The prevalence of chronic disease increases with age, and older people have much to gain from medicines that delay disease, cure disease or manage symptoms. However, older people are also at increased risk of adverse drug events. With increasing age, comes increasing inter- and intra-individual variability. Therapeutic aims, pharmacokinetics, pharmacodynamics, safety and efficacy vary significantly between individuals of the same age and change over time in older patients. Therefore, it is important to review all medicines appropriately in older patients to achieve relevant outcomes.1
Robust and frail older adults
In considering the potential benefits of medicines, it is important to differentiate robust from frail older adults. There are several objective definitions for frailty, such as the frailty phenotype (≥3 of unintentional weight loss, exhaustion, weakness, slow walking, low physical activity) and accumulation of medical, functional or social deficits.2–4 Robust older people are usually mobile and functionally independent, and medications are used to prevent and treat illness, alleviate symptoms and delay functional decline and mortality. In contrast, frail older people have reduced resilience to external stressors, are often less mobile, more functionally dependent, and may reside in a residential aged care facility. In frail older people, medical therapy may be predominantly aimed at relieving symptoms and maintaining function.5
Risks from medicines in older adults include adverse drug reactions, hospitalisation, functional impairment, geriatric syndromes (eg. confusion, falls, incontinence, frailty) and mortality.6 These risks increase with patient characteristics such as age, multiple comorbidities, low body weight, frailty and impaired hepatic or renal function. Risks are also associated with specific medicines exposures such as polypharmacy, drug-drug interactions, drug-disease interactions, drug-geriatric syndrome interactions,7 drugs with anticholinergic and sedative properties,8 anticoagulants, and antiplatelet and hypoglycaemic agents.9
Optimising medication use
Assessment of an older patient's medical therapy requires clear understanding by the doctor and patient of the therapeutic aims, and the potential and actual benefits and risks of the patient's medications. Key steps in optimising therapy in the older patient are outlined in Figure 1. Treatment guidelines are often based on clinical trials in young or robust older patients and must be applied with caution to frail older adults. Furthermore, guidelines rarely provide recommendations for patients with comorbidities.10
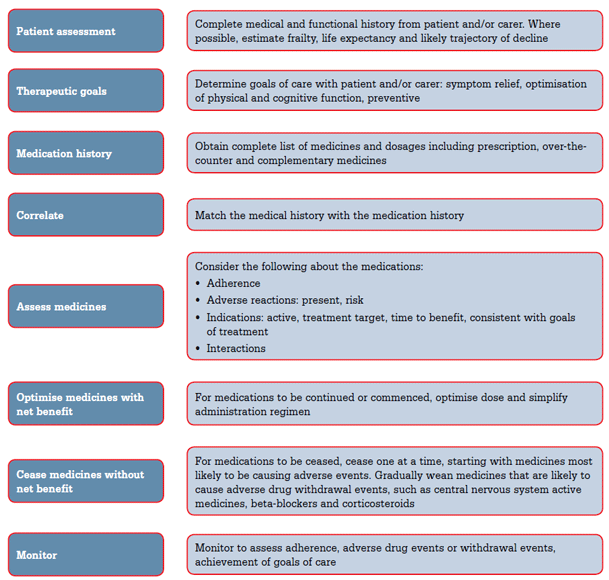
Figure 1. Key steps in optimising an older patient's medical therapy11,35
Understanding the available evidence
There is little evidence that many drug classes commonly prescribed for older patients reduce mortality.11 This may be due to multiple 'competing risks' for mortality,12 changes in pathophysiology, pharmacology and the increasing impact of adverse events of medicines.11
There is robust evidence to show major risks from polypharmacy and adverse drug reactions in old age.6,9,13 The limited evidence on deprescribing in older people suggests that it can be done safely.14–18 While efficacy is best assessed by randomised controlled trials (RCTs), adverse drug events are better assessed by observational studies and are more frequent in clinical practice than in RCTs.19
Estimates of benefits and risks of commonly used drug classes in general practice are summarised in Table 1 and described below.
Table 1. Estimates of benefits and risks of commonly used medication classes in older people
| Medication | Benefit | Study population* | RR, OR, HR (95% CI) | NNT | NNH |
|---|
| Statins |
Secondary prevention of all cause mortality |
≥60 years24–26 |
RR=0.85 (0.78–0.93) |
28 to prevent death over 5 years |
10–20 to experience myalgia |
| Antihypertensives |
Secondary prevention of all cause mortality |
≥60 years28 |
RR=0.90 (0.84–0.97) |
84 to prevent death over a mean time of 4.5 years |
Two to experience any overall adverse events over a mean time of 4.5 years |
| |
Secondary prevention of
all-cause
mortality |
≥80 years28 |
RR=1.01 (0.90–1.13) |
|
|
| Aspirin |
Primary prevention of all cause mortality |
Mean age of 57 years20 |
OR=0.94 (0.88–1.00) |
120 to prevent one cardiovascular event over 6 years |
73 to experience a non-trivial bleed over 6 years |
| Bisphosphonates |
Zoledronic acid after hip fracture prevention of mortality |
≥50 years (84% of participants aged over 65 years)38 |
HR=0.75 (0.58–0.97) |
27 to prevent death over 3 years |
18 to experience pyrexia, 45 for myalgia, 45 for bone pain and 53 for musculoskeletal pain over 3 years |
| |
Zoledronic acid after hip fracture prevention of mortality in nursing home subgroup |
A sub-study of: ≥50 years (84% of participants aged over 65 years)38 |
HR=1.02 (0.51–2.05) |
|
|
RR = relative risk. The risk of developing an outcome in the group receiving the trial medication compared to the group not receiving the medication. RR <1, the outcome is less likely to occur in the medication group than in the control.
RR >1, the outcome is more likely to occur in the medication group
OR = odds ratio. The OR measures the ratio of the odds of an event (disease or death) occurring from specific causative agent to the odds of the event not occurring. The OR gives an idea of how strongly dependant the outcome is on the supposed causative agent. OR <1 suggests that the agent reduced the odds of the event occurring
HR = hazard ratio. The ratio of the chance of events (usually death) occurring in the treatment group as a ratio of the hazard occurring in the control group of a trial. HR <1 means that death is less likely to occur in a given time period in the treatment group than in the control
CI = confidence interval. This is a calculated range of the margin of error
NNT = number needed to treat. The number of patients who need to be treated for the stated time period for one patient to experience the stated benefit
NNH = number need to harm. The number of patients who need to be treated for the stated time period for one patient to experience the stated adverse event
* Most data is from robust older participants in randomised trials. When available, data from nursing home patients is included separately |
Antiplatelets and antithrombotics
Data from RCTs suggests that when prescribing aspirin for primary prevention of cardiovascular disease, we need to treat 120 people for 6 years to prevent one event (number needed to treat [NNT] = 120). However, there will be one major bleed among 73 people over the same time (number needed to harm [NNH] = 73).20 Similar NNH are observed in RCTs of aspirin in patients with atrial fibrillation.21 However, an observational study of warfarin use in frail adults over 70 years of age with atrial fibrillation found a higher likelihood of harm: for every six people treated for 6 months one had a major bleed.22
Statins
Lipid lowering drugs are taken by over 40% of Australians aged over 65 years.23 In older patients, the best evidence for statins is for secondary prevention of cardiovascular events and death in patients with coronary heart disease.24,25 In this setting, a doctor would need to treat 28 patients for 5 years to prevent one death (NNT=28). The cost per quality associated life year (QALY) in secondary prevention are $15 000 to 24 000 for all age groups (45–85 years). However, for primary prevention of coronary heart disease, cost per QALY increases from $26 000 to 90 000 at 65 years to $56 000 to 169 000 at 85 years.26
General practitioners may consider withdrawal of statins when potential benefits are no longer clinically relevant. In patients with severe physical or cognitive impairments or those in their last year of life, reducing the risk of vascular events or mortality may no longer be important. Statins may also be withdrawn if frail older patients have significant symptoms or signs consistent with adverse effects in a temporal pattern associated with statin use.27
Antihypertensives
A recent Cochrane review of antihypertensive (mainly thiazides) use in adults over 60 years of age with moderate to severe systolic or diastolic hypertension, showed reduction in overall mortality, cardiovascular mortality and cardiovascular morbidity in healthy people aged 60–80 years. However, among patients 80 years of age or over, there was limited data showing no reduction in total mortality.28 Several prospective, open label studies of withdrawal of antihypertensives in patients over 65 years of age found that this can be done safely without withdrawal effects. Overall, 20–85% of subjects remained normotensive 6 months to 5 years after cessation, with no increase in mortality.14
Sulfonylureas
The best evidence that sulfonylureas reduce microvascular complications of diabetes mellitus comes from the 20 year United Kingdom Prospective Diabetes Study (UKPDS), which recruited patients with a median age of 53 (+/– 8.6) years at baseline.29 Risks of hypoglycaemia increase with age and national guidelines recommend higher target blood sugar levels for older patients.30
Antibiotic use
Detection and treatment of infection is important and clinically effective in older patients. Infections are a major cause of death in old age, and not only affect the infected site, but also precipitate geriatric syndromes such as confusion and falls. Dosing of antibiotics, and all medicines, in frailty is complicated by reduced lean body weight, impaired hepatic and renal function31 and pharmacodynamic factors (increased risk of adverse events, impaired immune responses).5,11 Dose adjustment, monitoring of drug levels or clinical response may be required. Frail older adults in a pre-terminal state may choose not to treat infections with antibiotics.
Ethical principles
The ethical principles of beneficence, nonmaleficence, autonomy and justice can be considered in guiding medication use in older adults, both on an individual and a population scale.32
The effectiveness of medicines can be considered a marker of beneficence, the duty to do good. Only a small proportion of medications, such as analgesics, can be monitored clinically in individual patients. For other drugs, individual assessment of benefit is limited to surrogate markers or is not possible. Therefore assessment may rely instead on the likelihood of doing good, which is derived from the results of clinical trials that may not be generalisable to frail older people.19 Adverse drug reactions are good examples of avoidable (nonmaleficence) harm, they increase with frailty and age, and may present atypically as geriatric syndromes.5 There is also increased harm from polypharmacy6 and the 'prescribing cascade', where drugs are prescribed to treat the side effects of others.33
Respect for autonomy may be a challenge when patients are not competent to consent to treatment due to cognitive impairment. Carers – acting as decision makers – may be in conflicted positions. For example, the treatment of behavioural disturbances in dementia arguably accrues the risks of adverse events from medications to the patient, while the benefits reside chiefly with the carers. Consideration of numbers needed to treat and harm from research trials may be helpful so that patients can weigh up the risks in keeping with their personal priorities.34
The principle of justice denotes fair, unbiased treatment on the basis of medical need. In Australia, age-based economic rationing of medicines does not occur directly on a population level. However, some individuals and communities of older patients on multiple medications may not be able to afford them despite government subsidies.35
Management strategies
General practitioners are best able to understand the complete medical, functional and social issues that are in play when optimising medications. A multidisciplinary approach involving doctors (GPs and specialists), pharmacists and nurses, is the common feature of successful interventions to reduce polypharmacy.36,37 Nevertheless, this is a time-consuming, ongoing activity.
General practitioners may consider a medication review after significant transitions such as moving into residential aged care, hospitalisation or a new diagnosis. Medication review by a pharmacist, or a structured interview performed by the practice nurse during which the patient (or carer) brings all their medications, will assist the GP in compiling information from multiple sources.
General practitioners may also need to liaise with specialists to ensure agreement on therapeutic goals. Where reaching concordance is difficult, a second opinion from another GP or a geriatrician may help.
Patients and their families need to be reassured that medicines are being changed to achieve their therapeutic goals. It must be clear that if drugs are ceased it is because they are causing harm or are of no benefit, and not because the patient is 'not worth treating'. Close monitoring of, and discussion with, the patient throughout the period of medication change will determine whether goals are being met, detect any adverse events, and demonstrate to the patient that the change is part of an active treatment plan.
Key points
- Individual therapeutic goals and priorities influence the choice to continue or commence medications in primary care.
- Polypharmacy and drug related adverse effects may cause significant harm to older patients.
- GPs can safely deprescribe medications with patient and multidisciplinary support.
Conflict of interest: none declared.