Several patients have recently presented with a delayed diagnosis of acral melanoma (AM) at the Melanoma Institute Australia (MIA). MIA treats more than 1500 new patients with melanoma each year and 1% of these have AM. AM often has a poor prognosis and the purpose of this article is to raise awareness of AM to facilitate early diagnosis and treatment with the ultimate aim of improving patient outcomes.
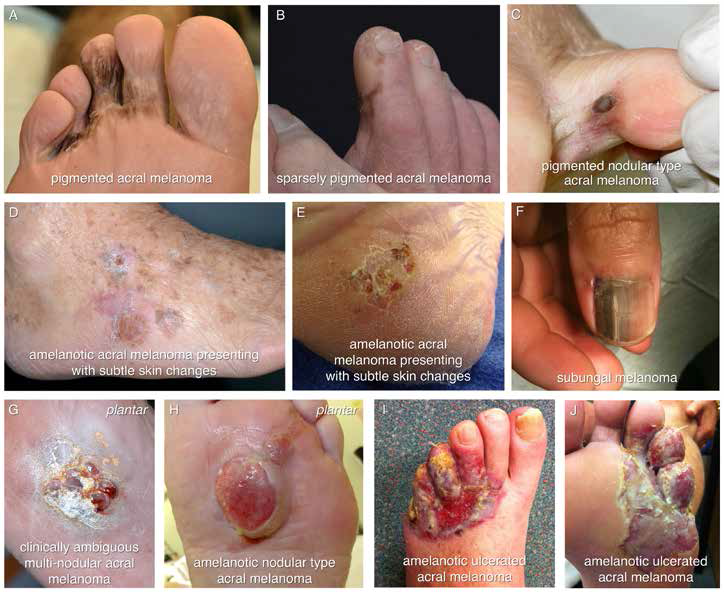 |
| Figure 1. The diverse clinical appearances of acral melanoma |
Case presentation
A 62-year-old man presented with a 2-year history of left foot symptoms. At presentation to MIA he had a reddish ulcerated lesion centered at the fourth toe and extending across the lateral three web spaces on both the plantar and dorsal surfaces of the foot. The lesion was amelanotic (Figure 1 I, J). His symptoms were of itch, pain and intermittent contact bleeding. He had seen multiple doctors and had been using topical antifungal treatments for almost 2 years. Partial biopsy demonstrated an intermediate thickness melanoma with a Breslow thickness of 2.5 mm. Adverse features included ulceration and a mitotic count of 1/mm2. Clinically, there was no evidence of in-transit metastasis (metastases in the skin or subcutis between the primary tumor and the regional lymph nodes), no palpable regional lymphadenopathy and a staging positron emission tomography–computed tomography (PET–CT) scan showed no evidence of distant metastatic disease. Complete excision required forefoot amputation and the final pathology was similar to that of the biopsy. The regional lymph node field, identified as the left groin by lymphatic mapping, was evaluated with sentinel node biopsy. One of two sentinel nodes contained metastatic melanoma. The patient subsequently underwent a completion left groin lymph node dissection and no further metastatic disease was identified in the operative specimen.
Discussion
Patients with AM most commonly present with lesions that have changed in size, colour or form, or that are bleeding, painful or itchy.7 The diagnosis of AM is often delayed because the patients have attributed their symptoms to much more common benign conditions, the most common being warts, fungal infections, haematomas and ulcers.7 When these benign pathologies fail to resolve with appropriate management the diagnosis of AM must be excluded. Other patients have failed to appreciate the presence of pathology on their soles, which may not have been inspected recently.
The aetiology of AM is poorly understood. Risk factors such as fair skin type, ultraviolet (UV) exposure, the presence of acral nevi and trauma may be identified in a patient’s clinical history, but have not been consistently associated with AM.7–10
Clinically, AM can be pigmented and appear as a classical melanoma with asymmetry, border irregularity, colour variation, a diameter >6 mm and evolution of the lesion (using the ABCDEs) (Figure 1 A, B and C). However, a large proportion of AM is amelanotic, appearing pink in colour, and making diagnosis much more difficult. It can be as subtle as a change in skin texture (Figure 1 D, E), appear pink and nodular (Figure 1 G, H) or ulcerated (Figure 1 I, J). As a consequence, AM can easily be misdiagnosed and a high index of suspicion is needed when managing lesions of the feet and hands. Subungual melanoma most commonly presents as longitudinal melanonychia (a brown-black stripe along the length of the nail) (Figure 1F)
but may also be amelanotic.
Dermatoscopy is an adjunct to clinical examination that can be used by clinicians experienced in the technique to assist with the diagnosis of AM. Specific dermatoscopic features are associated with pigmented lesions on the glabrous skin of palms and soles. Parallel ridge pattern and irregular diffuse pigmentation are findings that are highly specific for melanoma and can assist in differentiating AM from benign acral naevi.11 Dermatoscopy has not been well validated in the diagnosis of non-pigmented AM and biopsy should be performed when there is clinical concern.
The prognosis of AM is generally poor and this has often been attributed to delayed diagnosis. However, in a recent series, AM patients were stage matched with patients with non-acral cutaneous melanoma (NACM) of the limb and AM patients were shown to have a poorer disease specific survival (5-year survival 70% for AM compared to 83% for NACM)6 suggesting that AM is biologically more aggressive. This also correlates with a different spectrum of genetic mutations in AM compared to NACM. BRAF mutations occur in approximately 50% of all melanomas but are less common in AM. In contrast, KIT mutations, which are rare in sun-damaged skin, occur in only 3% of all melanomas, but have been identified in 11–23% of AMs.12,13
Early diagnosis prior to metastasis will provide the best chance of long-term survival in AM patients and diagnosis requires appropriate biopsy. The Clinical Practice Guidelines for the Management of Melanoma in Australia and New Zealand recommend that skin lesions suspected of being a melanoma be excision biopsied with 2 mm margins,4 but this can be difficult or inappropriate in the setting of acral lesions. The index of clinical suspicion is often low and the lesion often large. Consequently, an excision biopsy would cause significant functional impairment. Incision or punch biopsy techniques can be useful and the biopsy(s) should be taken from the area of greatest clinical concern and a peripheral non-necrotic area.4 When the index of suspicion is low and treatment has been initiated for a probable benign diagnosis a biopsy should be performed if the pathology fails to improve after one or two courses of appropriate treatment. The assessment and biopsy of subungual pathology can be complex and should be referred to specialist units. Specialist referral before biopsy may also be considered when the level of suspicion is high.
The diagnosis of AM should prompt referral to a specialist surgical oncologist. Treatment includes complete excision of the primary with an adequate margin, evaluation of the regional nodes and, sometimes, systemic staging. Wide excision of the primary site often requires skin grafting or digital amputation. In the absence of clinical regional lymph node metastases sentinel node biopsy is used to assess for microscopic nodal metastases. Microscopic or clinically detectable regional lymph node metastases are often treated with radical lymph node dissection. For high-risk lesions or if there is clinical concern PET-CT is used to identify distant metastases.
Conclusion
AM is an uncommon melanoma subtype. The clinical appearance of AM is often atypical and this can delay recognition and diagnosis. Clinicians need to maintain a high index of clinical suspicion and arrange appropriate biopsy to facilitate early diagnosis and treatment so as to give AM patients their best chance of cure.
Competing interests: None.
Provenance and peer review: Not commissioned, externally peer reviewed.