Prostate cancer is the most common non-cutaneous cancer diagnosed in Australian men, accounting for 30% of all new cancer diagnoses. In 2011, 13% of all male cancer deaths were due to prostate cancer.1 The incidence of prostate cancer is rising because of a high uptake of prostate-specific antigen (PSA) screening combined with increasing life expectancy. Multiple treatment options exist and the appropriate treatment choice depends on clinical and biopsy-proven pathological factors.2 Accurate pre-treatment diagnosis is essential to determine appropriate management.
Until recently, there was a lack of a suitable imaging modality for reliable detection of significant tumours in prostate cancer. New evidence suggests multi-parametric magnetic resonance imaging (mpMRI) has the potential to improve diagnosis and treatment of prostate cancer.
Issues with the current method of prostate cancer diagnosis
Screening for prostate cancer remains controversial. This is because of the poor sensitivity and specificity of PSA, and the issue of over-treating men with non-life threatening tumours.3 The current diagnostic technique requires trans-rectal ultrasound (TRUS)-guided biopsy prompted by an abnormal digital rectal examination (DRE) and/or increased PSA. DRE is a crude tool with low positive predictive value and high inter-observer variability.2 Although PSA levels correlate with prostate cancer risk, no threshold value of PSA provides an acceptable combination of sensitivity and specificity.4
When prostate cancer is suspected, the diagnosis must be confirmed with biopsy. This is typically a 12-core TRUS biopsy.5 Most prostate cancers are not visible on TRUS.6 The sampling nature of TRUS biopsy means that it is often by chance that the cancer is detected. TRUS biopsy will miss over 20% of all prostate cancers at the time of biopsy.7 An additional problem is undersampling of the anterior prostate, apex and midline, meaning many men are falsely reassured they are free of cancer.8 Transperineal biopsy provides access to all areas of the prostate but comes with increased costs and requires a general anaesthetic.9 The pick-up rate of transperineal biopsy is equivalent to a saturation TRUS biopsy, but has extremely low risk for infection.
In men with a raised PSA, 70–80% of initial biopsies are negative because of the sampling nature of TRUS and the limitations of PSA. Thus, more men are subjected to biopsy than is necessary.6 Although typically performed under local anaesthetic,TRUS biopsy has an appreciable complication rate and a 1–2% risk of sepsis, despite antibiotic prophylaxis.
Confounding the diagnostic pathway further is inaccuracy of grading on TRUS biopsy. It is well established that 30–40% of low-risk cancers diagnosed on TRUS are upstaged or upgraded in men who procede to radical prostatectomy.10 The goal of the urologist is to distinguish ‘clinically significant’ prostate cancers, which require treatment, from indolent cancers, which can be observed.
What is multiparametric MRI?
Prostate MRI using T1- and T2-weighted images was trialled in the 1980s but lacked sensitivity and specificity to justify use.11 Technical improvements together with the addition of functional parameters have improved accuracy. mpMRI combines the anatomical images of T1- and T2-weighted imaging with two or more functional sequences (Table 1).12 It is the only imaging modality with the spatial resolution and soft-tissue contrast necessary to accurately characterise localised prostate cancer. The recent availability of higher-field strength 3-Tesla magnets has reduced acquisition time and provides superior anatomical definition, compared with 1.5-Tesla.12 Endorectal coils were used with 1.5-Tesla systems but are not required in 3-Tesla systems, reducing cost, time and patient discomfort. A recent review suggests mpMRI reliably detects clinically significant prostate cancer and provides information regarding tumour location, volume, grade and stage.11
Table 1. Description of multiparametric MRI sequences
|
|
T2-weighted imaging (T2WI)
|
- Provides high-resolution images that clearly define prostate anatomy
- Normal peripheral zone is characterised by intermediate–high signal intensity due to its high water content
- Cancer has low signal intensity due to its dense cellularity, although this is not specific for cancer
- Gives the best assessment of prostate margins for extracapsular extension, seminal vesicle invasion, neurovascular bundle and adjacent organ involvement
- Reduced accuracy for cancer in the transitional and anterior zones as the baseline T2 signal is lower and BPH nodules are common here
- T2WI alone does not have sufficient sensitivity and specificity for the localisation of prostate cancer, hence, functional sequences are also required
|
|
T1-weighted Imaging
|
- Helps differentiate post-biopsy haemorrhage from tumour
|
|
Diffusion-weighted imaging (DWI)
|
- Measures the diffusion of water molecules through tissue
- Prostate cancer has a reduced diffusion of water, compared with normal prostate, due to its tightly packed cells
- Apparent diffusion coefficient (ADC) maps of the prostate are derived
- Interpretation must be combined with T2WI
- Sensitivity and specificity of DWI and T2WI combined is 85–90% when compared with radical prostatectomy findings
|
|
Dynamic contrast enhanced imaging
|
- A T1 sequence is run followed by a bolus of intravenous gadolinium followed by a rapid sequence of scans
- Malignancy causes changes such as increased blood flow, neo-vascularity and leaky capillaries
- Perfusion of a region of interest can be plotted against time to create a perfusion vs time curve.
- Three types of curve have been defined:
- Type 1: suggests normal prostate tissue
- Type 2: suggests BPH or prostatitis
- Type 3: suggests high grade prostate cancer
|
|
Magnetic resonance spectroscopy
|
- A functional technique that indirectly measures metabolite levels in the prostate by region of interest
- Cellular concentrations of choline and creatine increase in prostate cancer and correspond to volume and grade
- The concentration of citrate decreases as tumour volume and grade increase
- Technically challenging and time consuming, and often not included in a mpMRI protocol
|
|
BPH, benign prostatic hyperplasia
|
Reporting has been standardised using the Prostate Imaging – Reporting and Data System (PI-RADS; Table 2). This system improves consistency and objectivity in reporting, and has been prospectively validated.13,14
Overall, mpMRI in the hands of an experienced uro-radiology team, is emerging as a useful tool in the diagnosis and to help guide treatment of prostate cancer. It must be stressed that mpMRI is in its infancy and results can be variable, depending on the radiologist’s experience, the MRI scanner itself and software platform. At this time we believe mpMRI cannot replace prostate biopsy and that MRI should only be ordered by the treating urologist. We now outline the role of mpMRI in different stages of the diagnostic and treatment pathway.
Table 2. Explanation of the PI-RADS scoring system
|
|
Overall PI-RADS score
|
Likelihood of the presence of a clinically significant cancer
|
|---|
|
1
|
Extremely unlikely
|
|
2
|
Unlikely
|
|
3
|
Equivocal
|
|
4
|
Likely
|
|
5
|
Extremely likely
|
|
A score between 1 and 5 is given by the radiologist for each parameter in a region of interest based on objective findings. An overall score reflecting the likelihood of prostate cancer is then given for each region
of interest
|
Role in detection
When prostate biopsy is indicated, a pre-biopsy mpMRI can identify areas suspicious for cancer. Performing MRI prior to TRUS biopsy avoids post-biopsy changes of haemorrhage, inflammation and fibrosis, which can mimic tumours.11 Extra cores can be taken of suspicious areas in addition to the standard biopsy template, thus improving biopsy accuracy. When ultrasonography is used to perform the biopsy, this technique is called cognitive fusion (Table 3). MRI-guided prostate biopsy allows for in-bore MRI biopsy of any suspicious area shown on the MRI. MRI–TRUS fusion technologies are becoming available where MRI images are superimposed on real-time ultrasound images used to perform the biopsy. This has the benefit of not requiring real-time MRI during the biopsy.
In the future, MRI could be a possible second-line screening tool in men with a mildly elevated PSA and normal DRE, without other risk factors. If the MRI is deemed ‘low-risk’, these men could be followed with serial PSA and DRE rather than biopsy. At present there is insufficient evidence to justify this approach. Until further evidence is available it would be hazardous to omit TRUS biopsy on the basis of MRI.11
Table 3. MRI guided biopsy techniques
|
|
Cognitive TRUS-guided biopsy
|
- Urologist reviews MRI images, correlates this with real-time TRUS images and performs a ‘free-hand’ TRUS-guided biopsy of the MRI suspicious region
- Advantages:
- Simple
- No speciailised, expensive equipment required
- Uses standard biopsy technique
- Disadvantages:
- Larger margin of error
- No guarantee the manual biopsy will sample the MRI-suspicious region
|
|
In-gantry
(real time) MRI-guided biopsy
|
- An MRI-compatible biopsy device is inserted into the patient (under local anaesthetis +/– sedation) and repeated T2-weighted imaging performed until the biopsy trajectory is centred on the suspicious region. Biopsy needle is then deployed and images taken to confirm the biopsy needle has sampled the suspicious area
- Advantages:
- Guarantees sampling of the MRI suspicious area
- Disadvantages:
- Longer procedure time (1–2 hours)
- High cost
- Resource-intensive, requires prolonged access to MRI scanner
|
|
MRI-TRUS fusion-guided biopsy
|
- This is a hybrid of the two techniques above. MRI images are downloaded onto an ultrasound machine with special software. Ultrasound images are acquired and the software fuses the MRI onto corresponding USS images and coordinates the biopsy template to guide biopsy of the suspicious region
- Advantages:
- Fairly accurate sampling
- Cost between the 2 previous techniques
|
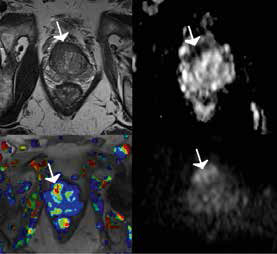 |
Figure 1. Multiparametric images of an anterior prostate tumour
Left upper corner: T2 weighted; right upper corner: apparent diffusion coefficient map; left lower corner: dynamic contrast enhanced map; right lower corner: diffusion-weighted image map |
Post-negative biopsy
The scenario of a rising PSA after one or more negative biopsies is the best established role of MRI, which localises a region suspicious for prostate cancer in over 30% of such patients.15 These tumours are typically found in the anterior prostate (Figure 1). To minimise post-biopsy changes, an interval of at least 8 weeks after biopsy is recommended before performing an MRI.11
Role in selection of patients for treatment
Distinguishing patients who will benefit from treatment from those who will not is a crucial step in the assessment of patients with prostate cancer. Gleason grade is the most important parameter when determining risk of mortality.4 mpMRI has particular accuracy in identifying cancers of Gleason grade 7 and above (ie intermediate and high-risk cancers). Table 4 explains risk stratification of prostate cancer.2,16 A recent study found mpMRI has a sensitivity of 87–100% for detection of Gleason 4 disease. However, the same study found that the specificity of mpMRI was much lower at 40%.15 The lower specificity is in part due to the range of benign conditions that can mimic the appearance of prostate cancer on MRI (Table 5). mpMRI could also reduce the detection of clinically insignificant prostate cancer as mpMRI has reduced sensitivity for low grade, low volume cancers.8
Table 4. Risk stratification of prostate cancer
|
|
Risk level
|
Criteria
|
|---|
|
Low
|
- DRE: impalpable disease or occupying less than half of one lobe of the prostate
AND
AND
|
|
Intermediate
|
- DRE: tumour involving more than one-half of one lobe of the prostate
OR
OR
|
|
High
|
- DRE: tumour involving both lobes or extending through the prostate capsule
OR
OR
|
|
DRE, digital rectal examination; PSA, prostate-specific antigen
|
Table 5. Differential diagnosis for low signal intensity regions on T2 weighted imaging
|
- Prostate cancer
- Haemorrhage
- Prostatitis
- Fibrosis
- Atrophy
- Radiotherapy
- Hormonal therapy
|
Role in active surveillance
Active surveillance is a management strategy used in selected patients to reduce prostate cancer over-treatment. Men with low-risk prostate cancer may be suitable.5 Patients are closely followed with regular DRE, PSA and repeat prostate biopsies. Curative treatment is initiated if disease progression occurs. The main limitation is being able to accurately classify a prostate cancer as low risk. Up to one-third of men commenced on active surveillance have upgrading of disease (eg from Gleason 3+3 = 6 to 3+4 = 7 or higher) on serial biopsy.17 In some men, this may represent true progression of cancer; however, in most cases it reflects inadequacy of the initial biopsy.
The ability of mpMRI to detect intermediate and high-risk cancers may aid in the selection of patients who can safely pursue active surveillance by identifying unsuitable patients who harbour high-grade or larger tumours that have been missed on biopsy.2 MRI may also have a role in monitoring patients, although further data are required before mpMRI can replace the regular biopsies recommended in active surveillance protocols.8
Role in treatment
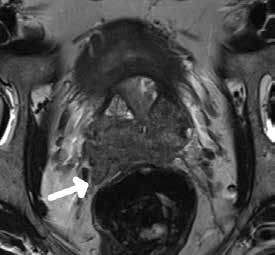 |
Figure 2. T2-weighted image showing a large high-grade prostate cancer with extraprostatic extension invading the neurovascular bundle
This patient would have a positive margin if aggressive nerve sparing surgery was performed |
Accurately locating extracapsular extension on MRI may aid in determining suitability of patients for nerve sparing radical prostatectomy without compromising surgical margin status (Figure 2).11 MRI may also have a role in avoiding side effects from radiotherapy if the tumour can be accurately identified and dose escalation performed for the particular region.8 mpMRI is also emerging as a useful modality when detecting recurrent prostate cancer following curative-intent radiotherapy.18
MRI- guided biopsy
MRI-guided biopsy may aid in the diagnosis of patients with a negative biopsy after a suspicious lesion is found on mpMRI. Biopsies are taken with real-time MRI to ensure the suspicious lesion is sampled. This is time-consuming, costly and available at a very limited number of centres throughout Australia. At present MRI-guided biopsy is a second-line technique that should only be requested by the treating urologist after standard biopsies have been performed.
Future possibilities
Focal therapy is a minimally invasive therapy involving the localisation and ablation of an area of cancer while sparing the remainder of the prostate.11 MRI may be of value when guiding focal treatment and follow-up. MRI-guided focal laser treatment for prostate cancer is a potential future technique in low-risk patients that is currently being explored by the authors. Focal therapy is experimental at this stage, but could be a promising treatment option in the future.8
Limitations of MRI
Reliable comparison of the literature is difficult because of differences in technical conduct of scans, interpretation and reporting. The combination of anatomical and functional parameters varies depending on indication, so different protocols are recommended for detection, compared with tumour and distant staging.11
At present, MRI of the prostate is not rebatable by Medicare. The Urological Society of Australia and New Zealand is lobbying the government to obtain Medicare rebates for MRI of the prostate and it is hoped that rebates will be available in the near future.
Conclusion
mpMRI is emerging as a very useful tool in the diagnosis and treatment of prostate cancer. Although it is an expensive technology, we believe it can impart significant cost savings by reducing the number of prostate biopsies performed, as well as improving patient care by aiding more rapid and accurate diagnosis.8 There can be improvements in the surgical margin rate, which also reduces the risk of future recurrences and the need for adjuvant or salvage radiotherapy. Further investigation is required to determine the optimal techniques, indications and interpretation of mpMRI.12 Presently, mpMRI cannot replace prostate biopsy and should only be ordered by urologists as part of an overall prostate cancer risk assessment. mpMRI should be performed by specially trained radiologists with adequate MRI technology using a standardised reporting system.11
Key points
- mpMRI can assess the prostate and identify regions of interest that may be cancers. These lesions are given a likelihood of being malignant using a standardised reporting system (PI RADS).
- The best established role of mpMRI is to guide biopsy in patients with a high clinical suspicion of prostate cancer but negative biopsy(s).
- MRI can be performed pre-biopsy where it can improve the accuracy/detection of prostate cancers.
- MRI can improve nerve sparing while maintaining margin status by identifying extraprostatic tumour extension.
- MRI-guided biopsy and MRI-TRUS fusion technologies are beginning to become available.
- A 3-Tesla MRI scanner with an experienced radiographer and radiologist is essential.
- The role of MRI remains experimental and further evaluation is required. At this time MRI cannot replace prostate biopsy and should only be ordered by the treating urologist.
Competing interests: None.
Provenance and peer review: Not commissioned, externally peer reviewed.