In the setting of a current reaction, it is important to make a thorough assessment of the nature and severity of the ADR, and where not obvious, refer to available information on the known side-effects of the medication in question. Where allergy is suspected, testing may be helpful. For example, serum tryptase in the case of suspected anaphylaxis, eosinophilia in a chronic reaction, and skin biopsy in the case of a rash. However, it is usually not possible to test for drug allergy during the reaction, and the drug under suspicion (usually the most recently started) will need to be stopped. In these cases, the medication thought to be causing the allergy will need to be stopped.
When a past reaction is described, verification and details should be sought from previous medical reports or contemporaneous records from other sources.
Table 1. Details of adverse drug reactions to be noted in the clinical record
| Detail | Reason |
|---|
| Generic and trade name of drug associated with adverse drug reaction |
Trade name relevant – excipients may rarely be involved in the reaction |
| Dose, frequency and route of drug |
Side-effects, intolerance reactions may be dose-related |
| Timing of onset and offset of reaction in relation to commencement and cessation of drug |
To assess likelihood of causality |
| Reaction description |
May allow classification of adverse drug reaction type |
| Reaction severity, management required (eg. additional medications, hospitalisation) |
May determine level of future contraindication |
| Original indication for use of drug (eg. infection) |
Could this have caused the reaction rather than the medication? |
| Cofactors (eg. fever, other simultaneous medications) |
The reaction may have been caused by an interaction between cofactors and the drug in question |
Record in the patient file as much detail as possible regarding the ADR, rather than simply ‘allergy to drug X’ (Table 1). This will enable an assessment of the risk of future use of the drug and related medicines. The nature of the ADR may allow classification according to mechanism (Table 2, Table 3).
Table 2. Terminology of adverse drug reaction mechanisms
| Classification | Mechanism |
|---|
| Allergy (= hypersensitivity) |
Immunological |
| Side-effect |
Pharmacological |
| Intolerance (= sensitivity) |
Pharmacological plus susceptibility; unknown |
| Other terms |
|---|
| Idiosyncrasy |
Genetic susceptibility to adverse drug reaction |
| Pseudo-allergy |
Various mechanisms – mimics allergy |
| Toxicity* |
Excess dosage |
| Interaction* |
Drug combinations |
| * Not further discussed in this article |
Table 3. Examples of mechanistically different adverse drug reactions
| Allergy | Side-effect | Intolerance |
|---|
| Enalapril – rash |
Enalapril – cough |
Enalapril – hypotension |
| Codeine – anaphylaxis |
Codeine – constipation |
Codeine – hallucinations |
| Nortriptyline – rash |
Nortriptyline – dry mouth |
Nortriptyline – postural hypotension |
Allergy
Allergic ADRs are a distinct subgroup because the mechanisms (if not the reason for their occurrence) are well understood. The classic system of Gell and Coombs1 (Table 4) still provides a good basis for understanding drug hypersensitivity reactions, although the vast majority of reactions fall into either type 1 (acute IgE mediated: urticaria or anaphylaxis) or type 4 (cell mediated: delayed rash or organ pathology). Type 4 has recently been divided into four subtypes according to the dominant effector cells: broadly, 4a – macrophage, 4b – eosinophil, 4c – cytotoxic T-cell and 4d – neutrophil.2 Each of these can be represented by a specific cutaneous drug allergy reaction (Table 5).
Table 4. Features of drug allergy
Mechanism (immunological)
(Gell and Coombs) |
- Type 1 – IgE mediated (acute allergy)
- Type 2 – antibody-dependent cytotoxicity
- Type 3 – immune complex
- Type 4 – cell mediated (delayed hypersensitivity)
|
| Natural history |
- Prior exposure required for sensitisation
- Tends to become worse on repeated exposure
- Often long lasting due to immunological memory
- Cross-reaction based on structure of drug (not action)
|
| Clinical features |
- Rash
- Angioedema
- Anaphylaxis
- Organ inflammation – lung, kidney, liver
|
Table 5. Extended Gell and Coombs classification
| Type | Effector cells and cytokines | Examples |
|---|
| Type 4a |
Macrophage, IFN gamma (Th1) |
Contact dermatitis
Maculopapular drug rash |
| Type 4b |
Eosinophil, IL-5 (Th2) |
Urticaria (delayed)
DRESS (drug rash eosinophilia systemic symptoms) |
| Type 4c |
Cytotoxic lymphocytes |
Toxic epidermal necrolysis
Stevens-Johnson syndrome |
| Type 4d |
Neutrophils, IL-8 |
Acute generalised exanthematous pustulosis |
Idiosyncrasy is a term used to refer to an individual’s susceptibility to a drug ADR based on genetic variation. Genetic susceptibility may predispose to side-effects, intolerance reactions or allergic reactions (Table 7). In these cases, genetic testing prior to drug administration will reduce the risk of ADRs.
Pseudo-allergy refers to adverse reactions that produce clinical features that mimic allergy but are not mediated by immunological mechanisms. Inhibition of angiotensin converting enzyme can lead to an excess of tissue bradykinin triggering angioedema, which is often thought of as an allergic disorder but in this case is related to the pharmacological effect of the drug. There may be an element of idiosyncrasy because there may be an underlying genetic polymorphism in those who are susceptible to this side-effect.3
The principles of the natural history of drug allergy reactions are shown in Table 4. Prior tolerance of a drug is not evidence against allergy to the drug, indeed, prior exposure is necessary for sensitisation. It is safest to assume that where an allergic reaction has occurred, it is most likely to occur again on subsequent exposure, and the severity of the reaction may be similar or worse. However, there are frequent exceptions; reactions may sometimes be of lesser severity, particularly after a prolonged interval, and in some cases, careful testing reveals that the allergy has resolved and the patient may safely receive the drug. Accidental re-exposure sometimes occurs and with luck, is tolerated without reaction, but deliberate re-exposure is not recommended unless careful consideration is given to the risks.
Cross-reactivity is when an immunological reaction develops specifically to drug 1, but because of similarity in molecular structure, the antibodies and/or T-cells also react against drug 2 (even without any previous exposure to drug 2). This allows some prediction of risk of reactivity to structurally related drugs. A proportion of patients will have a highly specific immune response and react only to drug 1 whereas others have a less specific response and react to both drug 1 and drug 2; the percentage of patients who react to both can be expressed as the cross-reactivity risk. For example, virtually all penicillin allergic patients will react to amoxycillin because both drugs have a beta-lactam/thiazolidine core, but only a small proportion will cross-react with cephalosporins which have a beta-lactam/dihydrothiazine core.
As the allergic reaction is mediated by a limited range of immunopathogenic mechanisms, diverse drugs can cause similar types of reactions, depending on which immunological effectors are activated. For example, an IgE-mediated anaphylactic reaction will be similar regardless of which drug triggers it.
A patient who gives a history of a drug allergy may not be allergic for several reasons:
- the reaction may have been due to the disease not the drug, or a combination of drugs, or a different drug than the one to which it was attributed
- it may be a conditional ADR in which the reaction only occurs with particular cofactors
- it may be a misattribution because the drug has been misremembered, or a parent has the allergy and believes their child will also be allergic, or the child’s sibling actually had the allergy
- in some cases, the nature of the reaction has been exaggerated; it may have been a mild intolerance or minor side-effect that is now called an allergy
- it is possible that there was a genuine and significant allergic reaction but the allergy has resolved due to the passage of time.
Tests are available for the diagnosis of drug allergy but they are limited. Specific IgE for some drugs can be detected on blood testing (Figure 1). Specialist referral for evaluation of drug allergy may be indicated (Table 6) as additional procedures may clarify the diagnosis (Figure 1).
Table 6. Indications for referral to a clinical immunology and allergy specialist
|
Drug allergy history uncertain and/or allergic reaction occurred in the distant past
AND
Indication to use the drug to which the patient is thought to be allergic and alternatives are not available or not optimal
OR
For consideration of desensitisation, where drug allergy history is definite or confirmed but there is a strong indication to use the drug
|
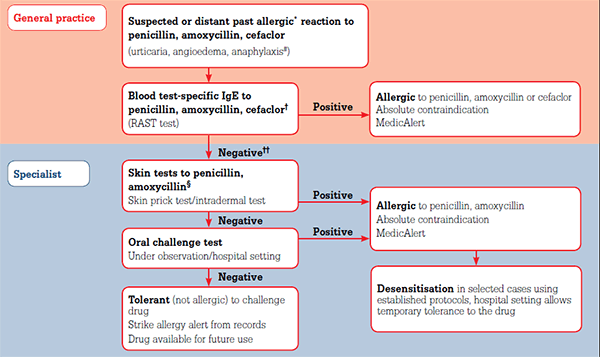
Figure 1. Drug allergy testing
* If allergic reaction is recent and clinically highly likely, no need for testing
# Blood tests, skin tests valid only for type-1 (IgE-mediated) acute allergic reactions
† Blood tests are currently only available routinely for these drugs, not for the majority of other drugs
†† Negative blood test does not exclude allergy, requires further evaluation, refer to clinical immunology and allergy specialist
§ Skin testing – not well validated for cephalosporins, may proceed directly to graded-dose challenge depending on risk/benefit assessment
This general schematic may vary in some specialist clinics depending on availability of research protocols and local expertise
Other drugs can be tested by graded-dose oral or parenteral challenge in appropriate circumstances
Side-effect
Side-effects are ADRs which do not depend on an immunological reaction against the drug but on its pharmacological effects. They are more common and usually well recognised for each particular drug. The reasons why only some individuals suffer from side-effects may relate to variation in the metabolism of the drug and/or the production of active metabolites, however, the mechanisms for individual variations in response are often unknown. Susceptibility factors may be intrinsic (such as genetic polymorphisms, age, gender, race) or acquired (such as interacting drugs, illness, hepatic or renal dysfunction). Some side-effects are an absolute contraindication to future use of the drug, whereas in other cases the drug could be cautiously re-tried. If a side-effect occurs to a particular drug, it is likely that other drugs with a similar pharmacological action (eg. within the same therapeutic class) will produce the same effect so therefore should usually also be avoided.
Intolerance
Intolerance (sensitivity) is a poorly defined term that may refer to an unusually low threshold to the pharmacological side-effect of a drug (eg. hyperemesis with codeine may be referred to as opiate intolerance) or to an adverse reaction that might be atypical or not understood according to pharmacology. For example, patients may feel non-specifically unwell on a medication. In practice there is a considerable overlap between the terms intolerance and side-effect and it is often difficult to classify ADRs into one of these categories.
Table 7. Idiosyncrasy – genetic predisposition to adverse drug reactions
| Genotype | Drug | Nature of reaction | Classification of reaction |
|---|
| Thiopurine methyltransferase (TPMT) |
Azathioprine |
Myelosuppression |
Side-effect |
HLA-B*5701
HLA-B*1502 (Han Chinese) |
Abacavir
Carbamazepine |
Hypersensitivity syndrome
Stevens-Johnson syndrome |
Allergy |
Non-steriodal anti-inflammatory drugs (NSAIDs) can produce both true allergic reactions and pharmacological reactions (referred to by convention as NSAID intolerance or aspirin intolerance) (Table 8). This is important because in a case of allergy to a specific NSAID, another NSAID can be used, whereas in NSAID intolerance, all COX-1 inhibitory NSAIDs will need to be avoided.4
Table 8. NSAIDs – allergic reactions or pharmacological intolerance4
|
NSAID (specific drug) allergy
|
Anaphylaxis to one particular NSAID (eg. diclofenac, celecoxib)
Tolerance of other NSAIDs
|
Probably IgE mediated; clinical characteristics of an immunological reaction
|
|
NSAID intolerance
|
Urticaria, angioedema or asthma to multiple NSAIDs
|
Pharmacological; inhibition of COX-1, increased leukotriene production
|
Conclusion
In conclusion, there are two parts to the patient ADR record:
- the detailed description of the prior ADR, and
- an interpretation of this to decide the risk of future use of the same or related drugs.
Unfortunately, some medical software packages do not allow detailed ADR information records, but only have the capacity to record ADRs as an allergy.
Future use of a drug when an ADR has been recorded could fall into one of three categories:
Drug can be used with caution: Risk of ADR is low but not negligible, and/or any ADR that could occur, based on prior reactions, is very unlikely to be dangerous. An observation period after the first dose may be advisable, or the patient warned to stop the drug if symptoms occur. In some cases it might be possible to ‘treat through’ side-effects or counteract them with other medications
Relative contraindication: There is a moderate risk of an ADR but the risk of a dangerous reaction is low; use only if the indications are strong and there are no suitable alternatives; observe patient or warn of possible side-effects; treat through if possible
Absolute contraindication: Risk of a dangerous adverse reaction is high; in some cases it might be possible to use the drug under specialist hospital management (using desensitisation protocols) but otherwise the drug should not be used.
Table 9 lists examples of drug recommendations based on ADR history.
A MedicAlert is indicated if the ADR was severe and the risk is moderate or high, particularly for drugs which might be used in emergency management and/or are given parenterally, whereas the indication for a MedicAlert is not as strong where the reaction has been mild or moderate and for drugs which are used electively.
Table 9. Examples of drug recommendation based on adverse drug reaction history
| Previous adverse drug reaction | Recommendation* |
|---|
| Anaphylaxis to penicillin |
Absolute contraindication penicillins, cephalosporins |
| Rash to amoxycillin (benign) |
Relative contraindication amoxycillin
Caution cephalosporins |
| Angioedema to enalapril |
Absolute contraindication ACEIs
Caution angiotensin-2 receptor blockers |
| * Advice may be modified after specialist testing |
Competing interests: None.
Provenance and peer review: Commissioned; externally peer reviewed.