Echocardiography is appropriate for use in patients with known or suspected cardiac pathology. Findings on TTE can provide valuable diagnostic and prognostic information that may impact significantly on clinical management. However, an understanding of the significance of such findings is necessary in order to trigger appropriate clinical decision making or to implement evidence based therapies.
Echocardiography should not generally be used as a screening tool in asymptomatic patients in primary care. However, as is the case in other imaging modalities, there is the potential for incidental findings in the patient referred for an unrelated indication or with symptoms out-of-keeping with the degree of reported abnormality.
This article discusses the impact of TTE findings on patient management using seven case studies as examples.
Case study 1
An asymptomatic 76-year-old man is found to have a soft systolic murmur on routine examination. The rest of his cardiovascular examination is normal. Apart from his age, he has no significant risk factors for cardiovascular disease. He is referred for a TTE, which shows that his left ventricular systolic function is depressed, with a globally reduced ejection fraction of 30%, but no valvular pathology.
Discussion
Systolic or contractile function of the left ventricle is quantified by measuring left ventricular ejection fraction (LVEF). An LVEF above 50% is regarded as normal, with degrees of severity being mild (40–50%), moderate (30–40%) or severe (<30%). A reduction in LVEF is common in older populations. In the Canberra Heart Study, Abhayaratna and colleagues2 observed that LVEF was reduced in 6% of subjects aged 60–86 years. Half of these cases had no symptoms of heart failure. These results indicate that symptomatic heart failure with reduced ejection fraction (HF-REF) represents only the ‘tip of the iceberg’ in terms of the prevalence of significantly impaired left ventricular function. In older patients, therefore, a reduction in LVEF could occasionally be an incidental finding in those referred for reasons other than heart failure symptoms. Left ventricular systolic dysfunction is a common endpoint of a range of cardiac pathologies. A list of potential causes is shown in Table 1.
Table 1. Causes of global left ventricular systolic dysfunction
- Ischaemic heart disease
- Viral myocarditis
- Idiopathic dilated cardiomyopathy
- Infiltrative – haemochromatosis, amyloidosis
- Diabetes
- Drug-induced/toxins, including alcohol
- Familial cardiomyopathy
- Connective tissue disease
- Peripartum
|
Patients presenting with symptomatic HF-REF are at increased mortality risk due to progressive heart failure or ventricular tachyarrhythmias, despite advances in pharmacological and device management.3 In addition, patients with no heart failure symptoms and a reduction in LVEF are at risk of developing symptomatic heart failure and an associated increase in mortality.4
The finding of a reduced LVEF in a patient presenting with shortness of breath confirms the diagnosis of HF-REF. In these cases, standard heart failure therapy should be initiated.5,6 In addition, in patients incidentally found to have a reduced ejection fraction of <50%, an angiotensin converting enzyme inhibitor (ACEI) should be commenced in order to prevent progression to symptomatic heart failure, as this reduces mortality over the long term.7,8 In patients who progress to symptomatic heart failure, a long-acting beta-blocker should also be commenced.5,6
Case study 2
A 60-year-old man with a past history of type 2 diabetes and hypercholesterolaemia presents with shortness of breath. He describes no other cardiac or respiratory symptoms. He takes metformin 850 mg twice per day and atorvastatin 20 mg/day. Cardiorespiratory examination is normal and full blood examination and urea, creatinine and electrolytes are also normal. His electrocardiogram (ECG) shows inferior T wave inversion. He is referred for a TTE, which shows an old inferior myocardial infarct with scar formation and a normal LVEF. A stress echocardiogram reveals hypokinesis in the territory of the left anterior descending artery following exercise. A coronary angiogram shows triple vessel disease and the patient is referred for coronary artery bypass surgery.
Discussion
Regional variation in left ventricular wall motion, either at rest or during stress, is strongly suggestive of ischaemic heart disease. There may be areas of hypokinesis (decreased myocardial thickening), akinesis (no thickening), dyskinesis (paradoxical motion) or prior infarction with scar formation. Patients with ischaemic heart disease may present with classic angina pain, atypical features or, as in Case 2, an ‘anginal equivalent’ such as exertional dyspnoea. Patients may also present with heart failure symptoms due to an ischaemic cardiomyopathy. Resting TTE may be helpful in detecting very significant regional wall motion abnormalities such as previous infarction, but stress echocardiography is far more sensitive in detecting areas of myocardial ischaemia.
Approximately one-quarter of myocardial infarctions remain unrecognised9 and may be an incidental finding on TTE at a later stage. These ‘silent’ myocardial infarctions are more common in the setting of diabetes10 and hypertension.11 Regardless, the prognosis of unrecognised myocardial infarction is similar to symptomatic infarction9 and standard treatment for ischaemic heart disease (including aspirin, statin, beta-blocker and ACEI therapy) should be commenced.
Case study 3
An 80-year-old woman presents with two episodes of heart palpitations in the past week. She states that they felt irregular and rapid but denies other cardiac symptoms. Abnormal findings on examination include an irregular pulse and a hyperkinetic apex beat, which is undisplaced. She has a history of hypertension treated with telmisartan, hydrochlorothiazide and amlodipine. Full blood examination, thyroid stimulating hormone and urea, creatinine and electrolytes are normal. A 12-lead ECG confirms atrial fibrillation with a rapid ventricular rate of 120 bpm with no other significant abnormalities. A TTE reveals concentric left ventricular hypertrophy with normal LVEF.
Discussion
Transthoracic echocardiography can detect cardiac pathology resulting from chronic, suboptimally controlled hypertension, including left ventricular hypertrophy and increased left ventricular mass. Mild left ventricular dilatation or diastolic abnormalities may also be present. Atrial fibrillation is also common in the setting of hypertensive heart disease (as in Case 3) or other structural heart disease. Therefore, an echocardiogram may be indicated to evaluate for cardiac pathology associated with atrial fibrillation.
Left ventricular hypertrophy, either by voltage criteria on a 12-lead ECG or measured by echocardiography, is associated with an increased risk of symptomatic heart failure and mortality.12 In a patient with a history of hypertension, left ventricular hypertrophy should trigger more aggressive treatment of hypertension to achieve recommended target levels.13 Regression of left ventricular hypertrophy is associated with a decrease in cardiovascular events14 and heart failure hospitalisations.15
In Case 3, a beta-blocker would serve dual functions as an additional antihypertensive agent as well as rate control for atrial fibrillation. A decision regarding the need for anticoagulation depends on clinical, not echocardiographic, parameters (see Resource).
Symmetrical hypertrophy on echocardiography may also occur in the setting of aortic stenosis, which would be seen on the same scan. However, increased left ventricular wall thickness in the absence of a history of hypertension or aortic stenosis, and usually with marked diastolic abnormalites, may suggest cardiac amyloidosis, an infiltrative disorder with a poor prognosis. In this case, or in cases where the aetiology remains unclear, referral to a cardiologist is indicated. Cardiac magnetic resonance imaging (MRI) and endomyocardial biopsy may be necessary to clarify the diagnosis.
Asymmetric left ventricular hypertrophy, often with dynamic outflow tract obstruction, suggests a diagnosis of hypertrophic obstructive cardiomyopathy. This finding should also trigger cardiology review, for detailed evaluation of risk factors for sudden cardiac death, treatment of symptoms and genetic counselling for family members.
Different causes of increased left ventricular wall thickness are illustrated in Figure 1.
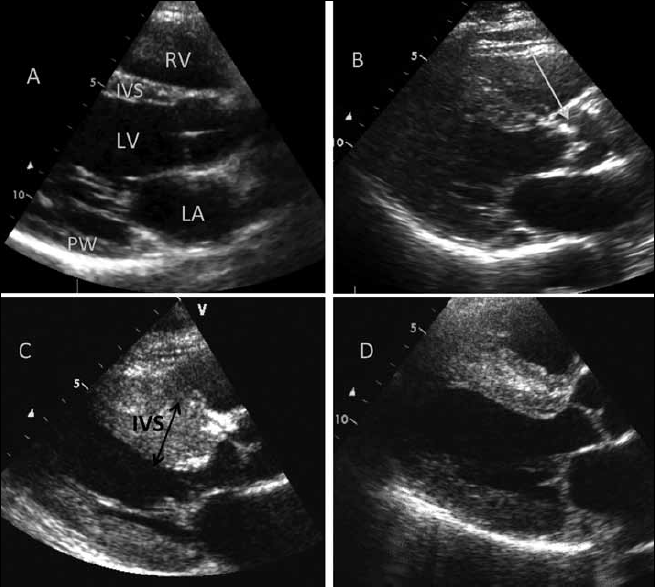
Figure 1. Causes of increased left ventricular (LV) wall thickness
A) Normal wall thickness (IVS = interventricular septum; PW = posterior wall) in parasternal long axis view of the LV;
B) Severe calcific aortic stenosis (arrow) causing pressure overload with LVH;
C) Hypertrophic cardiomyopathy: asymmetrical thickening of the septum (IVS);
D) Amyloid cardiomyopathy: symmetrical wall thickening in the absence of hypertension suggests infiltrative disease
Case study 4
An 80-year-old man presents with exertional shortness of breath. There are no other current cardiac or respiratory symptoms but he has a past history of ischaemic heart disease, smoking and diabetes. He takes enalapril 5 mg/day, metformin 500 mg twice per day and aspirin 100 mg/day. Cardiorespiratory examination and full blood examination and urea creatinine and electrolytes are normal. ECG shows left atrial enlargement but no significant ST or T wave changes. He is referred for TTE, which reveals a normal LVEF with moderate diastolic dysfunction and a dilated left atrium. There is evidence of a pseudonormal diastolic filling pattern and increased filling pressures. The patient was also referred for spirometry, which revealed a mild fixed obstructive defect suggestive of mild chronic obstructive pulmonary disease (COPD).
Discussion
Establishing a diagnosis of heart failure with preserved ejection fraction (HF-PEF) can be challenging. Although a normal LVEF on an echocardiogram is a requirement for the diagnosis, demonstration of significant abnormalities of diastolic function or their relationship to symptoms is often more difficult.
Even for experienced echocardiologists, the assessment of left ventricular relaxation and filling during diastole is challenging, and understanding of this complex process is still in evolution. Mild abnormalities of diastolic function are reflected in the ‘impaired relaxation pattern’, which is often seen in the setting of ischaemia, hypertension and diabetes and in people over the age of 60 years.16 Indeed, in the Canberra Heart Study,17 24% of the cohort exhibited mild diastolic abnormalities in the setting of a normal LVEF. The finding of mildly impaired diastolic function should therefore be regarded as a relatively normal finding in older asymptomatic patients.
In contrast, moderate to severe abnormalities of diastolic filling are less common, found in 6% of Canberra Heart Study subjects with normal LVEF.17 Such abnormalities are reported on TTE as a ‘pseudonormal’ or ‘restrictive’ pattern, usually with an increased Tissue Doppler E/E ratio >15. These patterns reflect significant diastolic dysfunction with elevated left atrial pressure and are more likely to explain dyspnoea.
However, significant diastolic dysfunction may not always be seen on resting TTE.18 Evidence of increased filling pressures16 and, occasionally, alternative causes of dyspnoea such as dynamic left ventricular outflow tract obstruction, may only be apparent after stress.18 As in Case 4, respiratory causes such as COPD should also be considered before diagnosing HF-PEF, as the cause of dyspnoea in a former smoker. Referral to a cardiologist may be necessary if, as is frequently the case, there is some question over the diagnosis. A combination of a number of tests, including chest X-ray, echocardiography, natriuretic peptides and respiratory function may need to be considered before making a diagnosis.
Prognosis is poor in patients presenting with HF-PEF.19 Any abnormalities of diastolic function on TTE are associated with an increased risk of heart failure and increased mortality within the general population,20 which may reflect the impact of underlying conditions such as hypertension, diabetes and ischaemia on prognosis. However, there is as yet no evidence of a mortality benefit from pharmacological intervention for this condition, although improvements in exercise tolerance may be seen.21 Treatment of patients with abnormalities of diastolic function is generally directed toward control of ischaemia, optimisation of blood pressure and glycaemic control in diabetes, as well as managing comorbidities such as COPD.
Case study 5
An 84-year-old woman is found to have a systolic murmur audible over the aortic area on routine cardiac examination. She says she is not short of breath on exertion, but admits she is fairly inactive. She does not describe any other cardiac or respiratory symptoms. A TTE reveals moderate calcific aortic stenosis with normal LVEF (mean pressure gradient 35 mmHg, aortic valve area of 1.1 cm²).
Discussion
The finding of a previously undocumented cardiac murmur should prompt evaluation for suspected valvular disease. Symptomatic patients found to have at least moderate valve pathology should be referred to a cardiologist for further evaluation. In an asymptomatic patient (such as in Case 5), monitoring involves periodic clinical and TTE re-evaluation and education of the patient to recognise the early symptoms of heart failure. However, in patients with significant valvular disease who deny any symptoms but are fairly inactive, stress echocardiography may be useful in determining whether the patient is truly asymptomatic.
Regurgitant lesions tend to be better tolerated than stenotic valvular disease, and are generally associated with a more benign clinical course. Milder degrees of valvular dysfunction are associated with a better prognosis than severe lesions and progress slowly over some years. Echocardiography may therefore be performed at greater time intervals in patients with milder degrees of valvular dysfunction than in those with more severe valvular disease (Table 2).22 Development of symptoms should prompt re-evaluation.
Table 2. Appropriate use of echocardiographic monitoring for valvular disease22
| | Lesion | Appropriateness |
|---|
|
Native valvular stenosis
|
Mild stenosis without change in clinical state or cardiac examination
|
Routine surveillance >3 yearly (appropriate)
|
|
Moderate/severe stenosis without change in clinical state or cardiac examination
|
Routine surveillance >1 yearly (appropriate)
|
|
Native valvular regurgitation
|
Mild regurgitation without change in clinical state or cardiac examination
|
Routine surveillance >3 yearly (uncertain)
|
|
Moderate/severe regurgitation without change in clinical state or cardiac examination
|
Routine surveillance >1 yearly (appropriate)
|
Although some authors advocate early surgical intervention for subjects with asymptomatic severe valvular pathology,23 surgery is generally not performed until a patient develops symptoms or evidence of ventricular dysfunction. Close echocardiographic monitoring is useful in identifying echocardiographic parameters which are associated with adverse outcomes.
Case study 6
An asymptomatic 34-year-old Afghan refugee reports a possible history of rheumatic fever as a child and is referred for a TTE. The TTE shows mild pulmonary hypertension with an estimated right ventricular systolic pressure of 45 mmHg and normal right ventricular function. There is no evidence of rheumatic valvular disease. Left ventricular systolic and diastolic function appear normal.
Discussion
Pulmonary hypertension may be caused by cardiac or respiratory pathology. When identified during an echocardiogram, the sonographer will seek evidence of specific cardiac causes such as left-sided valvular disease, diastolic dysfunction and intracardiac shunts. If no cardiac causes are detected on TTE, respiratory causes such as COPD and chronic pulmonary emboli or idiopathic pulmonary arterial hypertension should be considered.
Case study 7
A 25-year-old asymptomatic woman from India is referred for echocardiography following a chest X-ray for immigration purposes, which revealed cardiomegaly. On TTE she is found to have right ventricular dilatation with normal right ventricular function. Colour flow imaging demonstrates an atrial septal defect with significant left to right shunting.
Discussion
The finding of dilatation of the right ventricle, with or without pulmonary hypertension and right ventricular dysfunction, suggests volume overload as a cause. In young patients, echocardiographic evaluation should be undertaken, with particular targeting of the interatrial septum to search for an atrial septal defect with shunting. Patients found to have an atrial septal defect with significant left to right shunting (with an elevated ratio of pulmonary to systemic circulation, Qp:Qs >1.5) and right ventricular enlargement should be referred for percutaneous or surgical closure.24 A more common finding of a patent foramen ovale may be seen in 25% of the population and may be regarded as a normal variant.
Right ventricular systolic dysfunction may occur as a result of respiratory disease (cor pulmonale) or pulmonary hypertension of any cause. It may also be involved as part of a myocardial infarction or a generalised dilated cardiomyopathy. Primary right ventricle cardiomyopathies such as arrhythmogenic right ventricular cardiomyopathy are much less common.
Resource
Samardhi H, Santos M, Denman R, Walters D, Bett N. Current management of atrial fibrillation. Aust Prescr 2011;34:100–4. Available at www.australianprescriber.com/ magazine/34/4/100/4.
Conflict of interest: David Prior has received project grant support from the NHMRC; has served on advisory boards for Servier and Actelion; has received funding for lectures by Boehringer Ingleheim, CSL, Merck Sharp & Dohme, Astra Zeneca and Novartis and funding for travel by Sanofi and GlaxoSmithKline. Duncan Campbell has received research support from the NHMRC and NHF and has received funding for lectures from The Australasian Renin Academy.
Acknowledgement
St Vincent’s Institute of Medical Research is supported in part by the Victorian Government’s Operational Infrastructure Support Program.