lymph nodes if following up a previous melanoma
- Ask about lesions of concern on the scalp or those concealed by underwear. Only examine these areas with verbal consent from the patient
|
The role of dermatoscopy
Dermatoscopy is an important diagnostic tool in both melanoma and nonmelanoma skin cancer and can enhance diagnostic accuracy. Learning to use a dermatoscope follows a J curve, with diagnostic accuracy of skin cancer being reduced in new adopters of the dermatoscope. Practise is very important.
It is useful to make a provisional diagnosis and treatment decision before dermatoscopic examination. In the author's experience, revision of the provisional diagnosis following dermatoscopy most often leads to excision of benign lesions. Sometimes it helps to avoid excision of a benign lesion; rarely does it help discover an unsuspected skin cancer. For more detailed information about dermatoscopy see the article by Rosendahl et al in this issue of Australian Family Physician.
The role of medical photography and computer assisted diagnosis
Medical photography is increasingly popular and there is some evidence to support its use in high risk patient populations to enhance early diagnosis of melanoma.9 However, the photographic appearance of benign naevi are also known to evolve over time and change in the appearance of a mole is not pathognomonic for melanoma.10 Photographs can reassure patients that a complete examination has been performed, provide a baseline to identify new primary melanomas in the distant future, supplement medical records and help educate patients. However, photographs may provide a false sense of security, diminish diagnostic accuracy and delay excision of melanoma – particularly if 'technicians' perform the principal examination with the doctor reviewing photographs or when changes in serial photographs are used to diagnose melanoma. If a lesion is sufficiently worrisome to photograph it may be best to excise it.
Technological issues are common and the author's preference is to take the photograph with the patient's own mobile telephone or print the photograph and hand it to the patient rather than rely on clinic storage and retrieval.
However, a labelled photographic report can be very useful to facilitate communication between GPs and dermatologists (Figure 1). Photography is also useful for patients with multiple atypical naevi where baseline full body photography can assist serial clinical examination and minimise excisions.
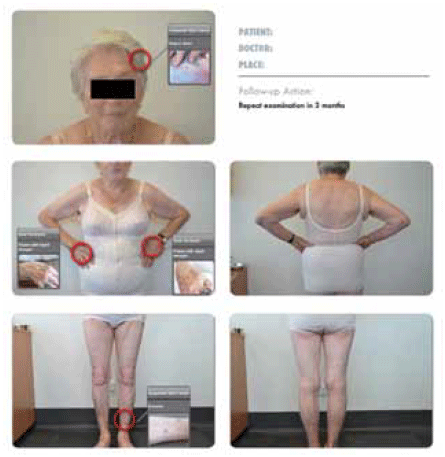
Figure 1. Skin report. A series of skin photographs with individual lesions labelled with their diagnosis to facilitate communication of diagnosis to patients and their referring doctor
Diagnosis of skin cancer
Two-thirds of all skin cancers are basal cell carcinomas (BCCs), 30% are squamous cell carcinomas (SCCs), 1.5% in situ melanomas and about 1% are invasive melanomas. The remaining 1% is made up of about a dozen very rare conditions.11 For simplicity, keratoacanthoma and Bowen disease can be considered as variants of SCC.
Site of the lesion
Nodular BCCs occur most commonly on the face, trunk and shins; superficial BCCs on the forearms, trunk and legs; and morpheoic BCCs on the nose and cheeks. Squamous cell carcinoma is common on the hands, forearms, ears, lips, scalp, forehead and shins; invasive melanoma is most common on the trunk and legs; and in situ melanoma is most common on the face and scalp.
Most lesions will have appeared within the past 12 months and been present for at least a month. As a rule of thumb, most cuts/scratches/lesions that are going to heal will have done so within a month.
Next steps
Simply put, there are only three possible diagnoses for a skin lesion: clearly malignant, clearly benign and too close to call.
- If you are 100% sure that the lesion is a skin cancer then arrange for it to be removed. Each GP knows the limits of their surgical skill and should refer as necessary
- If there is a specific diagnosis and you are certain that the lesion is benign, then no treatment is required
- If you are only 99% sure that the lesion is benign, a definitive diagnosis is required. Options include diagnostic biopsy, excisional biopsy or referral to a dermatologist. Access to specialists varies and patients usually find it disconcerting to wait weeks or months for a definitive diagnosis for a 'suspicious lesion'. Hence, GPs often excise lesions they feel are low (but not zero) risk. Consequently the number of lesions needed to be excised for every skin cancer removed is approximately 20 for melanoma and three for NMSCs. Some public hospitals and specialist centres provide rapid assessment clinics for GP referred patients with suspicious lesions.
Three useful rules of thumb
- If the history and examination are discordant, ignore the history
- If a lesion has been previously biopsied and found to be benign, but you are still concerned, biopsy it again
- If the patient leaves the room but you are still concerned about a lesion you did not biopsy, call them back.
Interpreting pathology reports
When a pathologist examines a histological slide of a skin lesion there are also only three possible diagnoses: clearly malignant, clearly benign and too close to call.
If it is clearly benign or clearly malignant the report is generally succinct and the bottom line will contain few words. If it is a melanoma you should receive a detailed synoptic report.
If the lesion is too close to call the report will often be wordy, ambiguous, have a number of caveats and leave you confused. Call the pathologist to discuss the report. They may recommend review by a specialist dermatopathologist. Occasionally the experts fail to reach a consensus and these types of lesions need to be managed as if they are a skin cancer. If the lesion is a suspected melanoma, re-excision of the scar and multidisciplinary review is recommended.
Common benign lesions encountered during skin checks
A clear understanding of benign lesions can reduce confusion and unnecessary excisions during skin checks. The top 10 benign lesions are:
- solar keratosis (Figure 2)
- diffuse superficial actinic porokeratosis (Figure 3)
- seborrhoeic keratosis (and the benign lichenoid keratosis variant) (Figure 4)
- benign melanocytic naevus (junctional, compound or intradermal) (Figure 5)
- chondrodermatitis nodularis helices chronicus (Figure 6)
- blue naevus (Figure 7)
- benign fibrous papule (Figure 8)
- sebaceous hyperplasia (Figure 9)
- capillary haemangioma (Figure 10)
- dermatofibroma (Figure 11).
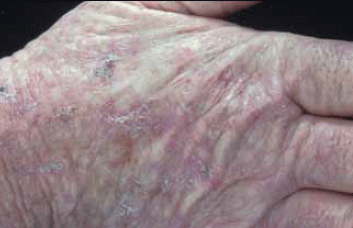
Figure 2. Solar keratosis
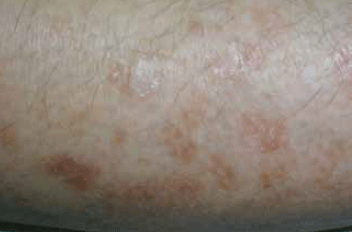
Figure 3. Diffuse superficial actinic porokeratosis
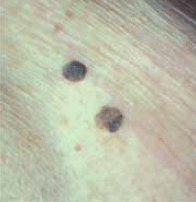
Figure 4. Seborrhoeic keratosis
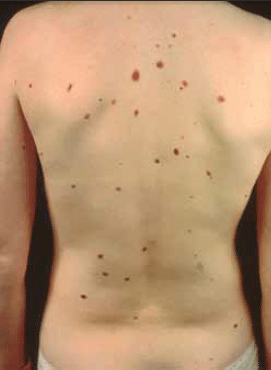
Figure 5. Benign melanocytic naevus
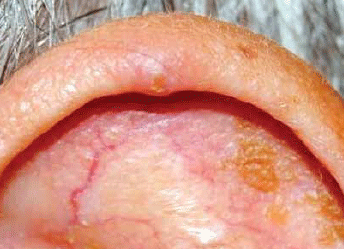
Figure 6. Chondrodermatitis nodularis helices chronicus

Figure 7. Blue naevus
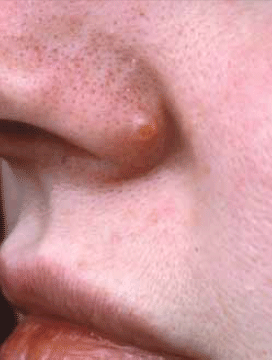
Figure 8. Benign fibrous papule
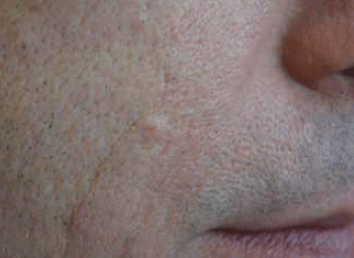
Figure 9. Sebaceous hyperplasia

Figure 10. Capillary haemangioma
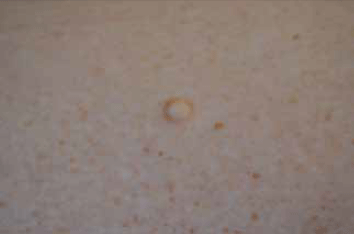
Figure 11. Dermatofibroma
Solar keratoses are usually flat, red and scaly and feel like sandpaper. By definition they are less than 10 mm in diameter. Induration may occur on the backs of the hand and forearms. Tenderness on lateral pressure requires a biopsy to exclude SCC. Solar keratoses can be pigmented with no pigment network on dermatoscopy. A biopsy may be required to exclude in situ melanoma.
Diffuse superficial actinic porokeratosis (DSAP) tend to occur on the shins and forearms. They are often mistaken for solar keratosis and treated with liquid nitrogen – this highlights their distinctive thread-like circumferential elevation of the outer border.
Seborrhoeic keratoses are usually easy to diagnose. They are multiple, large brown, well demarcated, waxy spots that look like they are stuck onto the skin. When they are flat, pink or inflamed (benign lichenoid keratosis) they can be mistaken for BCC or in situ melanoma. Skin cancers may arise within a seborrhoeic keratosis. A superficial shave biopsy is helpful if the diagnosis is uncertain.
Benign melanocytic naevi are not present at birth; first appearing around the age of 5–10 years as flat pigmented macules. The macules progressively increase in number until the age of 15–20 years. When the melanocytes are confined to the junction between the dermis and epidermis they are called 'junctional naevi'. They become raised after 5–10 years while retaining their pigmentation as the melanocytes proliferate and migrate deeper into the dermis. When melanocytes are found in the junction and the deeper dermis they are called 'compound naevi'. Eventually the melanocytes at the junction disappear leaving only melanocytes in the deeper dermis. These intradermal naevi then persist 10–20 years before eventually disappearing altogether.
Chondrodermatitis nodularis helices chronicus presents as a painful nodule on the ear helix. There may be a central crust. Biopsy may be required to exclude SCC.
Blue naevus are often present for many years and are noticed incidentally on a routine skin check. They are deep blue rather than black, perfectly round and no pigment network is seen with dermatoscopy. New, large or rapidly enlarging lesions may require a biopsy to exclude nodular melanoma.
Benign fibrous papules are commonly mistaken for nodular BCC. They tend to occur on the nose and chin. They are firm on palpation. They rarely bleed spontaneously. Dermatoscopy can be used to exclude BCC as they lack telangiectasia.
Sebaceous hyperplasia tends to occur on the forehead, nose and upper cheeks. They are yellow or faintly orange. They look like mini-BCCs, but they do not grow over time and are often multiple. Dermatoscopy is useful as the lesions lack telangiectasia in their border and the central punctum is more easily seen.
Capillary haemangioma are usually pink. If thrombosis occurs they can turn black overnight. Examination with a dermatoscope shows distinctive blood filled lobules and allows the colour to be clearly seen.
Dermatofibroma present as pink or brown dermal nodules. Diagnosis is confirmed by rolling the lesion between the fingers; they feel like a split pea.
Limitations of skin checks
No technique provides 100% accuracy in skin cancer diagnosis. Amelanotic melanoma remains very hard to diagnose. Patients should be advised that if a new lesion appears or an existing lesion changes after their skin check, they should attend for re-examination.
Summary
Although current guidelines do not recommend routine skin checks, GPs often find themselves providing skin checks and evidence is growing for systematic screening. General practitioners should have a skin check routine that ensures they thoroughly examine all lesions. Dermatoscopy can assist in performing skin checks but its use is dependent on experience. Although common, the role of clinical photography is limited, reserved mainly for monitoring of patients with numerous atypical naevi. It is important to reach a diagnosis for each lesion and diagnostic biopsy should be performed where the diagnosis is uncertain. Specialist back-up is important and GPs should be familiar with how to access appropriate services in their area.
Conflict of interest: none declared.